Clear Sky Science · en
Geographic spillover of antimicrobial resistance from mass distribution of azithromycin
Why This Matters for Children’s Health
Azithromycin, a common antibiotic, has been used in mass drug campaigns to save the lives of young children in parts of sub-Saharan Africa, including Niger. These campaigns have cut child deaths but raised a serious worry: could they also help drug‑resistant bacteria spread from treated villages to neighboring communities that never receive the medicine? This study asks whether such "spillover" of resistance is happening between villages, a key question for countries weighing the benefits and risks of expanding these programs.
Life‑Saving Campaigns and Their Hidden Risk
Earlier large trials showed that giving azithromycin twice a year to all young children in high‑mortality areas reduces deaths by about 14–18 percent. That success prompted the World Health Organization and governments like Niger’s to consider wider use of this strategy. Yet antibiotics do not just kill harmful bacteria; they also favor the survival of strains that can resist them. In previous work, the same research group saw clear signs of azithromycin‑resistant bacteria in treated children. What was not known is whether these resistant microbes stay put in treated villages or travel outward, undermining antibiotic effectiveness for others.
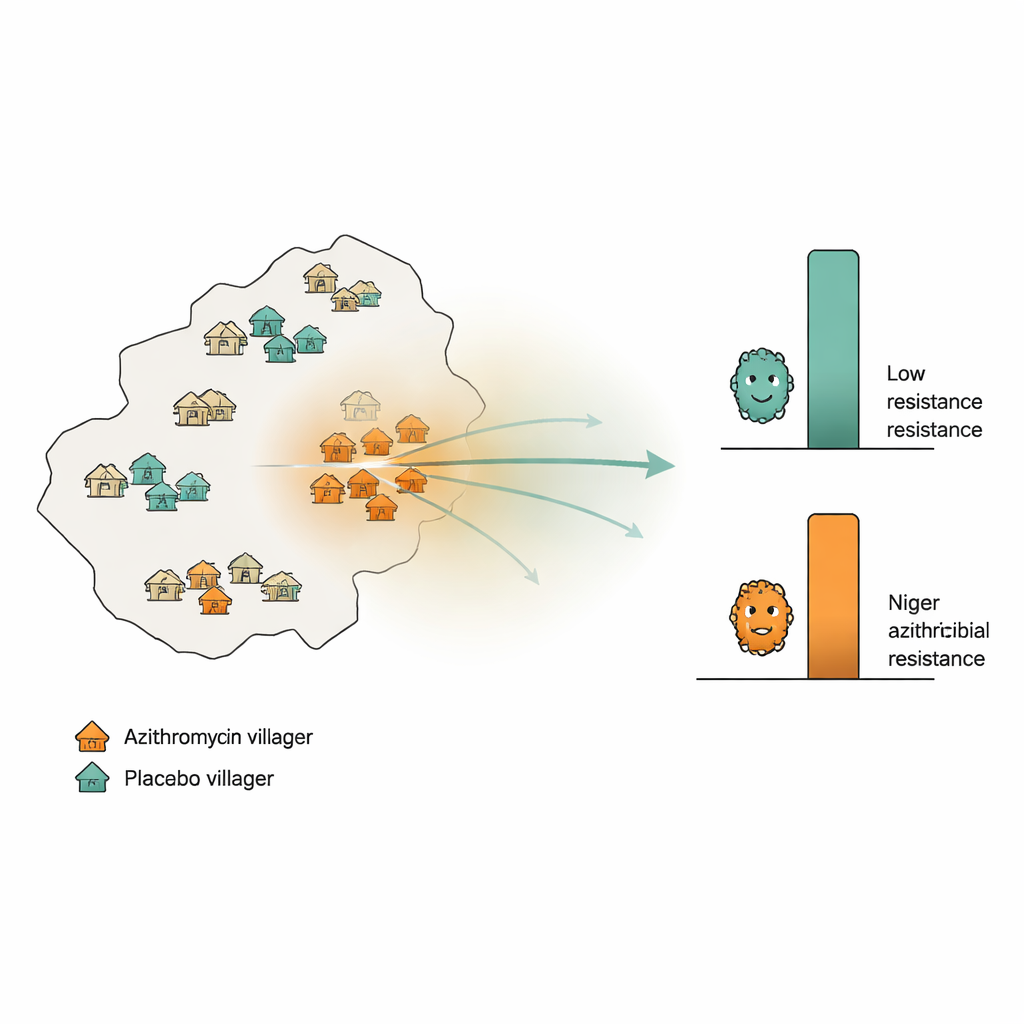
Tracking Resistance Across a Rural Landscape
The researchers took advantage of a major child‑survival trial in Niger’s Dosso region, where 594 villages had been randomly assigned to receive azithromycin or an inactive placebo twice yearly for two years. In addition, 30 separate "monitoring" villages were randomly chosen: half received azithromycin, half placebo. From these monitoring villages, the team collected rectal swabs from 10 children per village at the start of the trial and again after 24 months. Using deep DNA sequencing, they measured how many genes linked to resistance against macrolide antibiotics (the class that includes azithromycin) were present on average in each village. They also calculated, for every monitoring village, how intense azithromycin use was in the surrounding area—counting all doses given in nearby villages and giving more weight to villages that were closer.
Looking for Signs of Spread Between Villages
If resistance were spilling over geographically, the researchers expected to see higher levels of resistance genes in placebo villages that happened to be surrounded by heavily treated neighbors, compared with placebo villages in areas with little azithromycin use. They tested this idea in several ways: by looking at simple rank‑based correlations between nearby treatment intensity and resistance; by repeating the analysis across different distance bands (within 10, 20, or 30 kilometers); and by using a rigorous permutation strategy that shuffled treatment labels in the larger trial to see whether any apparent patterns could arise by chance. They also ran a "negative control" analysis that repeated the same calculations using placebo doses instead of azithromycin doses; any similar pattern there would suggest that confounding, not real spillover, was at work.
What the Data Revealed About Resistance
Across all these checks, placebo villages showed low levels of macrolide resistance genes that stayed close to their baseline levels, regardless of how much azithromycin was used around them. Statistical tests found no meaningful link between surrounding drug use and resistance in these untreated communities. In contrast, villages whose own children received azithromycin did show a marked rise in resistance genes over the two‑year period, consistent with direct selection from treatment—but this rise did not systematically grow stronger in areas with higher surrounding treatment intensity. When the researchers examined live pneumococcal bacteria from children’s noses, they again saw no connection between geographic treatment intensity and resistance. Analyses using different mathematical models and alternative definitions of treatment intensity reached the same broad conclusion.
Limits, Caveats, and the Bigger Picture
The study cannot completely rule out small spillover effects, especially given that only 30 monitoring villages were intensively tested, and it did not examine very fine‑scale spread within households or within single villages. It also assumes that distance and number of doses are the main drivers of any spread, and it focuses on a two‑year window; longer‑term effects or different program designs, such as those treating entire communities for eye disease, might show other patterns. Even so, the evidence suggests that in this setting, most of the resistance pressure from mass azithromycin campaigns is confined to the communities that actually receive the drug, rather than silently spreading across the landscape.
What This Means for Future Antibiotic Campaigns
For health officials in countries like Niger, these findings offer cautious reassurance. While mass azithromycin treatments do increase antibiotic resistance within treated villages, this study found no strong signs that such resistance is leaking into neighboring untreated villages over 24 months. That means previous estimates of resistance risk from these child‑survival trials are unlikely to be badly distorted by unrecognized geographic spillover. Policymakers still need to balance the life‑saving benefits of these campaigns against the local rise in resistance, but they can do so with more confidence that, at least in the medium term and at village‑to‑village scales, the problem is not rapidly radiating outward.
Citation: Srivathsan, A., Arzika, A.M., Maliki, R. et al. Geographic spillover of antimicrobial resistance from mass distribution of azithromycin. Nat Commun 17, 2152 (2026). https://doi.org/10.1038/s41467-026-68691-y
Keywords: antimicrobial resistance, azithromycin, mass drug administration, child mortality, Niger