Clear Sky Science · en
Functional and structural insights into interactions between β-Arrestin 1 and Gαs or Gαi1
Why this matters for health and medicine
Many of today’s medicines, from allergy pills to heart drugs and psychiatric treatments, work by targeting a huge family of cell-surface proteins called G protein–coupled receptors, or GPCRs. These receptors pass messages into the cell using two main helpers, G proteins and proteins called beta-arrestins. This study looks at how one beta-arrestin (beta-arrestin 1) directly talks to two types of G proteins inside the cell. Understanding this hidden conversation could help scientists design more precise drugs with fewer side effects by fine-tuning how signals flow inside our cells.
Two major messengers inside our cells
When a hormone or drug binds to a GPCR on the cell surface, it triggers two broad routes of communication. One route uses G proteins, which switch on and off like molecular timers by trading one small chemical (GDP) for another (GTP). The other route uses beta-arrestins, which can both shut down G protein signaling and start their own signaling pathways. For a long time, scientists mainly studied how receptors talk separately to G proteins or to beta-arrestins. More recent work hinted that G proteins and beta-arrestins might also bind to each other or even form large “mega-complexes” with the receptor. But the details of how these proteins connect and what that means for signaling have been unclear.
Measuring who binds to whom
In this study, the researchers focused on beta-arrestin 1 and two G protein “alpha” subunits: Gαs, which usually stimulates cell activity, and Gαi1, which generally dampens it. Using a sensitive binding technique, they tested how strongly purified beta-arrestin 1 and each G protein stick together under different conditions. They found that the key factor is not whether the G protein is “on” or “off,” but whether beta-arrestin 1 is in a more active, flexible form. A version of beta-arrestin 1 missing its tail — which behaves like a loose, active ensemble of shapes — bound strongly to both Gαs and Gαi1. In contrast, the resting form of beta-arrestin 1, or a form locked by a receptor-tail–like peptide, bound weakly or not at all. This suggests that G proteins preferentially recognize beta-arrestin 1 when it samples active-like shapes that may exist after receptor activation. 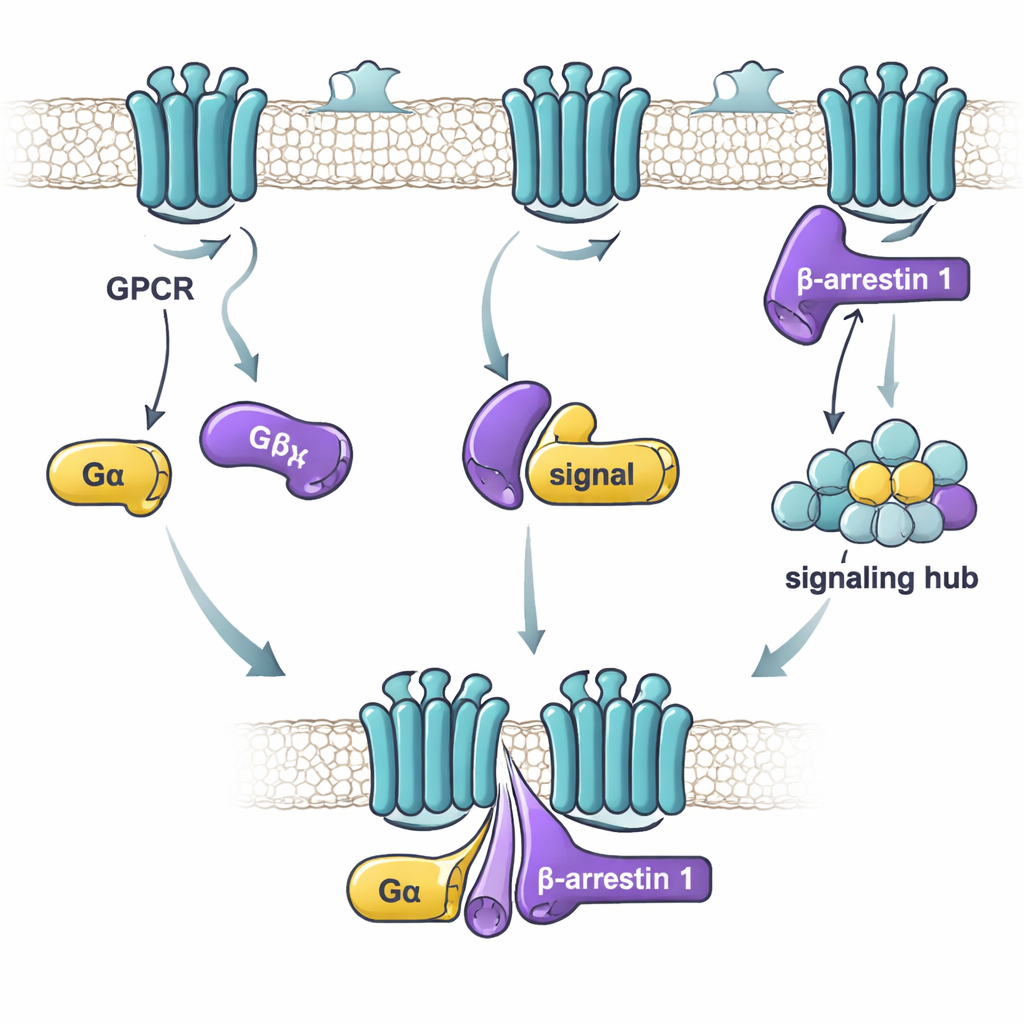
Watching subtle shape changes
To see how binding affects protein shape, the team used a method that tracks how quickly hydrogen atoms on the protein backbone swap with heavy hydrogen from the solvent. Slower exchange usually means a region has become more protected or rigid; faster exchange means it has become more flexible or exposed. When the active-like beta-arrestin 1 fragment bound to Gαs or Gαi1, both G proteins showed clear pattern changes in several regions, indicating they were reshaping around the complex. In contrast, beta-arrestin 1 itself changed only slightly, mainly in flexible loops. By working with isolated domains of the G proteins, the researchers also showed that both the main catalytic “Ras-like” portion and the surrounding alpha-helical portion contribute to binding. For Gαs, the helical region plays a particularly strong role; for Gαi1, both regions matter roughly equally.
Function without flipping the main switch
The next question was whether this direct contact alters how G proteins turn on. The team used a fluorescent GTP mimicking probe that lights up when a G protein switches from GDP to GTP, a standard readout of activation. Surprisingly, adding beta-arrestin 1 did not change the speed or extent of this nucleotide exchange for either Gαs or Gαi1. In other words, beta-arrestin 1 is not acting like a classic “on-switch” enzyme for G proteins. Instead, its influence shows up on its own structure. Using a cleverly engineered beta-arrestin 1 whose tail motion could be tracked by fluorescence, they found that active Gαs, but not Gαi1, boosts the release of beta-arrestin’s C-terminal tail when a receptor-derived peptide is present. In living cells, additional imaging and energy-transfer sensors confirmed that Gαs and beta-arrestin 1 come close together upon hormone stimulation and that Gαs helps push beta-arrestin 1 toward a more active conformation. 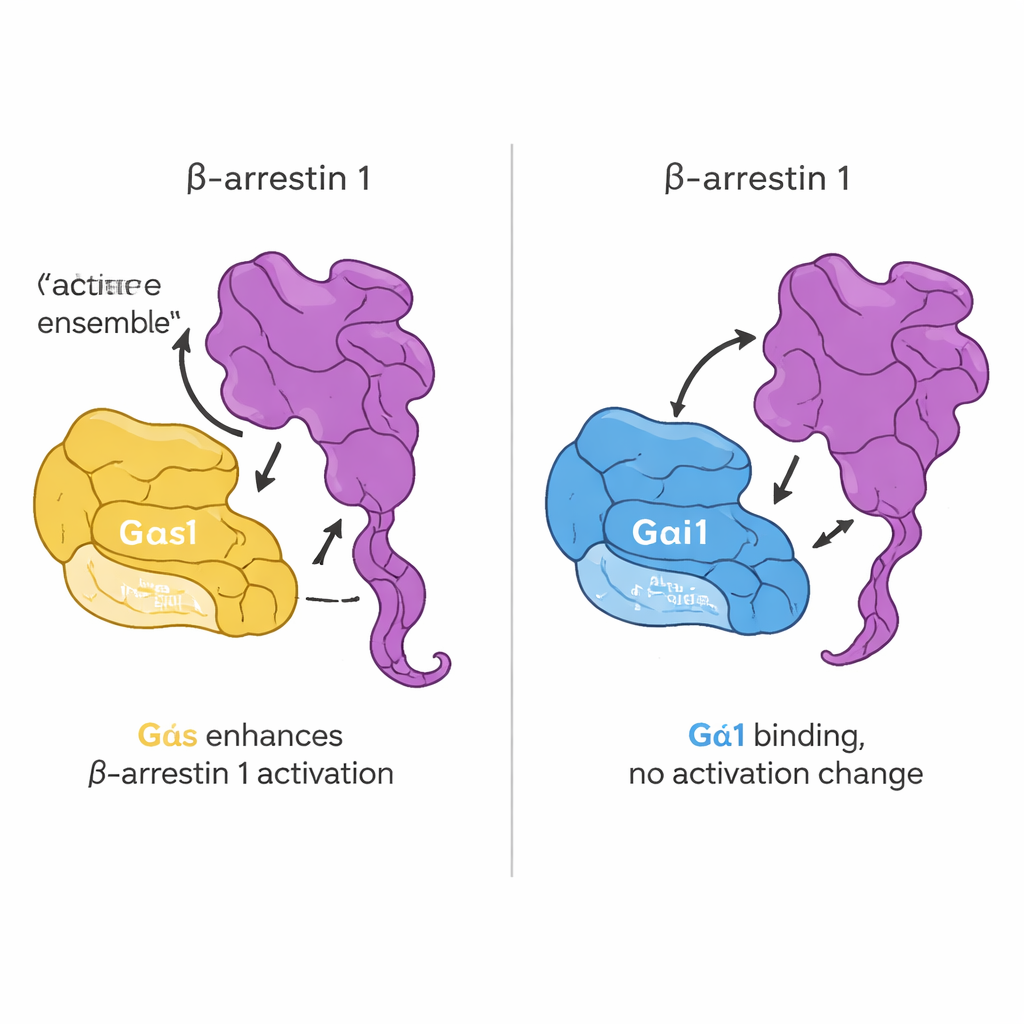
A new layer of control in cell signaling
Putting these results together, the study proposes that Gαs and Gαi1 both bind directly to beta-arrestin 1, but in slightly different ways and with distinct consequences. Neither G protein has its main “on/off” behavior changed by this contact. Instead, especially for Gαs, the interaction acts more like a fine-tuning knob on beta-arrestin 1, nudging it into or stabilizing a more active, tail-released form. This adds a previously underappreciated layer to GPCR signaling: not only can receptors coordinate G proteins and beta-arrestins, but G proteins and beta-arrestins can also modulate each other directly. In the long run, mapping these subtle interactions may help drug designers steer signaling along beneficial routes—enhancing helpful pathways while avoiding those that cause side effects.
Citation: Duan, L., Kim, H., Suh, Y. et al. Functional and structural insights into interactions between β-Arrestin 1 and Gαs or Gαi1. Nat Commun 17, 1879 (2026). https://doi.org/10.1038/s41467-026-68690-z
Keywords: G protein-coupled receptors, beta-arrestin, G protein signaling, cell signaling mechanisms, structural biology