Clear Sky Science · en
Epigenome-wide analysis identifies DNA methylation mediators of treatment-related cardiometabolic risk in survivors of childhood cancer
Why cancer’s after-effects linger in the body
Thanks to better treatments, most children with cancer now grow up. But many of these survivors face new health problems decades later, including obesity, high blood pressure, and diabetes-like conditions. This study asks a pressing question: how do treatments given in childhood leave such long shadows on the heart and metabolism? By looking at chemical tags on DNA in blood cells, the researchers uncover clues that past chemotherapy and radiation may “reprogram” the body in ways that raise long-term cardiometabolic risk.
Tracking hidden risks in grown-up childhood survivors
The team studied nearly 3,000 adults who survived childhood cancer and are followed in the long-term St. Jude Lifetime Cohort. These survivors had been treated with various chemotherapies and radiation to the brain or trunk (chest, abdomen, pelvis) as children, then returned years later for detailed health checks and blood tests. The scientists focused on five key cardiometabolic problems that are known to raise the risk of heart disease and early death: obesity, abnormal blood sugar, high triglycerides, high cholesterol, and high blood pressure. By comparing survivors with and without these conditions, they searched millions of spots across the genome for differences in DNA methylation—tiny chemical switches that help control how genes are turned on or off.
Chemical tags on DNA as a memory of treatment
Across the genome, the researchers found almost 1,900 DNA sites where methylation levels were linked to at least one of the five cardiometabolic conditions. Most of these sites were tied to a single problem, but a small group appeared to be “hubs” connected to several. Five key sites, located near two genes called CPT1A and LMNA, were associated with all five cardiometabolic risks at once. These genes are known to influence how the body handles fats and how cells maintain their structure, hinting that treatment may disrupt shared metabolic and cardiovascular pathways. Many of the methylation changes overlapped with signals previously seen in the general population, especially those related to inflammation, but others seemed unique to cancer survivors—suggesting that treatment leaves its own distinctive biological imprint.
Connecting therapy, DNA changes, and disease
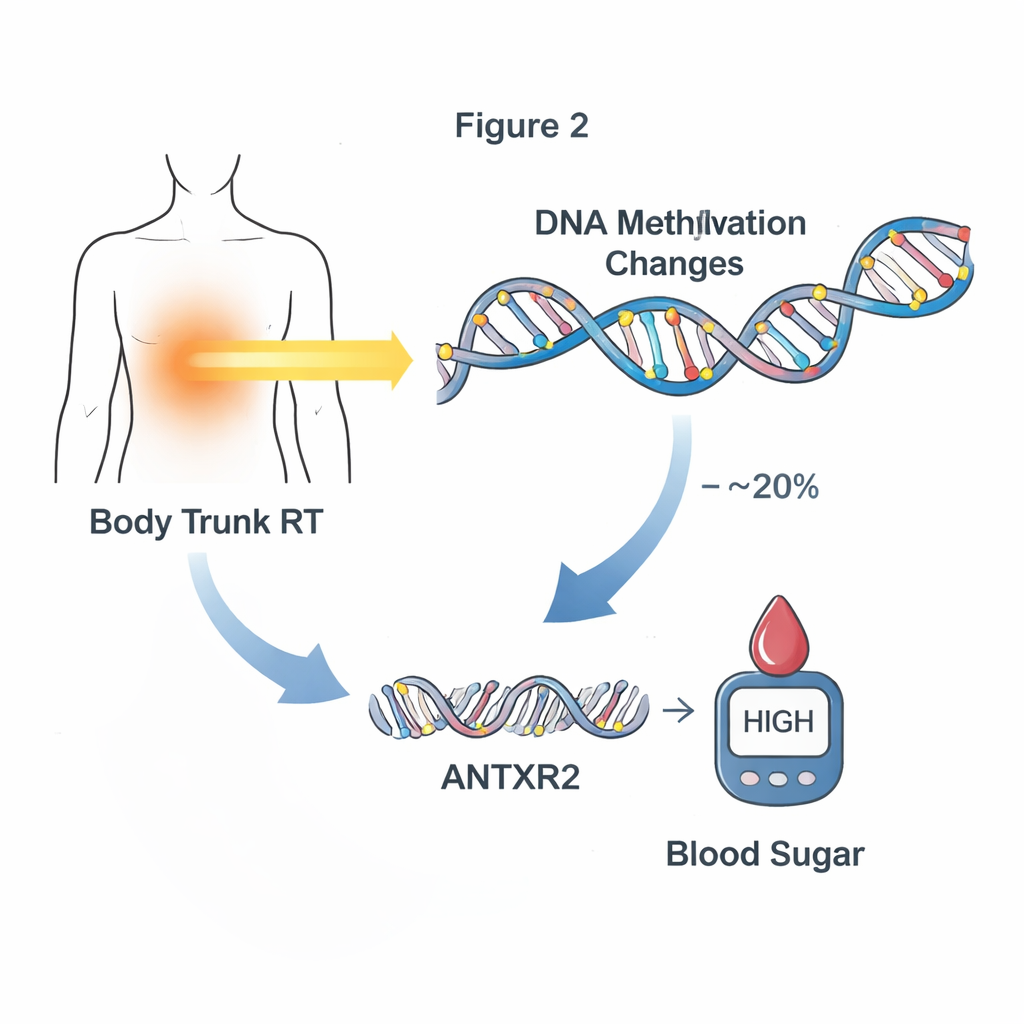
The next step was to test whether these methylation changes actually help explain how treatment leads to later disease, rather than just tagging along. The researchers focused on new or worsening cardiometabolic problems that developed only after the blood sample was taken, preserving the timeline from treatment to DNA change to disease. Using statistical mediation analyses, they identified 24 methylation sites that together explained up to about one quarter of the link between specific treatments and later cardiometabolic problems. Radiation to the brain and trunk showed especially strong patterns: groups of methylation sites together accounted for roughly 10–20% of the impact of these radiotherapies on abnormal blood sugar. One standout site, near a gene called ANTXR2, appeared to mediate about 20% of the effect of trunk radiation on later glucose problems.
From DNA marks to gene activity
To understand how these methylation marks might change biology, the team examined gene activity in a subset of survivors with both DNA and RNA data from their blood. They found dozens of methylation sites where higher or lower methylation was linked to changes in nearby gene expression. Among them was the key mediator site near ANTXR2, where methylation levels were strongly tied to how much of this gene was expressed, especially in survivors who had received trunk radiation. ANTXR2 helps regulate blood vessels and the surrounding tissue framework—systems that influence how glucose and insulin move through the body. This pattern suggests a chain of events in which radiation alters methylation, which shifts gene activity, which in turn may nudge the body toward abnormal blood sugar.
What this means for survivors and future care
For non-specialists, the key message is that childhood cancer treatments do not just damage tissues in the moment; they may also leave long-lasting “notes” on the DNA that shape health for decades. In this study, some of those notes—DNA methylation changes—help explain why certain survivors are more likely to develop obesity, high fats in the blood, or glucose problems linked to diabetes and heart disease. While these chemical tags are only part of the story, they could eventually serve as blood-based biomarkers to identify survivors at highest risk and to test whether lifestyle changes or new therapies can safely rewrite some of this epigenetic history. The work moves survivorship care a step closer to precision medicine: using molecular footprints of past treatment to guide earlier, more tailored prevention of heart and metabolic disease.
Citation: Eulalio, T., Kim, Y., Meng, X. et al. Epigenome-wide analysis identifies DNA methylation mediators of treatment-related cardiometabolic risk in survivors of childhood cancer. Nat Commun 17, 1979 (2026). https://doi.org/10.1038/s41467-026-68689-6
Keywords: childhood cancer survivors, DNA methylation, cardiometabolic risk, radiation therapy, epigenetics