Clear Sky Science · en
Co-translational determination of quaternary structures in chaperone factories
How cellular “assembly lines” keep big machines in shape
Inside each of our cells, thousands of protein parts must snap together into complex molecular machines, from the engines that copy DNA to the factories that build other RNAs. This study asks a deceptively simple question with big implications: how do cells decide, in real time, which helper proteins will escort each new part as it is being built, and how do they keep this process organized in the chaotic fluid of the cell?
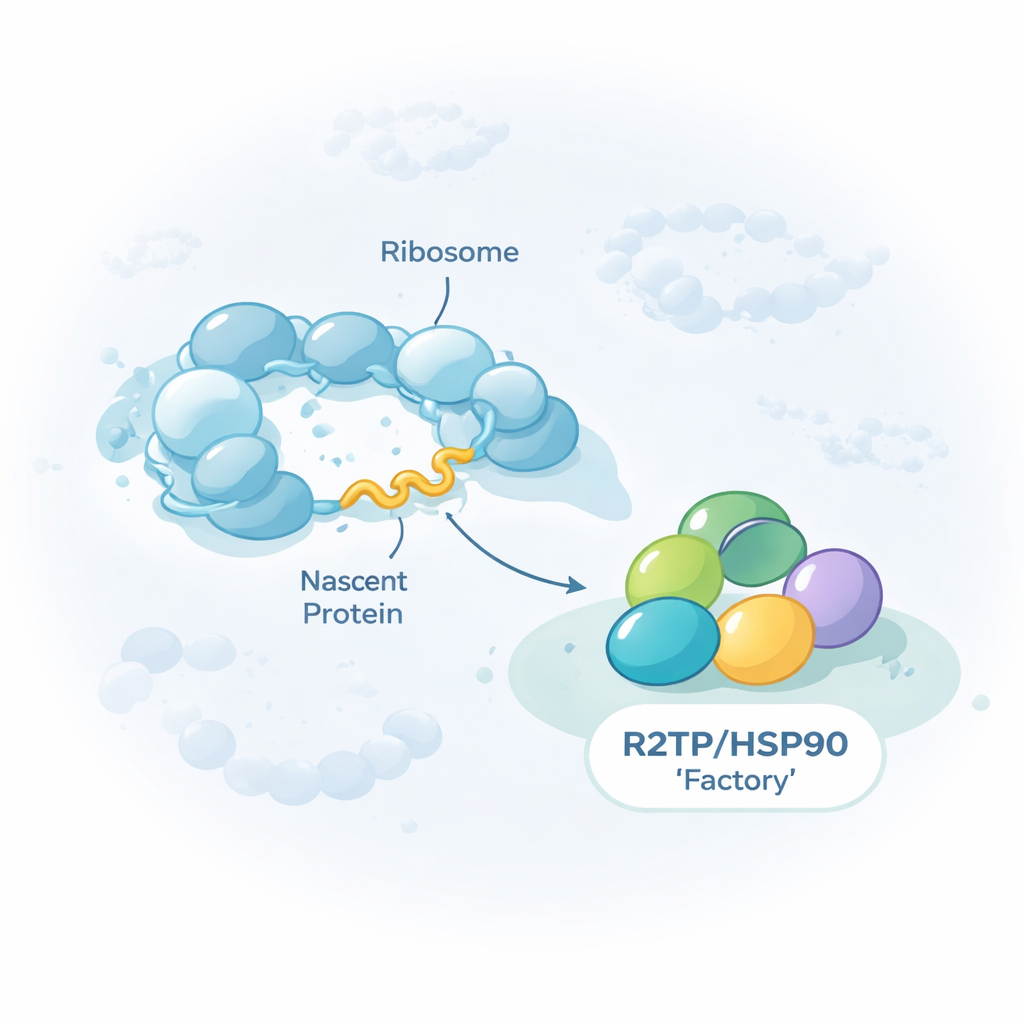
A general-purpose helper for cellular mega-machines
The work focuses on a group of helper proteins, or chaperones, known as HSP90/R2TP. Rather than folding single proteins, this chaperone specializes in assembling giant multi-part complexes, including the three nuclear RNA polymerases that transcribe DNA into RNA, certain RNA–protein particles, and large remodeling machines that reshape DNA packaging. The authors show that R2TP doesn’t just meet these client proteins after they are finished. Instead, it often attaches while each client is still being manufactured on ribosomes, the cell’s protein-making machines. By pulling down R2TP from human cells and sequencing the RNAs that came with it, they found around 150 messenger RNAs whose encoded proteins are contacted by R2TP during their own translation.
Helpers that ride along on growing protein chains
To test whether this binding truly happens during protein synthesis, the team used drugs that freeze or disrupt translation. When they used compounds that strip growing protein chains off ribosomes, most of the messenger RNAs disappeared from R2TP’s grip. But when they used a drug that simply pauses ribosomes and leaves the nascent chains attached, R2TP binding increased. Engineered reporter RNAs confirmed this: if translation was artificially stopped early, the chaperone no longer bound the message. Together, these results point to a clear rule of engagement: R2TP recognizes its clients primarily through the emerging protein chain as it comes out of the ribosome, rather than through the RNA sequence itself.
Chaperone-built “factories” that cluster specific messages
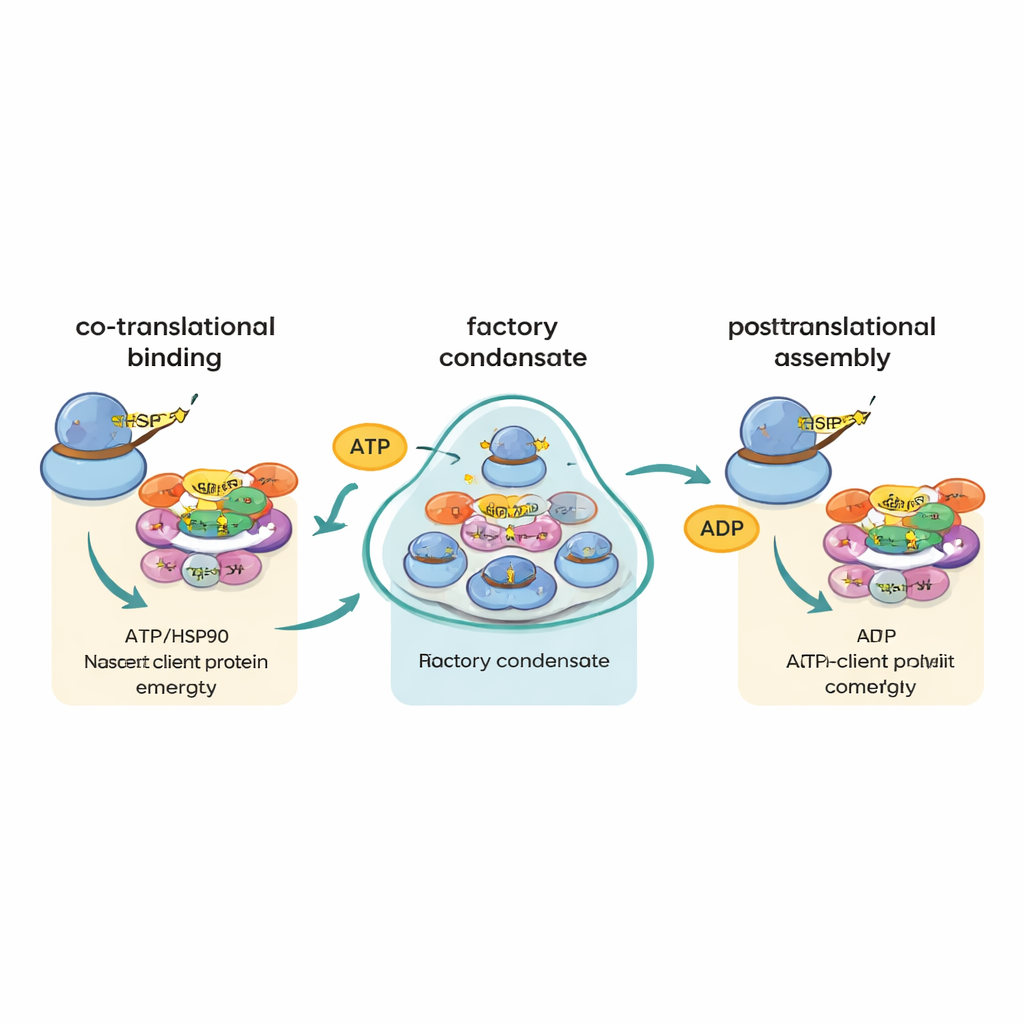
Using advanced single-molecule imaging, the authors then made a surprising discovery. Instead of seeing many different client messages randomly scattered, they observed that some key messages—especially those encoding major subunits of RNA polymerase II and a large splicing factor called PRPF8—often gathered in bright foci in the cytoplasm. These foci were enriched in R2TP and its partner HSP90, as well as in ribosomal components and the very proteins being made, showing that they are active translation sites. When translation or the energy-hungry ATPase activity of R2TP or HSP90 was blocked, these foci dissolved. The authors call these structures “R2TP factories”: specialized condensates where selected messenger RNAs and chaperones are concentrated to favor timely, protective contact between emerging protein chains and their assembly helpers.
Routing parts without matching their blueprints
One earlier idea was that messages encoding different parts of the same complex might find each other and be translated side by side, allowing their protein products to assemble immediately. The new work largely rules out this “co-localized blueprint” model. Even for protein pairs known to join in the same machine, their messenger RNAs rarely appeared together. Instead, the data support a different strategy the authors term “co-translational chaperone channeling,” or “cha-cha.” In this scheme, as each large or assembly-prone protein begins to emerge from the ribosome, R2TP and HSP90 latch on and determine its fate, guiding it through further steps of assembly. The factories themselves do not mainly bring matching messages together; rather, they boost the odds that vulnerable new proteins will promptly meet the right chaperone.
Why this hidden organization matters for health and disease
By revealing that R2TP and HSP90 not only safeguard fragile protein parts but also shape where and how they are translated, this study exposes an unexpected layer of intracellular organization. The cha-cha mechanism helps explain how cells reliably build huge molecular complexes even though the messages for their parts rarely share the same address. It also suggests why defects in chaperones or their energy cycles can destabilize vital machines like RNA polymerase and splicing factors, with consequences ranging from developmental disorders to cancer. In essence, the work shows that cells rely on dynamic chaperone “factories” to route freshly made parts into the right assemblies, keeping their internal machinery both efficient and orderly.
Citation: Philippe, M., Salloum, S., Slimani, F. et al. Co-translational determination of quaternary structures in chaperone factories. Nat Commun 17, 1978 (2026). https://doi.org/10.1038/s41467-026-68687-8
Keywords: protein chaperones, co-translational assembly, molecular machines, RNA polymerase, cellular quality control