Clear Sky Science · en
Structural basis for the dynamic conformations of AP-4 and its association with ARF1
How cells sort and ship their cargo
Every cell is a bustling warehouse, constantly sorting and shipping thousands of protein “packages” to the right destinations. When this delivery system goes wrong in nerve cells, the result can be serious brain and movement disorders. This study uncovers how one key shipping machine, called AP-4, changes shape and teams up with a regulator protein, ARF1, to load cargo into tiny transport bubbles and why its flexibility is so important for healthy neurons.
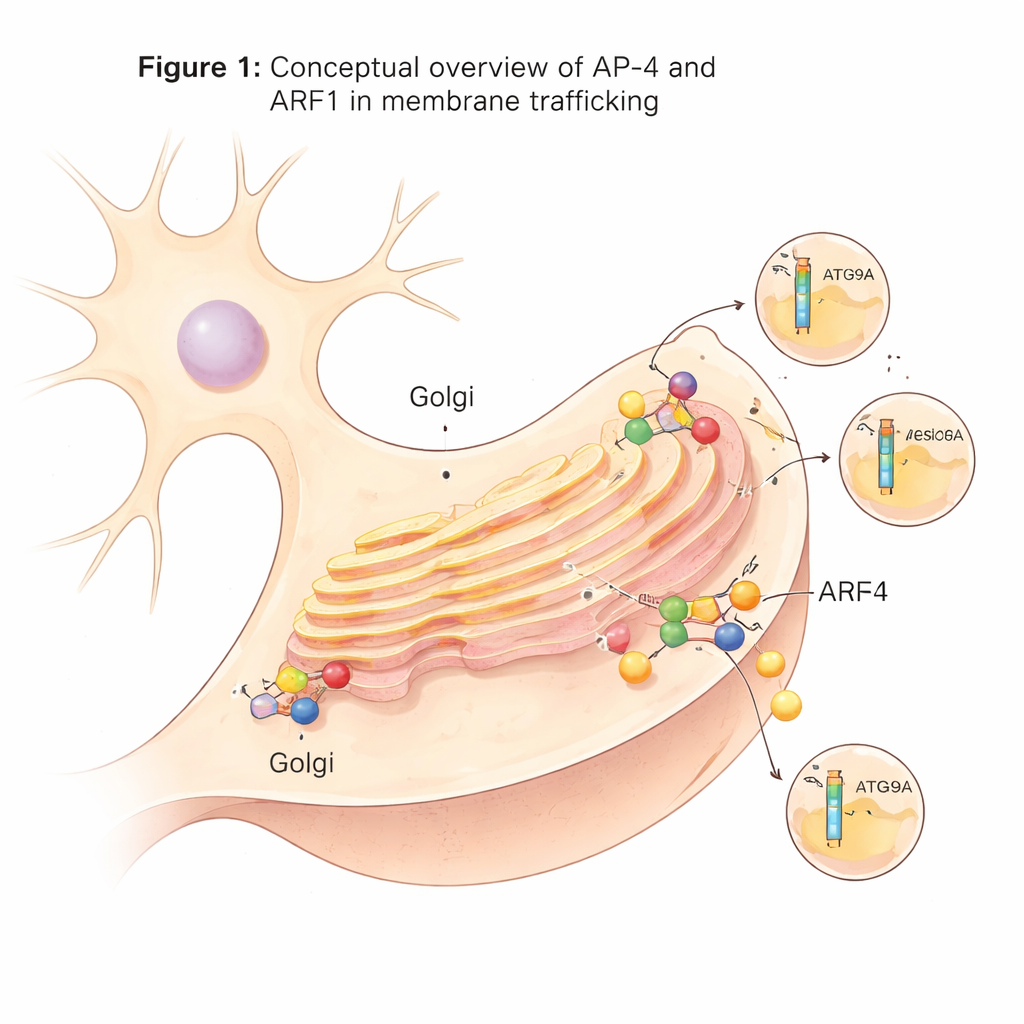
A specialized shipping machine in nerve cells
Inside cells, many transport vesicles bud from a central sorting station known as the trans-Golgi network. These vesicles rely on adaptor protein complexes, or AP complexes, which act like loading docks: they recognize cargo proteins and help form coated vesicles around them. AP-4 is one of these adaptors and is unusual because, unlike many others, it does not use the common scaffold protein clathrin. Even though AP-4 is made in many tissues, it plays an especially important role in neurons. When AP-4 is missing or defective in humans, crucial cargoes such as the autophagy-related protein ATG9A and certain glutamate receptors fail to move properly, leading to a severe neurodevelopmental condition known as AP-4-deficiency syndrome.
A shape-shifting complex at rest
The authors used cryo–electron microscopy, a powerful imaging method that can visualize large protein assemblies, to determine the three-dimensional structure of the AP-4 “core” complex in solution. AP-4 is built from four subunits that together form a bowl-like structure. They discovered that AP-4 does not sit in a single rigid shape. Instead, it naturally toggles between a “closed” form, in which one subunit (the medium subunit’s C-terminal domain) is tucked into the center of the bowl, and an “open” form, in which this piece swings out and becomes highly flexible. Additional single-molecule fluorescence experiments showed that this subunit actually samples at least three positions—closed, partially open, and fully open—revealing that AP-4 is intrinsically dynamic even before it binds to membranes.
ARF1’s role: recruiter, not master switch
Many related adaptor complexes are switched from an inactive to an active shape by binding to small regulatory proteins called ARFs. To see how this works for AP-4, the researchers solved structures of AP-4 bound to ARF1, a small GTP-binding protein that anchors to membranes. They found that ARF1 attaches mainly to one of AP-4’s large subunits near its N-terminus, in a way similar to other AP complexes. Surprisingly, ARF1 binding does not trigger a dramatic structural rearrangement in AP-4. The closed and open shapes still coexist, and the overall architecture of the complex remains largely unchanged. Biochemical tests confirmed that specific contact points between ARF1 and AP-4 are essential for membrane recruitment, but ARF1 alone does not lock AP-4 into a single active state.
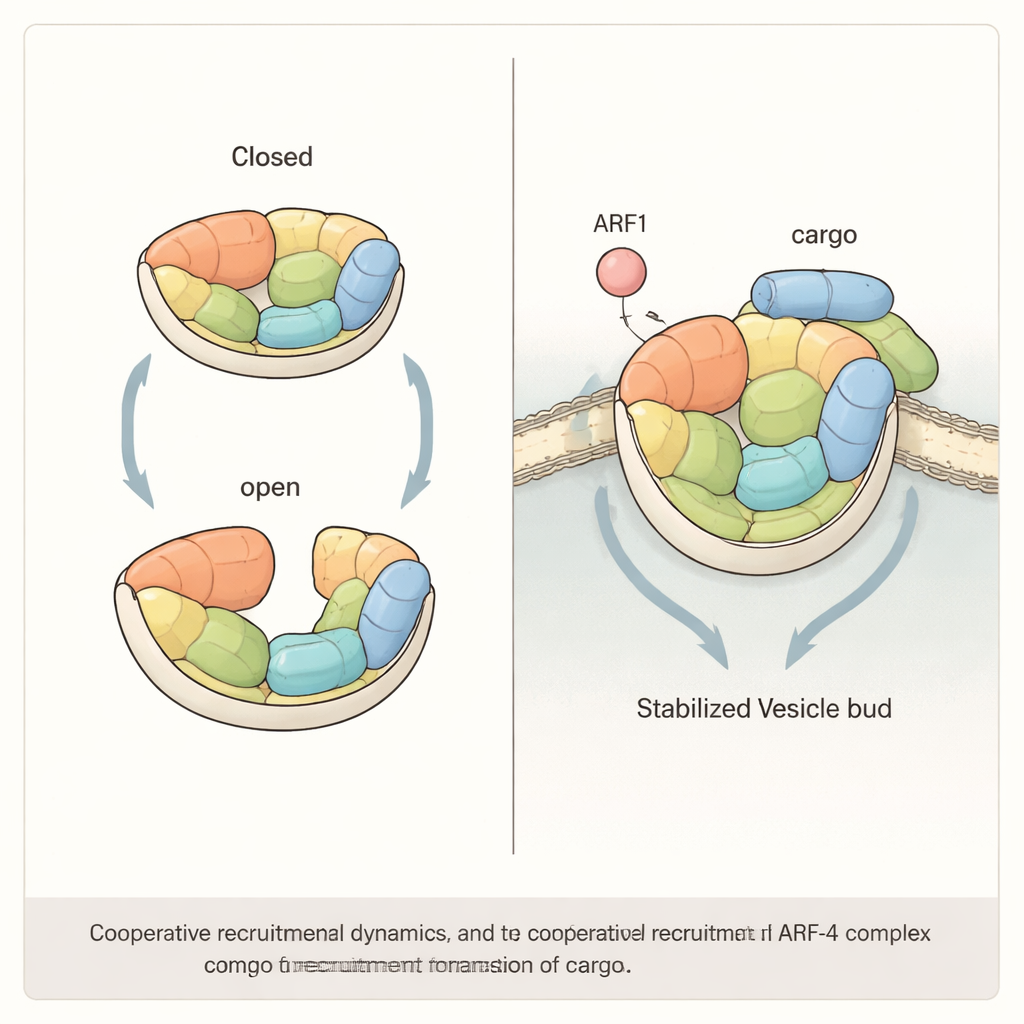
Teamwork between regulator and cargo
The study goes on to show that efficient recruitment of AP-4 to membranes likely requires cooperation between ARF1 and the cargo proteins themselves. By reconstituting the system on artificial beads and liposomes, the authors found that either ARF1 or a short tail from the cargo ATG9A can attract AP-4, but when both are present together they pull in significantly more AP-4 than expected from simply adding their individual effects. When the researchers engineered mutations that stiffened AP-4’s structure—reducing its ability to move between open and closed states—this synergistic effect was lost. In human cells lacking a key AP-4 subunit, restoring only such “rigid” mutant versions of the complex failed to rescue normal trafficking of ATG9A from the Golgi, underscoring that AP-4’s flexibility is not just a curiosity but a functional requirement.
Why flexibility matters for brain health
Overall, the work paints AP-4 as a shape-shifting adaptor that samples multiple conformations in solution and on membranes. Rather than acting as an on–off switch, ARF1 serves as a membrane-based recruiter that works together with cargo proteins to stabilize AP-4 in productive configurations for vesicle formation. Disrupting this balance—either by weakening ARF1 or cargo binding or by freezing AP-4 into one shape—impairs cargo export from the Golgi and can contribute to neurodevelopmental disease. By revealing the structural basis of AP-4’s dynamic behavior, this study offers a framework for understanding AP-4–related disorders and hints at future strategies to restore proper trafficking in affected neurons.
Citation: Wang, Y., Li, W., Qiu, Y. et al. Structural basis for the dynamic conformations of AP-4 and its association with ARF1. Nat Commun 17, 1897 (2026). https://doi.org/10.1038/s41467-026-68679-8
Keywords: membrane trafficking, AP-4 adaptor complex, ARF1, vesicle formation, neurodevelopmental disorder