Clear Sky Science · en
ZNF683+ NK cells govern chemotherapy sensitivity in advanced HPSCC via reshaping immune microenvironment
Why some chemotherapy works better for certain throat cancers
People with advanced hypopharyngeal cancer—a tumor hidden deep in the throat—often face harsh chemotherapy with uncertain results. Some tumors melt away, while others barely budge. This study asks a simple but crucial question: what is different inside the tumors that respond well? By looking at individual immune cells one by one, the researchers uncover a particular type of “guardian” cell that can tip the balance between success and failure of standard chemotherapy.
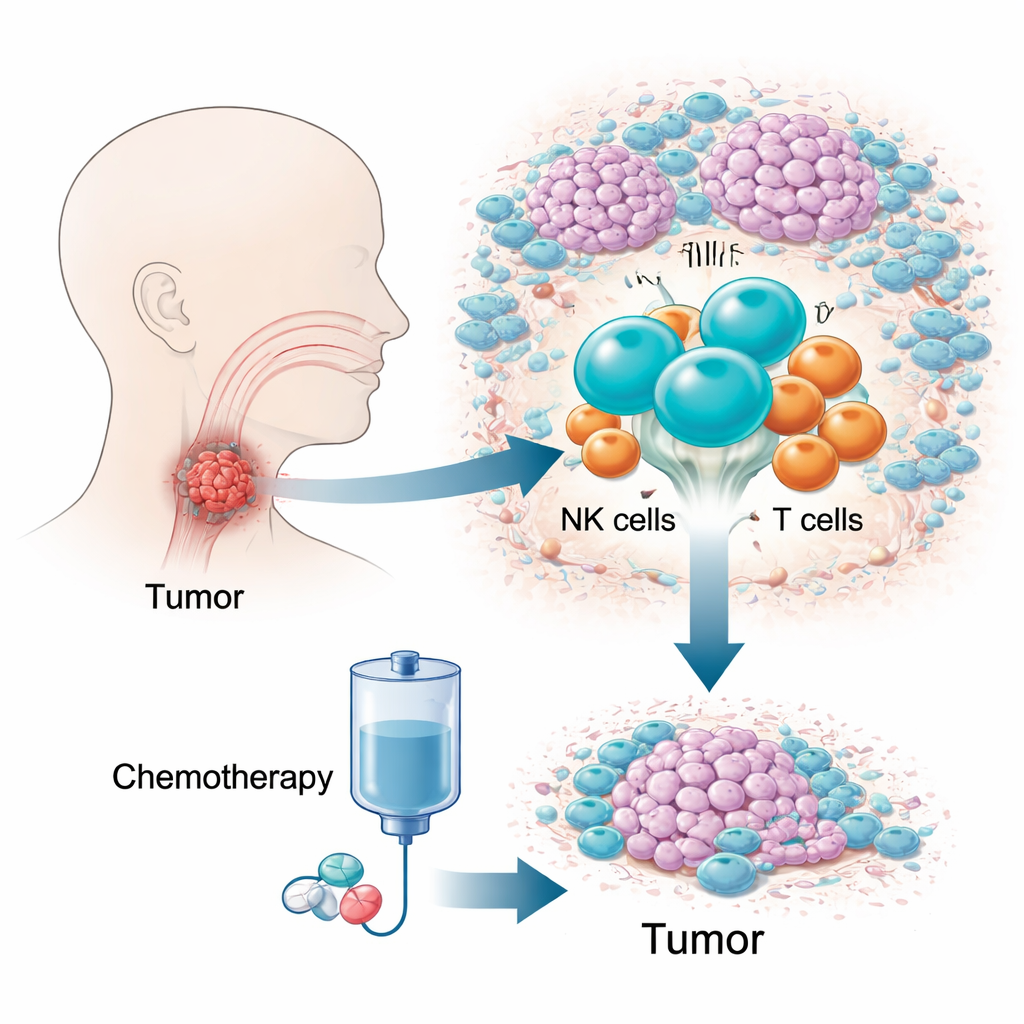
The challenge of a hidden, hard-to-treat cancer
Hypopharyngeal squamous cell carcinoma is one of the deadliest head and neck cancers, in part because it grows in a cramped, hard-to-examine area and causes few symptoms early on. Many patients are diagnosed late, when options are limited and five-year survival is low. A drug combination known as TPF chemotherapy is widely used because it can control disease while preserving the voice box. Yet 10–20 percent of patients show little or no response, losing precious time. Traditional genetic tests, which average signals across millions of cells, have struggled to explain this difference, prompting the authors to focus instead on the tumor’s living ecosystem of immune cells.
Reading the tumor’s immune landscape cell by cell
The researchers followed 12 patients over time, collecting tumor samples before and after two cycles of TPF chemotherapy and profiling more than 150,000 immune cells at single-cell resolution. They also examined an additional 41 patients to confirm key findings using imaging and flow cytometry. By mapping each cell’s gene activity and type, they built a detailed atlas of the tumor microenvironment: T cells, B cells, macrophages, dendritic cells, mast cells, neutrophils, and natural killer (NK) cells. Among all these players, one pattern stood out. Before treatment, tumors that later shrank had many more NK cells than those that resisted therapy, and the higher the NK cell fraction, the greater the eventual tumor shrinkage.
A special NK cell subset sets up T cells for success
Diving deeper, the team split NK cells into three groups based on their molecular features. Only one of these, marked by the transcription factor ZNF683 and largely sitting in the tissue surrounding tumor nests, strongly tracked with good chemotherapy response. Patients whose tumors harbored more of these ZNF683+ NK cells at baseline were far more likely to benefit from TPF. In mouse experiments, removing NK cells almost completely erased the benefit of chemotherapy, confirming that NK activity is not just a marker but a requirement for effective treatment. Intriguingly, these NK cells were not simply acting as direct cancer killers; instead, their gene programs were enriched for guiding and activating T cells.
How NK cells license potent tumor-killing T cells
To understand which T cells mattered most, the scientists followed individual T cell receptor clones before and after treatment. They found that in responders, a specific group of CD8 “effector memory” T cells, enriched for a molecule called GZMK, expanded dramatically after chemotherapy. These T cells produced high levels of the toxic cytokines TNF and interferon-gamma without showing deep exhaustion, making them powerful, durable killers. Spatial imaging revealed that, after treatment, these GZMK+ CD8 T cells clustered strongly in tumors from responders but not from non-responders. In lab co-culture tests using human cells, direct contact with ZNF683+ NK cells drove the growth of this GZMK+ CD8 T cell subset, an effect that vanished when the cells were separated by a barrier.
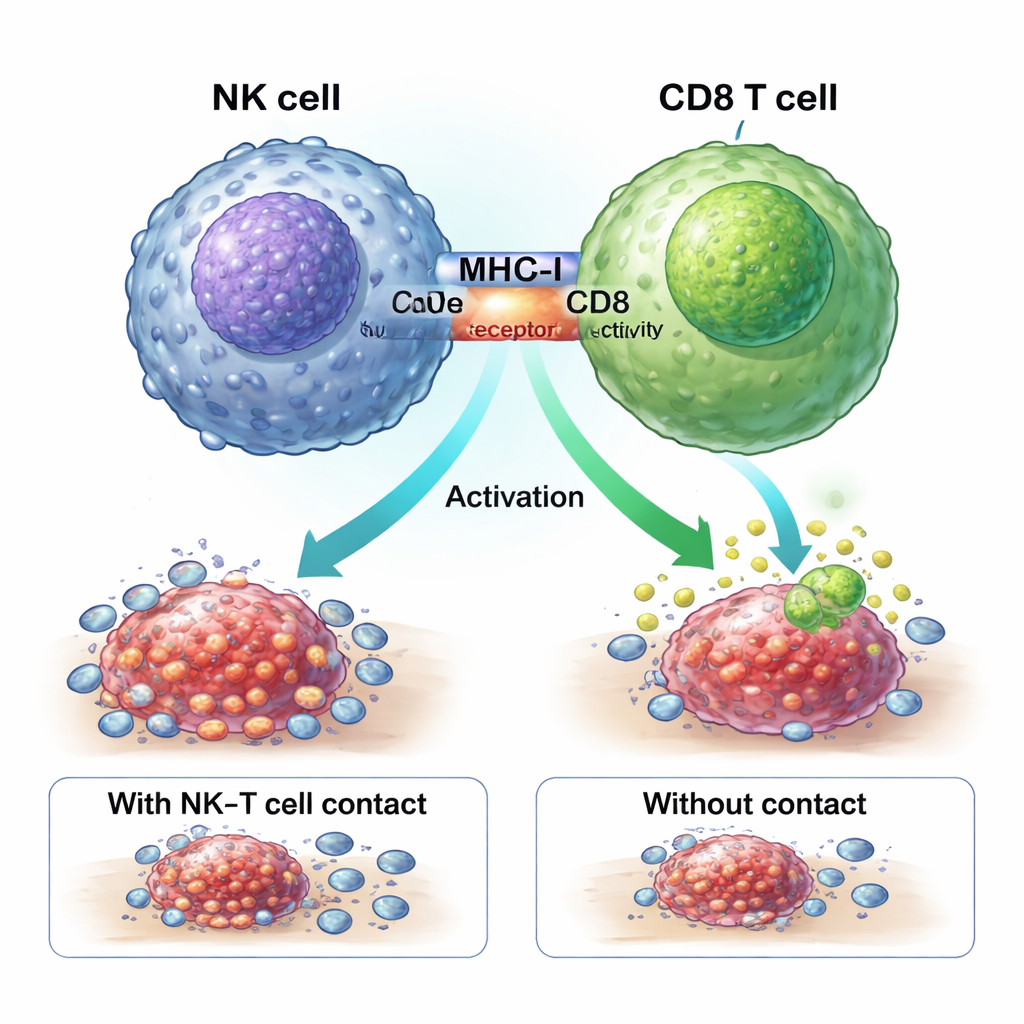
A physical handshake that turns on tumor fighters
The study shows that ZNF683+ NK cells boost chemotherapy response through a physical “handshake” with CD8 T cells. Molecules called MHC-I on the NK cell surface engage CD8 receptors on T cells at a close-contact zone, or immune synapse. Blocking MHC-I stopped NK cells from generating the potent GZMK+ CD8 T cells. In specially engineered mice lacking ZNF683 only in NK cells, these NK cells expressed less MHC-I and were much poorer at activating CD8 T cells, reinforcing the idea that ZNF683 programs NK cells to become effective coaches for T-cell fighters rather than simple executioners.
What this means for patients
For lay readers, the key message is that not all immune cells in a tumor are equal. This work identifies a particular NK cell subset as an advance scout that prepares the battlefield before chemotherapy begins. Tumors rich in these cells are primed to unleash waves of strong CD8 T cells once drugs hit, leading to much better shrinkage. Measuring the abundance of this NK–T cell axis before treatment could help doctors predict who will benefit from TPF chemotherapy and who might need alternative or additional therapies. In the future, treatments that boost or mimic these ZNF683+ NK cells could turn resistant hypopharyngeal cancers into ones that are far more sensitive to standard chemotherapy.
Citation: Li, G., Xiao, W., Wu, H. et al. ZNF683+ NK cells govern chemotherapy sensitivity in advanced HPSCC via reshaping immune microenvironment. Nat Commun 17, 2069 (2026). https://doi.org/10.1038/s41467-026-68676-x
Keywords: hypopharyngeal cancer, chemotherapy resistance, natural killer cells, tumor immune microenvironment, CD8 T cells