Clear Sky Science · en
Photo-induced selective N-N bond construction via harnessing nitrene release and transfer
Lighting Up a New Way to Join Nitrogen Atoms
Nitrogen atoms sit at the heart of many medicines, crop-protection agents, and advanced materials. Chemists are very good at attaching nitrogen to carbon, but directly connecting two nitrogen atoms is much harder. This study introduces a light-powered, metal-free method to snap nitrogen atoms together in a controlled way, opening a simpler route to a wide range of useful molecules.
Why Connecting Nitrogen to Nitrogen Is Hard
Many natural products and drugs contain pairs of nitrogen atoms bonded to each other, a pattern that can tune how a molecule behaves in the body or in materials. Existing ways to build these N–N units usually start from pre-made nitrogen–nitrogen fragments, such as hydrazines or diazo compounds, and then modify them step by step. Directly forging the N–N bond from simple amines is appealing but difficult: nitrogen is highly electronegative, so two nitrogen atoms do not naturally form a stable, nonpolar bond. Past successes have relied heavily on transition-metal catalysts and often work only for narrow families of molecules, limiting their usefulness for discovery chemistry.
Using Light to Tame a Reactive Middleman
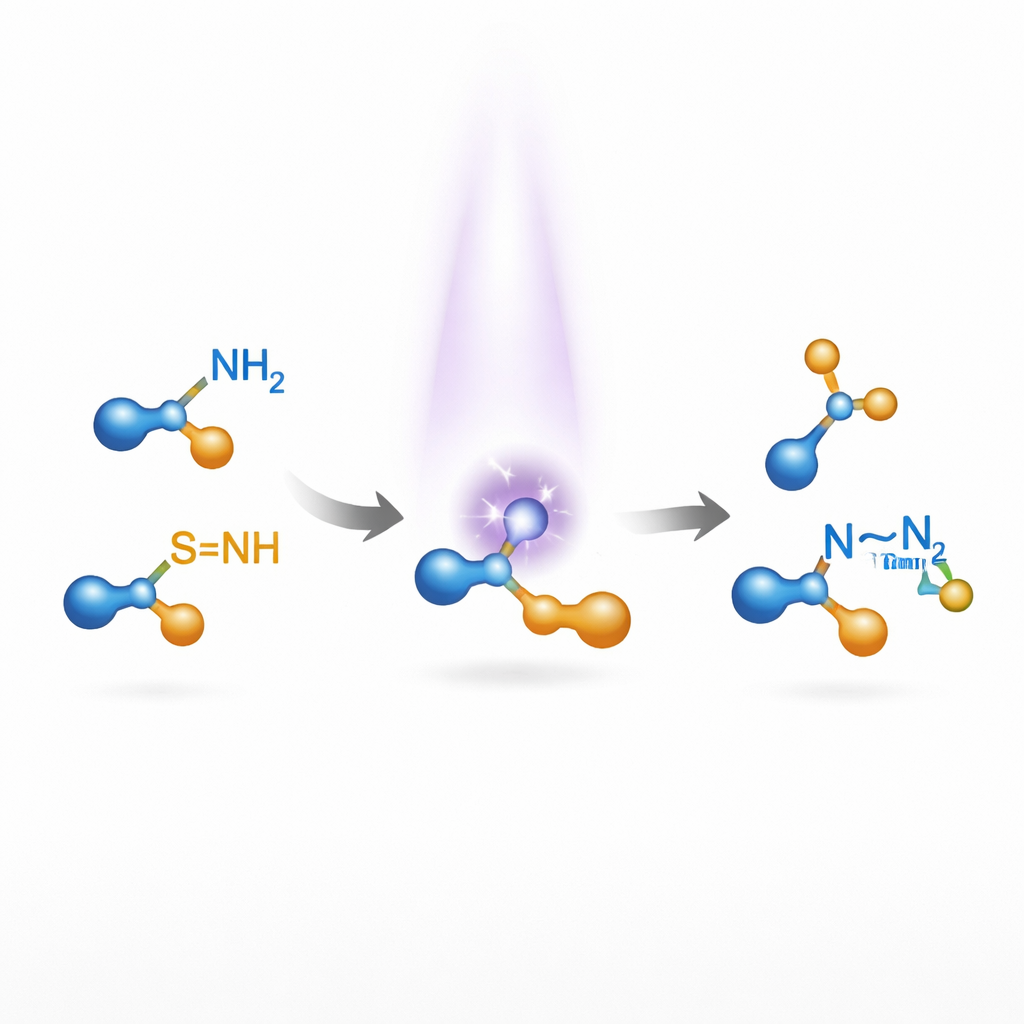
The authors turned to nitrenes—highly reactive, short-lived nitrogen species that can, in principle, insert into other bonds. Traditional nitrene chemistry typically needs metal catalysts and can suffer from side reactions because nitrenes are so energetic. The team asked whether they could use light to generate nitrenes in a gentler, more controlled way, without any metals, and then use them to link simple amines into N–N–containing products. Their key insight was to use sulfilimines, a family of molecules that are easy to tailor and that absorb ultraviolet and near-visible light. When illuminated, these sulfilimines can split in a way that releases a nitrene fragment while recovering a benign sulfur-containing by-product.
A Broad, Metal-Free Recipe for Nitrogen Pairs
After systematic testing, the researchers identified a particular sulfilimine that, under 365-nanometer light in chloroform, reacts efficiently with many amines to give hydrazide products—molecules featuring a freshly made N–N bond next to a carbonyl group. They optimized factors such as the sulfilimine structure, solvent, and light intensity to favor the desired product over common by-products like ureas. Under these mild conditions, a wide variety of amines, including aromatic and aliphatic types and cyclic variants, successfully coupled. They also showed that many different sulfilimines, bearing both aromatic and aliphatic acyl groups and sulfonyl groups, participate well, yielding dozens of distinct N–N–containing products. Importantly, the method can be applied late in a synthesis to modify complex, biologically active molecules such as common anti-inflammatory drugs and chiral building blocks, demonstrating its practicality for medicinal and materials chemistry.
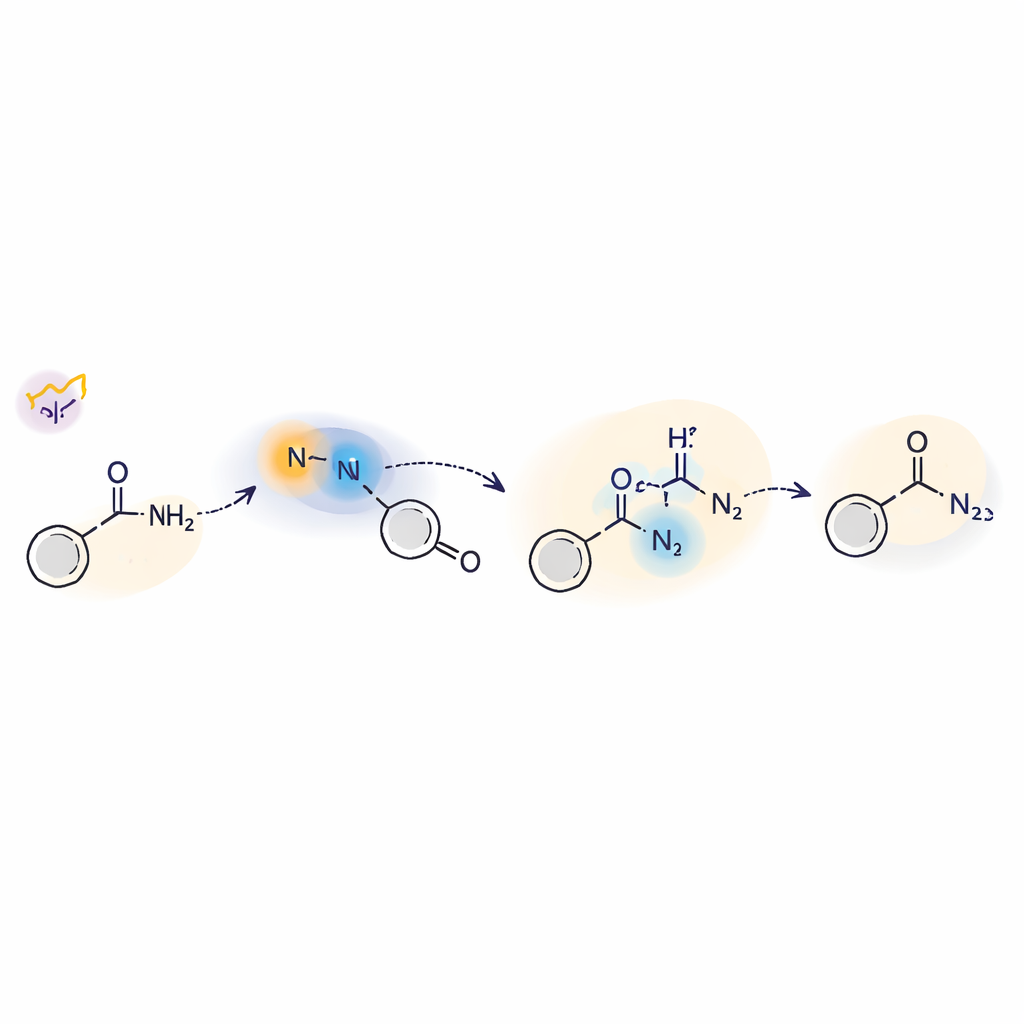
Peering Under the Hood of the Light-Driven Step
To understand how this light-triggered chemistry works, the team combined experiments and theory. They used radical traps, isotope-labeling, and electron paramagnetic resonance to show that free nitrene intermediates and nitrogen-centered radicals appear during the reaction. Time-resolved laser spectroscopy revealed two key forms of the nitrene: a short-lived triplet state and a longer-lived singlet state. The singlet form reacts directly with amines by a kind of nucleophilic attack, while the triplet can participate in hydrogen-atom transfer steps. Computer calculations supported a mechanism in which light excites the sulfilimine, its sulfur–nitrogen bond cleaves, and the resulting nitrene then engages the amine through both singlet and triplet pathways before settling into the final hydrazide structure.
What This Means for Future Molecules
This work shows that carefully designed sulfilimines can act as “nitrene reservoirs” that release reactive nitrogen units on demand under light, without the help of metals. By harnessing both the singlet and triplet personalities of nitrenes while keeping their concentration low, the method achieves selective formation of N–N bonds across a broad range of building blocks. For non-specialists, the takeaway is that chemists now have a simpler, cleaner, and more flexible way to stitch nitrogen atoms together, which could speed up the creation of new medicines, agrochemicals, and nitrogen-rich materials.
Citation: Yu, M., Feng, J., Wang, X. et al. Photo-induced selective N-N bond construction via harnessing nitrene release and transfer. Nat Commun 17, 2084 (2026). https://doi.org/10.1038/s41467-026-68674-z
Keywords: nitrene chemistry, photo-induced reactions, N–N bond formation, sulfilimines, hydrazides