Clear Sky Science · en
Harnessing PV-oxirene for the modular synthesis of α-Oxy carbonyls
Why tiny chemical linkers matter
Many modern medicines, crop protectants, and advanced materials rely on small molecular “connectors” that hold larger pieces together and subtly tune how they behave. One such connector family, called α‑oxy carbonyls, can change a drug’s stability, a peptide’s shape, or a plastic’s properties. Yet these promising linkers have been surprisingly hard to make in a flexible, mix‑and‑match way. This article describes a new, mild method that lets chemists quickly build many different α‑oxy carbonyls from simple starting materials, opening fresh possibilities in drug design, chemical biology, and materials science.
Building a better chemical toolbox
α‑Oxy carbonyls appear in nature and technology alike—from anti‑cancer agents and antibiotics to biodegradable polymers and herbicides. A closely related group, α‑hydroxy acids, can even be slotted into protein‑like chains to fine‑tune how these chains fold and interact, which in turn affects how they recognize biological targets. Existing routes to these linkers rely on multi‑step sequences, harsh reagents, or narrow sets of building blocks. These limits make it difficult for chemists to quickly explore new structures or to modify complex drug molecules at a late stage of development.
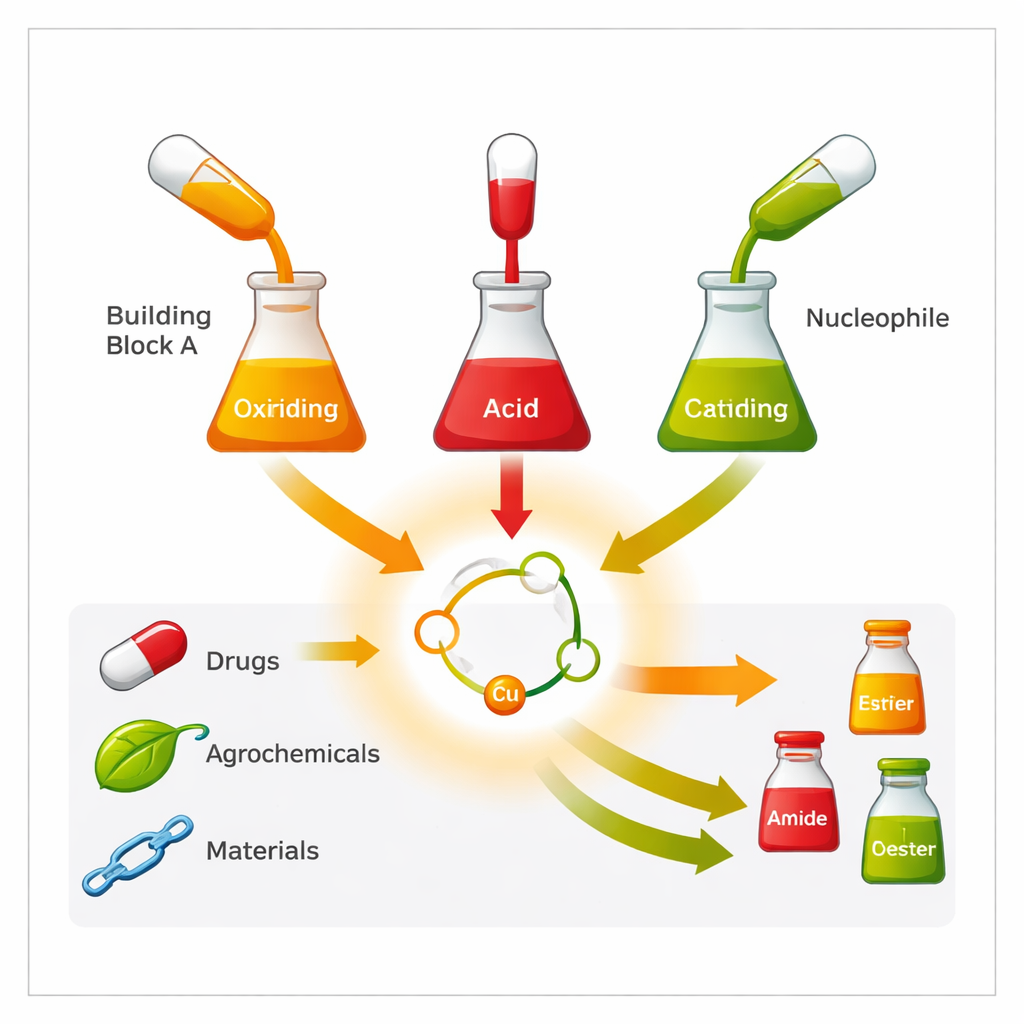
A new three‑piece assembly line
The researchers introduce a copper‑based reaction that stitches together three simple ingredients in one pot: a specially designed “ylide” reagent, an ordinary carboxylic acid (a group found in countless natural products and drugs), and a third partner that brings in nitrogen, oxygen, sulfur, selenium, or water. Under mild conditions and without precious metals, these three pieces are combined into α‑oxy carbonyl products such as esters, amides, thioesters, and related structures. The reaction tolerates many other sensitive features—double bonds, nitriles, sulfides, and complex ring systems—showing that it can be used directly on intricate, drug‑like molecules.
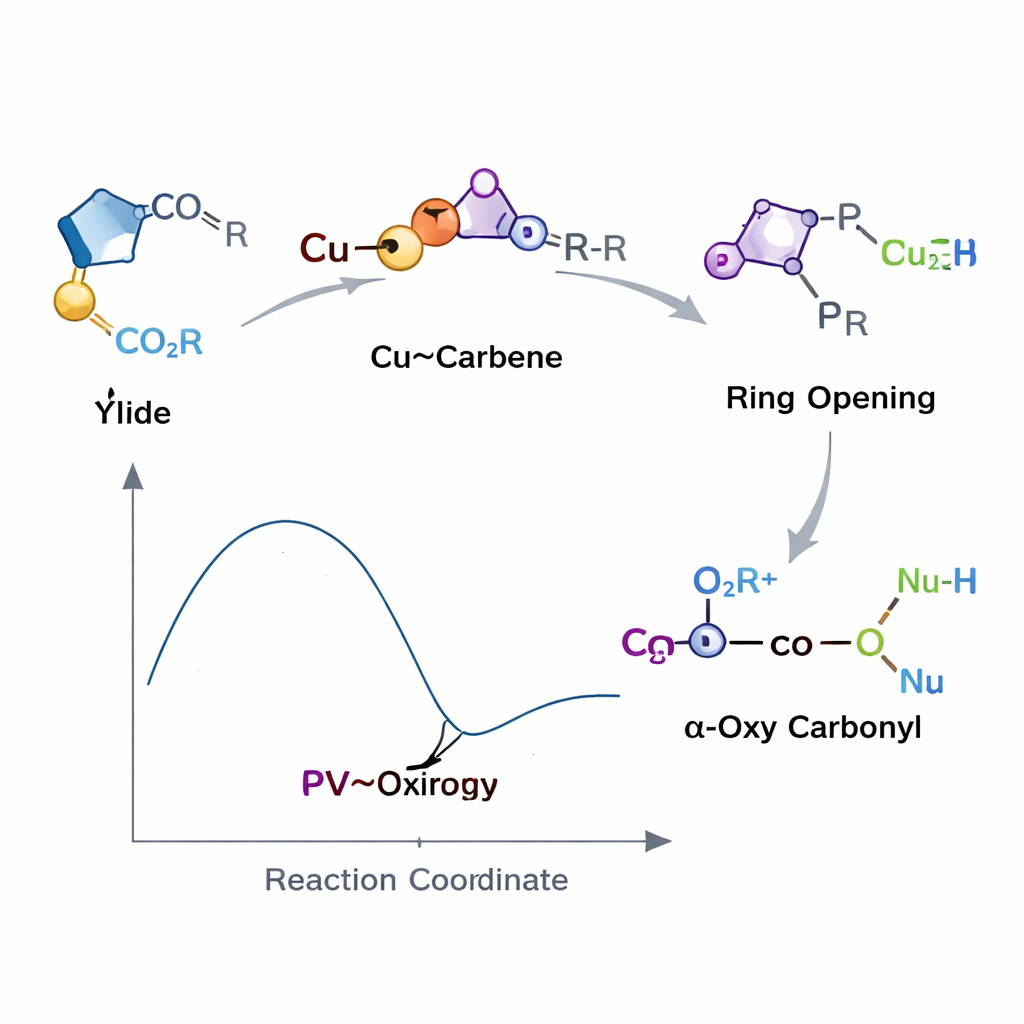
Harnessing a fleeting ring for control
Behind this new method lies an unusual, highly strained ring called an oxirene, long considered a short‑lived curiosity rather than a practical tool. By embedding a phosphorus‑containing group into the ring and generating it briefly with the help of copper, the team turns this reactive structure into a controllable waystation. The carboxylic acid first becomes a charged partner that attacks the oxirene at a preferred position, steering the reaction so that only one main product forms instead of a messy mixture. The resulting intermediate then passes its acyl fragment to the third nucleophile, delivering the final α‑oxy carbonyl in a step that resembles a carefully choreographed relay race.
From peptides to herbicides and beyond
To showcase what this chemistry can do, the authors build short peptide‑like chains in which normal links are replaced by ester links derived from α‑hydroxy acids. Such “depsipeptides” are valuable probes for understanding how small changes in backbone structure affect biological function, but they are typically difficult to access. The same strategy also produces a commercial herbicide, Lactofen, in a single step from a known precursor, and quickly generates new analogues that would be cumbersome to make by older routes. In addition, by combining the new reaction with a few straightforward steps, the team demonstrates a way to lengthen carboxylic acids by one carbon while introducing an α‑hydroxy group, a transformation prized in medicinal chemistry.
What this means going forward
In practical terms, this work offers chemists a versatile, user‑friendly tool for installing α‑oxy carbonyl linkers almost anywhere a carboxylic acid and a nucleophile are present, even in complex, bioactive molecules. On a deeper level, it shows that a once‑esoteric intermediate, the oxirene, can be tamed and put to work in everyday synthesis. By turning a fleeting ring into a controllable step in an assembly line, the method broadens the menu of molecular connectors available for designing drugs, probing biological systems, and engineering new materials.
Citation: Huang, S., Duan, D., Luo, J. et al. Harnessing PV-oxirene for the modular synthesis of α-Oxy carbonyls. Nat Commun 17, 1918 (2026). https://doi.org/10.1038/s41467-026-68671-2
Keywords: alpha-oxy carbonyls, modular synthesis, copper catalysis, depsipeptides, carboxylic acid functionalization