Clear Sky Science · en
Glycine photosynthesis via C−N coupling of waste plastic and nitrate over diatomic Pd−B catalyst
Turning Trash into Building Blocks of Life
Plastic bottles, food containers and polyester clothes are everywhere—and so is the waste they leave behind. At the same time, many factories release nitrate-laden wastewater that can pollute rivers and seas. This study shows how both problems can be addressed at once: by using sunlight and a specially designed catalyst to turn discarded plastic and nitrate into glycine, a simple amino acid widely used in food, medicine and agriculture.
Why Glycine and Waste Matter
Glycine is one of the basic building blocks of proteins and is produced on the scale of hundreds of thousands of tons per year. Conventional industrial methods, especially the classic Strecker process, rely on toxic cyanide compounds and harsh reaction conditions, raising safety and environmental concerns. Meanwhile, global production of the plastic PET—used in bottles, textiles and packaging—exceeds 100 million tons annually, and more than 80% of it ends up in landfills or the environment. When PET is chemically broken down, one of the products is ethylene glycol, a low-value liquid that is costly to purify. The authors ask a simple question: instead of treating ethylene glycol and nitrate as waste, could they become feedstocks for making valuable glycine using only sunlight?
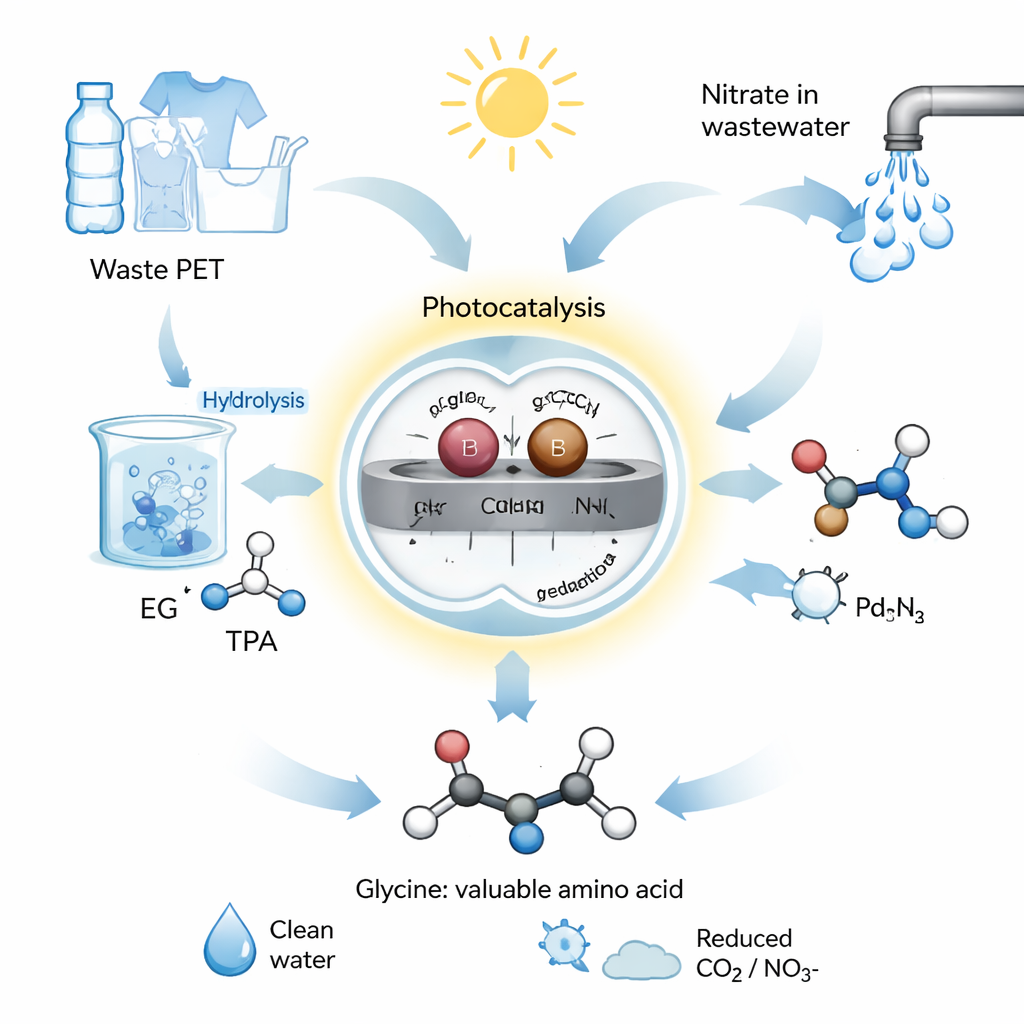
A Sun-Driven Chemical Shortcut
The team designed a photocatalytic system, meaning it uses light to drive chemical reactions. They built a catalyst from graphitic carbon nitride, a semiconductor, and anchored pairs of individual palladium (Pd) and boron (B) atoms on its surface. These paired atoms act like a microscopic two-person team, each handling a different half of the chemistry. In their process, PET waste is first hydrolyzed—split apart in alkaline water—to release ethylene glycol. This ethylene glycol and nitrate from wastewater are then placed in water with the Pd–B catalyst and exposed to simulated or natural sunlight. Under these mild conditions, the system converts the mixture into glycine with a high yield and a selectivity of over 92%, meaning very little unwanted by‑product is formed.
How the Atomic Duo Works
The catalyst’s success hinges on how it manages short‑lived reaction intermediates. Boron sites on the surface are “hole‑rich,” meaning they readily accept positive charge when the material absorbs light. At these B sites, ethylene glycol is gently oxidized—stripped of hydrogen—to form glycolaldehyde, an unstable molecule that normally tends to over‑oxidize into acids or even carbon dioxide. Palladium sites, in contrast, are “electron‑rich.” They use the light‑generated electrons to reduce nitrate stepwise into ammonium or ammonia. The key step is a carbon–nitrogen coupling between glycolaldehyde and these nitrogen species to form ethanolamine, which is then further oxidized, again mainly at B sites, to give glycine. By stabilizing glycolaldehyde just long enough to react, and by keeping electrons and holes spatially separated, the Pd–B pair steers the chemistry away from wasteful side reactions and toward the desired amino acid.
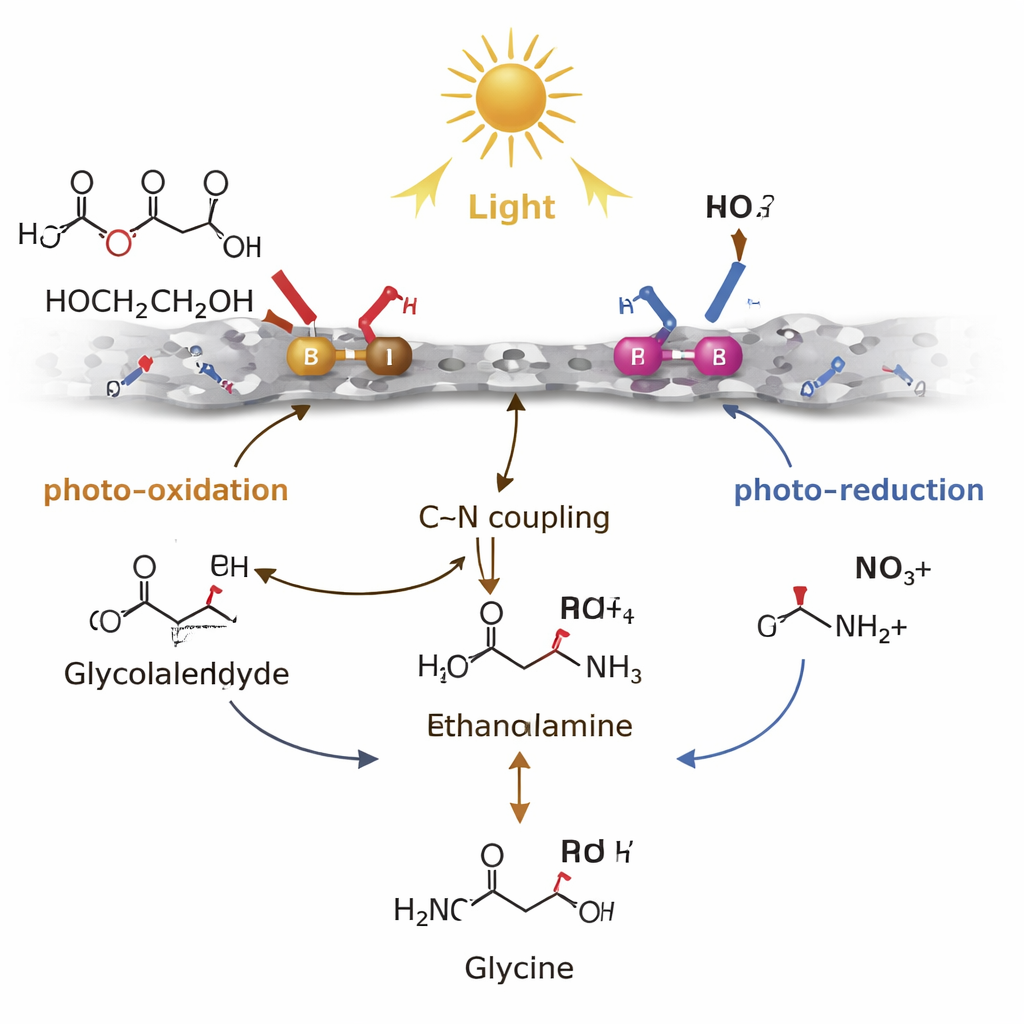
From Lab Mechanism to Real-World Waste
The researchers confirmed each part of this pathway with a suite of techniques, tracking radicals, intermediates and nitrogen products in real time. They compared different catalyst materials and metals and found that g‑C3N4 supports with isolated Pd–B pairs performed best, achieving glycine production rates of 2.9 millimoles per gram of catalyst per hour. The catalyst stayed active over repeated cycles, and its structure remained stable. Importantly, the team moved beyond pure laboratory chemicals. They demonstrated gram‑scale glycine production starting from real post‑consumer PET—powder, bottles, clothing, bags and more—combined with nitrate solutions and even complex wastewater. They also showed that related alcohols from biomass, such as glycerol, can serve as alternative carbon sources, broadening the approach’s relevance.
A Win–Win Route for Waste and Climate
To bring the concept closer to practice, the authors tested their process under natural sunlight focused by a simple Fresnel lens. The system consistently produced glycine with high selectivity, and modeling suggests that scaling it up could avoid substantial emissions of carbon dioxide and prevent nitrate discharge into the environment. In plain terms, this work outlines a way to convert piles of used plastic and polluted water into a useful amino acid, using only light and a finely tuned catalyst. While engineering challenges remain before industrial deployment, the study highlights how atomic‑level catalyst design can turn two major waste streams into a single, valuable chemical product.
Citation: Ya, Z., Li, M., Fu, D. et al. Glycine photosynthesis via C−N coupling of waste plastic and nitrate over diatomic Pd−B catalyst. Nat Commun 17, 1887 (2026). https://doi.org/10.1038/s41467-026-68666-z
Keywords: glycine, plastic recycling, photocatalysis, nitrate wastewater, single-atom catalysts