Clear Sky Science · en
Multilayer oxide protection layer with multiple tunnelling paths for efficient and durable Si-based photocathode
Turning Polluted Water and Sunlight into Useful Fuel
Excess nitrate pollution in water is a growing problem, but those same nitrate ions can be turned into ammonia, a key ingredient for fertilizers and chemicals. This article describes a new way to build solar-powered electrodes, based on silicon, that can both survive in harsh, alkaline liquids and efficiently convert nitrates into ammonia. The work tackles a long-standing conflict between getting good performance and keeping delicate materials from corroding.
Why Silicon Needs Body Armor
Silicon is the workhorse of modern electronics and an excellent absorber of sunlight, which makes it attractive for solar-driven chemistry. In photoelectrochemical devices, light shines on silicon to create charged particles that drive reactions such as splitting water or converting nitrates to ammonia. The catch is that silicon is chemically fragile in water, especially in strong acids or bases, and will quickly corrode if left bare. Past attempts to protect it have relied on ultra-thin metal or transparent oxide films. Thin films allow charges to pass but fail over time, while thicker ones last longer but block the flow of charges, leaving engineers stuck between efficiency and durability.
A Layered Shield with Many Shortcuts
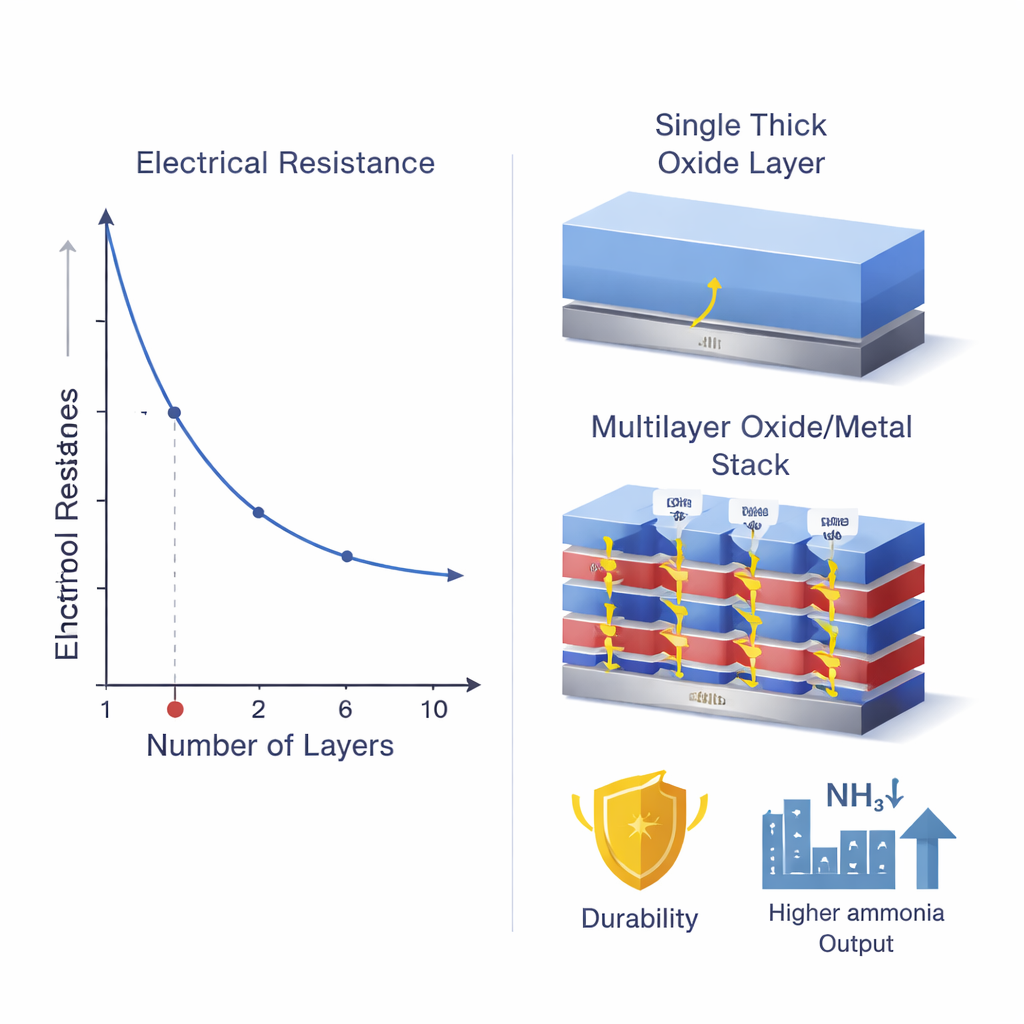
To escape this trade-off, the researchers designed a new protective “body armor” made from many repeating nanoscale layers of an oxide and a metal. Instead of a single thick oxide film, they stack units of titanium dioxide (an oxide) and iron (a metal) into a fixed total thickness of about 36 nanometers. By adjusting how many times this oxide/metal unit is repeated, they can tune both how easily charges move and how well the silicon is shielded from the liquid. Computer simulations and electrical measurements showed that when the stack is divided into six very thin oxide/metal units, electrons can travel along multiple tunneling paths through the layers with surprisingly low resistance. This design keeps the total barrier thick enough to resist corrosion, but perforated with many quantum “shortcuts” for electrons.
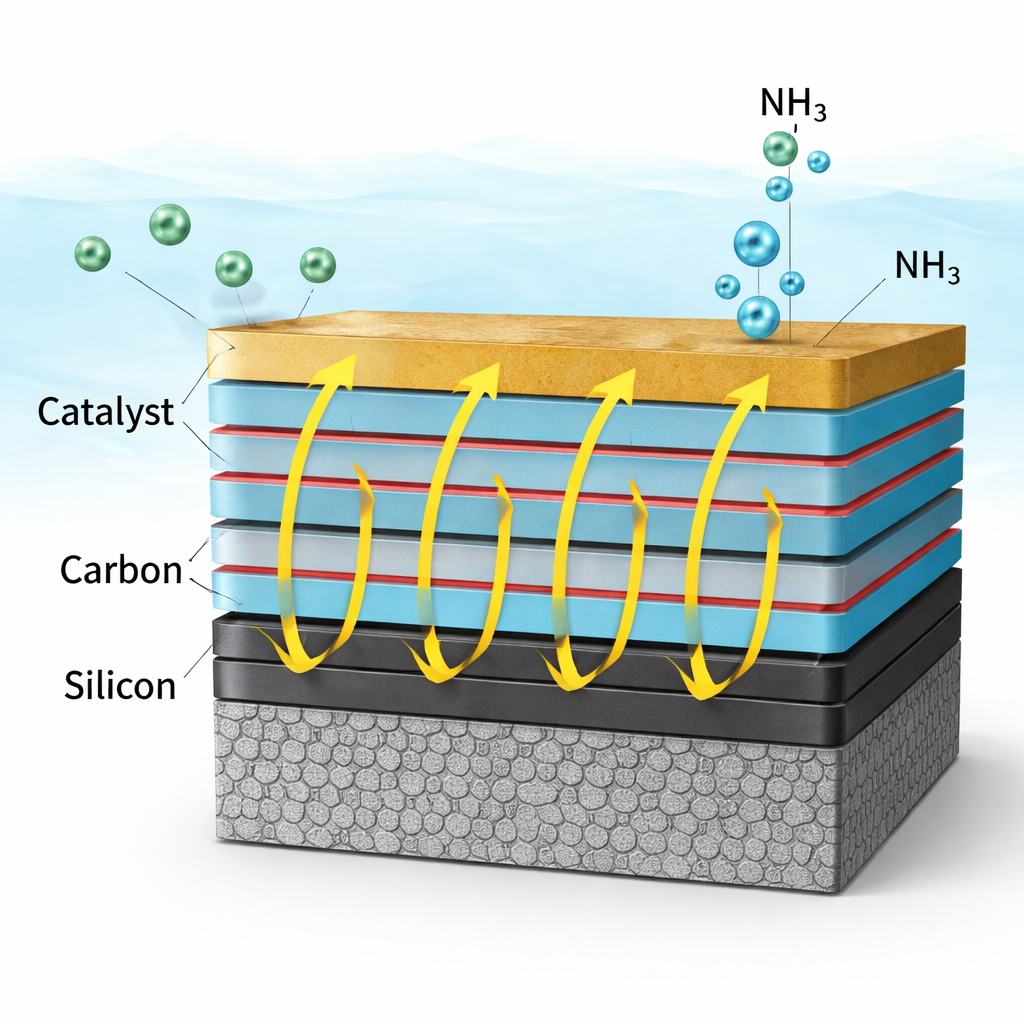
Building and Testing the Solar Nitrate-to-Ammonia Electrode
The team then turned this concept into a working device. They started with a textured silicon wafer that efficiently traps light, added a thin carbon layer to help shuttle electrons, and coated it with the multilayer oxide/metal protection stack. On top, they deposited a thin iron–copper alloy that speeds up the chemical reaction converting nitrate to ammonia. When this photocathode was placed in a strongly alkaline solution containing nitrate and exposed to simulated sunlight, it generated high currents while operating near the thermodynamic limit where hydrogen gas would otherwise form. The best-performing version, with six repeating oxide/metal layers, produced more ammonia at higher efficiency and lower applied voltage than versions with fewer or more layers, confirming the predicted “sweet spot” in resistance.
Balancing Speed, Stability, and Versatility
Beyond raw output, the new protection strategy improved how quickly and cleanly charges moved through the device. Electrical tests under light showed the six-layer structure had the lowest internal resistance and the fastest travel time for photogenerated electrons to reach the catalyst surface, which reduced energy losses from recombination. Impedance measurements and surface potential mapping revealed a stronger built-in electric field at the surface, helping to pull electrons toward the reaction sites. At the same time, the thick but cleverly structured barrier endured more than 100 hours of operation in harsh alkaline conditions, with only slow, measurable loss of material. The concept proved flexible as well: swapping titanium dioxide or iron for other oxides and metals, such as cerium oxide and palladium, still gave strong performance when the stack was tuned to six units.
From Cleaner Water to Better Solar Chemistry
In plain terms, this work shows how to give a sensitive silicon device a tough protective coat that does not slow it down. By slicing a protective oxide film into many ultrathin layers separated by metal, the researchers created multiple quantum pathways for electrons while preserving thickness against corrosion. The result is a silicon-based photocathode that can efficiently turn nitrate pollution into useful ammonia under sunlight, and that lasts long enough to be practically relevant. Because the multilayer approach can be applied to different oxides and metals, it offers a general blueprint for durable, high-performance coatings in a wide range of solar and electrochemical technologies.
Citation: Zhou, Y., Cheng, Z., Lyu, Y. et al. Multilayer oxide protection layer with multiple tunnelling paths for efficient and durable Si-based photocathode. Nat Commun 17, 1871 (2026). https://doi.org/10.1038/s41467-026-68665-0
Keywords: photoelectrochemistry, silicon photocathode, nitrate reduction, multilayer oxide protection, solar ammonia synthesis