Clear Sky Science · en
Low temperature and rapid photothermal oxidation of liquid gallium for circular hydrogen production
A New Way to Make Clean Fuel
Hydrogen is often called the fuel of the future: when burned, it produces water instead of smoke or soot. But most hydrogen today is made from fossil fuels or with expensive, power-hungry machines. This study explores a different path, using a soft, silvery metal called gallium together with ordinary water—and even seawater—to create hydrogen in a reusable, circular process powered largely by sunlight.
Turning Light and Metal into Fuel
The researchers discovered that tiny droplets of liquid gallium can react with water to release hydrogen gas when they are heated by light. Gallium melts just above room temperature, so under mild warming it becomes a flowing liquid. When light shines on the droplets, they absorb the energy and heat up, which speeds up their reaction with water. During this reaction, the gallium is converted into a solid compound called gallium oxyhydroxide while hydrogen bubbles are released. Because gallium is liquid, the solid layer that forms on its surface naturally flakes off instead of sealing it, so fresh metal remains exposed and the reaction can continue rapidly.
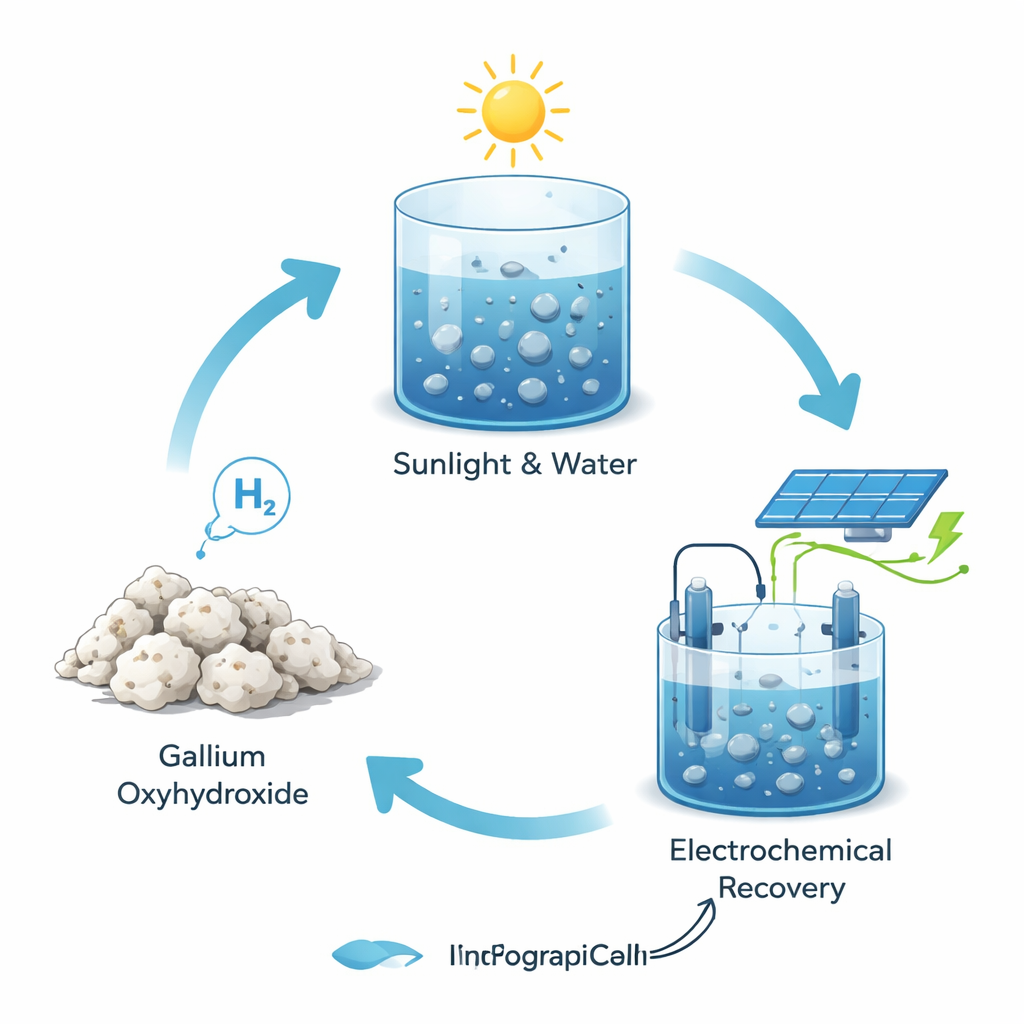
A Circular Loop Instead of a One-Way Use
Most chemical reactions that produce hydrogen consume their metal ingredients for good, leaving behind stubborn waste materials. In contrast, this work shows that the solid gallium oxyhydroxide left after hydrogen production is not a dead end. It can be dissolved in a mild acid and turned back into liquid gallium using standard electrochemical equipment—essentially, a battery-like setup that runs in reverse. Powered by electricity, ideally from renewable sources such as solar or wind, this recovery step regenerates almost all of the original gallium. That means the same batch of metal can be cycled again and again, forming a closed loop for hydrogen production rather than a throwaway process.
Fast, Low-Temperature Hydrogen from Many Waters
In practical tests, the team created gallium droplets by using sound waves to break up melted metal into many small particles. Smaller droplets gave a larger surface area and absorbed light more efficiently, which made hydrogen production both faster and more complete. Under strong but realistic light levels equivalent to concentrated sunlight, 0.2 grams of gallium droplets in water could be fully converted to the solid product in about an hour and a half, producing hydrogen in amounts that matched the theoretical maximum. Importantly, this performance held up not only in purified water but also in salty solutions and in real seawater collected from the coast, without needing a separate desalination step.
Why Light Matters More Than Simple Heating
The scientists compared several ways of driving the reaction and found that shining light directly on the droplets was far more effective than just heating the water to the same temperature. Light does two jobs at once: it warms the gallium, and it also interacts with the thin layer of solid material that forms on the droplets, helping to move electric charges across the interface. Those charges make it easier for the metal to keep reacting with water. Experiments using different lamps, a solar simulator, and focused natural sunlight all showed that visible light in particular is very good at triggering this “photothermal” behavior, combining light and heat to accelerate hydrogen production.
From Lab Concept to Future Energy Carrier
From an energy-accounting viewpoint, the researchers estimate that the full cycle—from gallium reacting with water to produce hydrogen, through regeneration of the metal—could reach a round-trip efficiency of about 13 percent when solar light is treated as free input. While gallium itself is not cheap, it is reusable, relatively non-toxic, and easy to transport and store as a compact energy carrier. The study suggests a future scenario in which gallium could be shipped to coastal sites, reacted with seawater under sunlight to produce hydrogen on demand, and then returned for regeneration using clean electricity. In simple terms, the work demonstrates a promising way to turn sunshine and seawater into a storable, clean fuel using a recyclable liquid metal, bringing the vision of a hydrogen-based energy system a step closer.
Citation: Campos, L.G.B., Allioux, FM., Fimbres Weihs, G. et al. Low temperature and rapid photothermal oxidation of liquid gallium for circular hydrogen production. Nat Commun 17, 1890 (2026). https://doi.org/10.1038/s41467-026-68664-1
Keywords: hydrogen production, liquid gallium, solar energy, seawater fuel, circular chemistry