Clear Sky Science · en
Regulation of AP1 adaptor assembly by the bi-handed chaperone MEA1
Why this hidden cell helper matters
Inside every human cell, thousands of tiny parcels shuttle proteins to where they are needed or to the cellular “recycling center” for breakdown. This traffic keeps our cells healthy and our immune defenses in check. The paper summarized here uncovers a previously mysterious protein, called MEA1, that turns out to be essential for building one of the key sorting machines that load these parcels. Understanding how MEA1 works helps explain how cells control important cargos, including molecules that switch antiviral immune responses on and off.
Traffic control at the cell’s shipping hub
Much of the cell’s shipping activity runs through a structure called the Golgi, a stack of flattened sacs that acts like a central post office. From the Golgi, cargo can be sent out for use at the cell surface or rerouted to internal compartments for degradation. A protein complex known as AP1 sits at the heart of this route. AP1 recognizes tags on cargo proteins, helps bend the membrane, and recruits an outer coat of clathrin to form small transport vesicles. Without correctly assembled AP1, crucial cargos build up in the wrong places and normal signaling pathways, including those linked to immunity, can go awry. 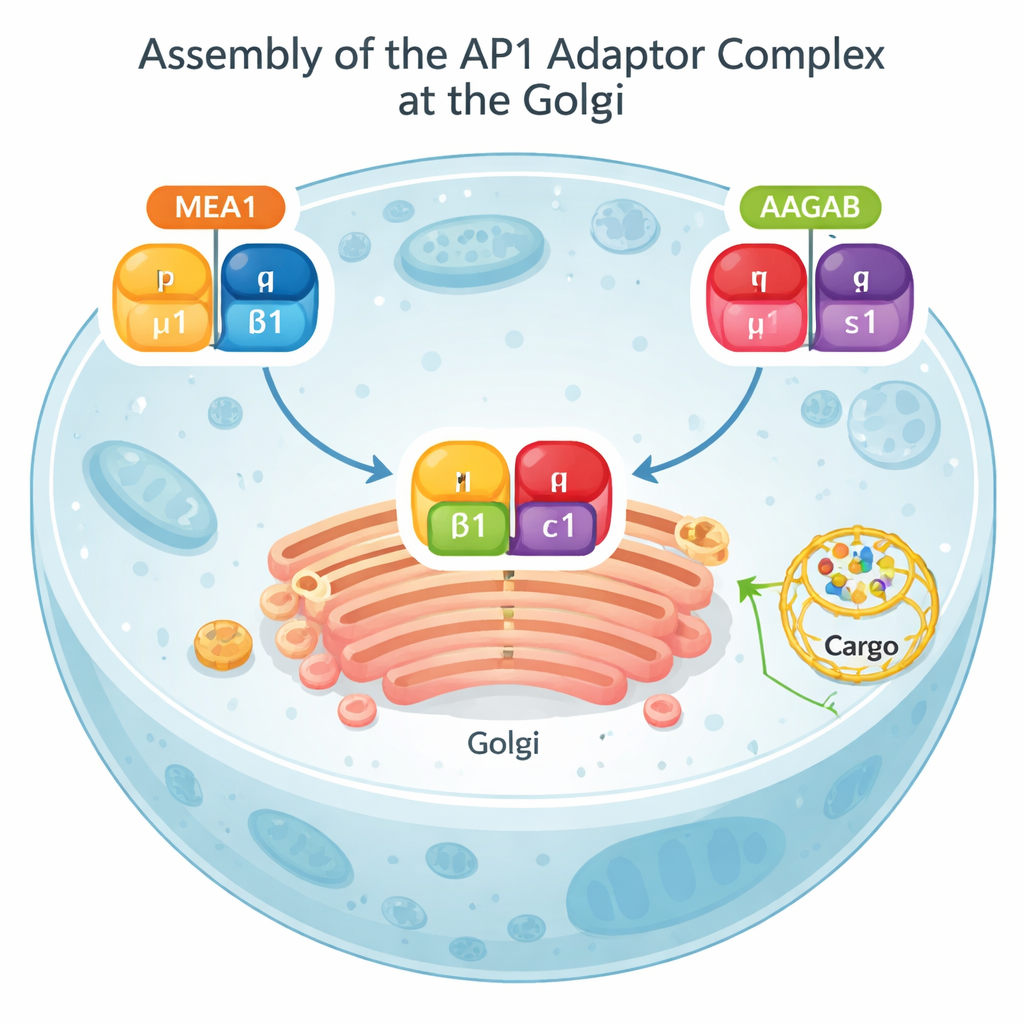
Finding a missing assembly worker
The authors set out to identify proteins that physically associate with AP1 and might help it assemble. They mined a large protein–interaction database, filtered out candidates unlikely to work with AP1, and then used an artificial-intelligence tool, AlphaFold, to predict how the remaining proteins might contact AP1’s individual parts. This search highlighted MEA1, a small and previously uncharacterized protein present in many tissues. Follow-up experiments in human cells confirmed that MEA1 binds two specific AP1 subunits, known as μ1 and β1, both when tested on their own and when examined in their natural, unmodified forms.
MEA1 keeps AP1 intact and cell cargo moving
To probe what MEA1 actually does, the researchers deleted the MEA1 gene in human cell lines. When MEA1 was missing, all four pieces of AP1 were drastically reduced, and characteristic AP1 spots near the Golgi nearly disappeared. This loss had clear functional consequences. One AP1 cargo, the folate receptor FOLR1, accumulated at the cell surface instead of being sent inward, a hallmark of defective AP1 sorting. Another cargo, the immune signaling protein STING, was no longer efficiently packed into vesicles bound for lysosomes, the cell’s recycling centers. As a result, activated STING and its partner kinase TBK1 persisted at high levels, indicating that the “off switch” for this antiviral pathway was not working properly. Reintroducing MEA1 restored AP1 levels and corrected these trafficking defects.
A two-handed chaperone and a collision-based build process
Biochemical tests and AlphaFold models revealed that MEA1 behaves like a “bi-handed” assembly chaperone: its front portion grips the μ1 subunit, while its back portion grips β1. On their own, μ1 and β1 are unstable and tend to clump or be destroyed; bound to MEA1, they become soluble and ready to assemble. Another known chaperone, AAGAB, performs a parallel job for the remaining two AP1 subunits, γ and σ1. When MEA1–μ1–β1 and AAGAB–γ–σ1 complexes meet, their subunits combine into a complete, four-part AP1 adaptor. At that point, both chaperones let go and return to the cell fluid, while the newly formed AP1 moves to membranes to do its sorting work. This “dual-chaperone collision” mechanism shows that AP1 assembly is carefully orchestrated rather than left to chance encounters.
Broader lessons about cellular quality control
Beyond AP1, the study hints at a more general strategy that cells may use to build many large protein machines. MEA1 can also weakly assist with another adaptor complex called AP2, and together with AAGAB and a third chaperone named CCDC32, it illustrates a broader pathway the authors term “Chaperone-assisted Adaptor Protein Assembly.” In this scheme, specialized chaperones stabilize fragile subunits, bring the right pairs together, and temporarily block cargo-binding sites until the complex is fully formed and safe to deploy. 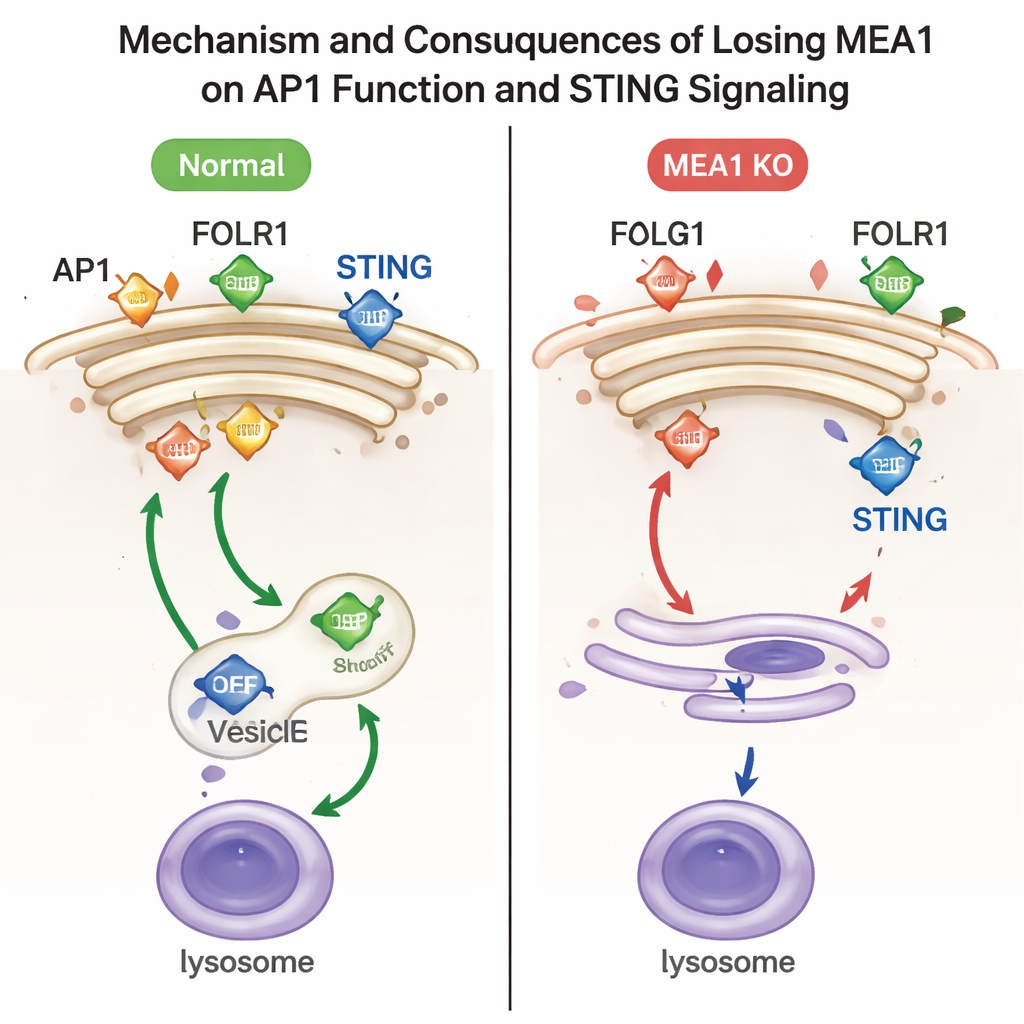
What this means for health and disease
For non-specialists, the key takeaway is that MEA1 is a crucial behind-the-scenes worker that helps assemble AP1, a core sorting machine in our cells. When MEA1 is absent, AP1 falls apart, traffic jams form, and important regulators like STING are not properly switched off. This new understanding may eventually help explain certain immune disorders or other diseases tied to faulty protein traffic, and it reveals a general principle for how cells safely build complex molecular machines from unstable parts.
Citation: Wan, C., Wu, J., Ouyang, Y. et al. Regulation of AP1 adaptor assembly by the bi-handed chaperone MEA1. Nat Commun 17, 1876 (2026). https://doi.org/10.1038/s41467-026-68662-3
Keywords: protein trafficking, adaptor protein AP1, molecular chaperones, STING signaling, cell biology