Clear Sky Science · en
Molecular recognition of thyroglobulin by sortilin
How Thyroid Cells Decide When to Release Hormone
Thyroid hormones help set our metabolic “thermostat,” influencing everything from heart rate to body temperature. These hormones are built and stored inside a giant protein called thyroglobulin. This study uncovers how another protein, sortilin, helps thyroid cells choose which form of thyroglobulin to pull back inside the cell so that hormone can be released into the bloodstream—a decision that ultimately affects how much thyroid hormone our bodies see.
A Storage Protein Waiting to Be Processed
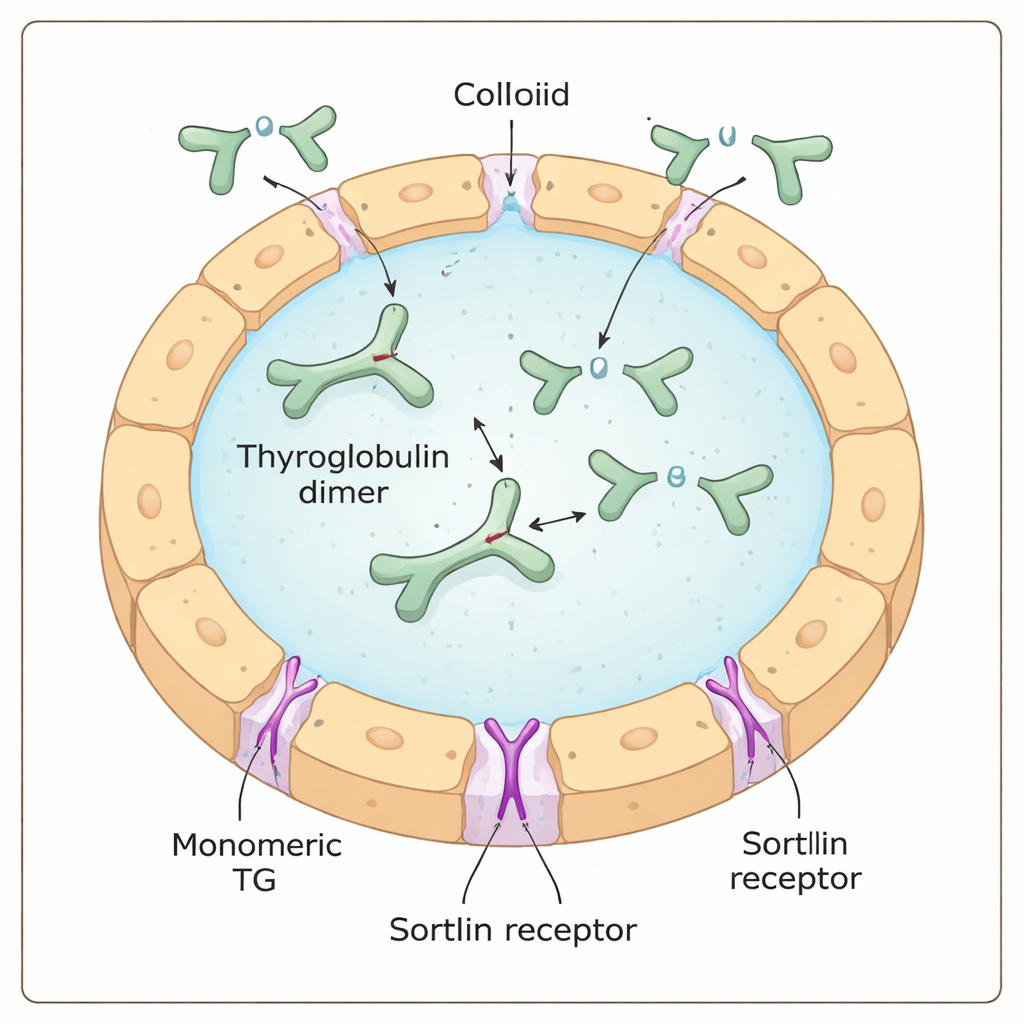
Thyroglobulin is a massive, Y-shaped protein produced and secreted by thyroid cells into a gel-like pool called the colloid. There, it acts as both raw material and warehouse for thyroid hormone: specific building blocks (amino acids) within thyroglobulin are chemically modified with iodine to become thyroid hormones, still embedded in the larger protein. To actually free these hormones, thyroglobulin must be taken back into the cell, chopped up inside recycling compartments called lysosomes, and its hormone pieces transported out to the blood.
A Cellular Gatekeeper with a Hidden Preference
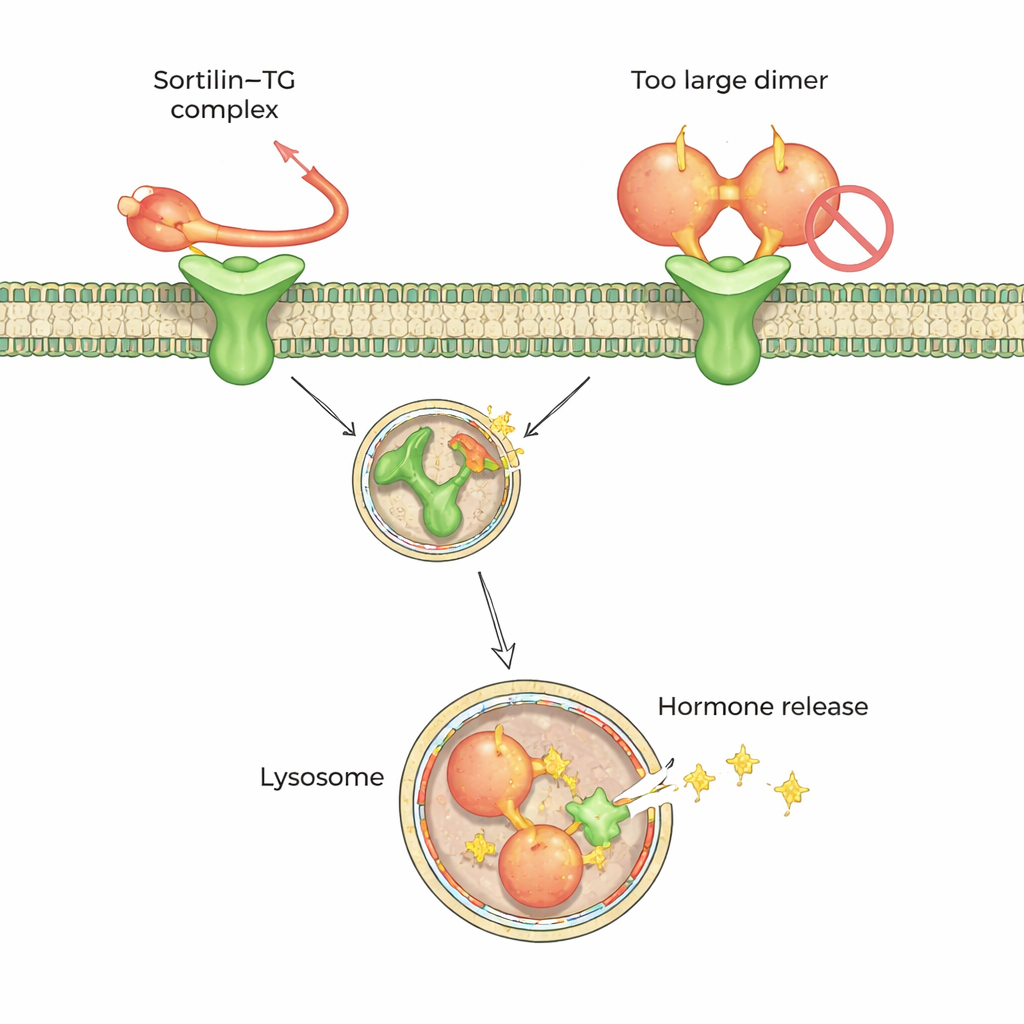
The receptor sortilin has been proposed as one of the “gatekeepers” that bind thyroglobulin at the cell surface and guide it inward. Earlier work suggested that sortilin prefers highly iodinated thyroglobulin, hinting that the receptor might somehow sense iodine content directly. Using a combination of biochemical tests, single-molecule mass measurements and cell-based uptake experiments, the authors instead found that sortilin strongly favors a single-unit, or monomeric, form of thyroglobulin over its more common paired, dimeric form. The more monomer in a sample, the more efficiently it formed complexes with sortilin and was internalized by thyroid cells, regardless of how much iodine it carried.
Zooming In on the Contact Point
To understand this preference at the atomic level, the team turned to high-resolution cryo–electron microscopy and cross-linking mass spectrometry. These methods showed that sortilin recognizes a short, floppy tail at the C-terminus (one end) of monomeric thyroglobulin. This tail threads into a central cavity of sortilin shaped like a ten-bladed propeller, docking at two small “hot spots” inside. Strikingly, in the dimeric form of thyroglobulin, part of the region around this tail is buried where sortilin cannot access it, explaining why the dimer is a poor partner. The data suggest that extracellular trimming or loosening of thyroglobulin—through natural protein degradation—helps convert dimers into monomers that sortilin can grab.
A Shared Docking Code for Many Cargo Proteins
Sortilin is not just a thyroid protein; it helps traffic many different molecules around the body, including factors linked to heart disease and brain disorders. By combining their structural work with advanced structure prediction tools such as AlphaFold and AlphaPulldown, the researchers compared how dozens of known sortilin partners might bind its propeller cavity. They found a recurring pattern: many cargoes present an unstructured peptide segment of about twenty amino acids that fits into the same pocket as thyroglobulin’s tail, sometimes running in the same direction as a known brain peptide called neurotensin, and sometimes in the opposite direction. Despite the reversed orientation, these peptides share similar features—an acidic or negatively charged group at one end, a bulky aromatic residue at the other, and a flexible, often proline-rich stretch in between.
Why Iodine Matters Less than Shape
Because the key contact between sortilin and thyroglobulin is this flexible tail, the authors tested whether decorating a hormone-forming tyrosine in the tail with extra iodine would change binding. It did not: a synthetic peptide carrying a fully formed thyroid hormone behaved almost identically to the unmodified version. Modeling showed that the iodinated ring sticks out into solvent without creating new tight contacts. Together with the uptake experiments, this supports a revised view: sortilin does not “count” iodine atoms on thyroglobulin. Instead, it senses whether the protein has relaxed or been partially degraded enough to expose its monomeric form and tail in the right way.
What This Means for Thyroid Health
To a non-specialist, the central message is that thyroid hormone release is controlled less by a built-in iodine sensor and more by a mechanical check on protein shape and flexibility. Sortilin acts like a scanner at the cell surface, looking for thyroglobulin molecules that have been loosened or trimmed into monomers, then pulling them inward for final hormone release and iodine recycling. This work clarifies a key step in thyroid hormone biology and hints that drugs designed to block sortilin—now being explored for other diseases—could unintentionally alter thyroid hormone handling by disrupting this recognition step.
Citation: Boniardi, I., Tanzi, G., Di Ianni, A. et al. Molecular recognition of thyroglobulin by sortilin. Nat Commun 17, 2004 (2026). https://doi.org/10.1038/s41467-026-68658-z
Keywords: thyroid hormone, thyroglobulin, sortilin, protein trafficking, endocytosis