Clear Sky Science · en
RoboA reinforces planarian stem cell fate through FoxA and Anosmin1a
How Worms Regrow Their Organs
Some flatworms can regrow almost any missing body part, from their feeding tube to their brain. This remarkable ability depends on adult stem cells that can turn into many different cell types on demand. But with so much freedom, how do these cells avoid building the wrong tissue in the wrong place—for example, making stomach cells in the head instead of neurons? This study uses the planarian flatworm to uncover how a small set of signals keeps highly flexible stem cells on the right track during regeneration.
The Regenerating Worm and Its Hidden Potential
The planarian Schmidtea mediterranea is a favorite model for studying regeneration because a large population of stem cells is spread throughout its body. One key organ is the pharynx, a muscular feeding tube that sits in the middle of the worm and connects to the outside when the animal eats. Earlier work showed that a gene called foxA is crucial for rebuilding this pharynx after injury, and that only certain stem cells near the pharynx normally switch on foxA. The new study asks a deceptively simple question: what stops stem cells in other regions—especially the head and brain—from also choosing a pharynx identity?
When Guidance Goes Missing
The researchers focused on a receptor protein called RoboA, found at low levels in many cell types, including stem cells. When they reduced RoboA activity using RNA interference, animals often grew extra, misplaced pharynges after being cut. Closer inspection revealed something more subtle: even in uninjured worms, turning down RoboA caused pharynx-like neurons and muscles to appear inside the brain region. These “ectopic pharynx neurons” behaved like normal pharynx cells by gene expression, but they were now in the wrong place. Importantly, the overall body plan and brain architecture remained largely intact, suggesting that RoboA was not reshaping the entire animal, but instead was fine-tuning what nearby stem cells became.
A Three-Player Switch for Cell Fate
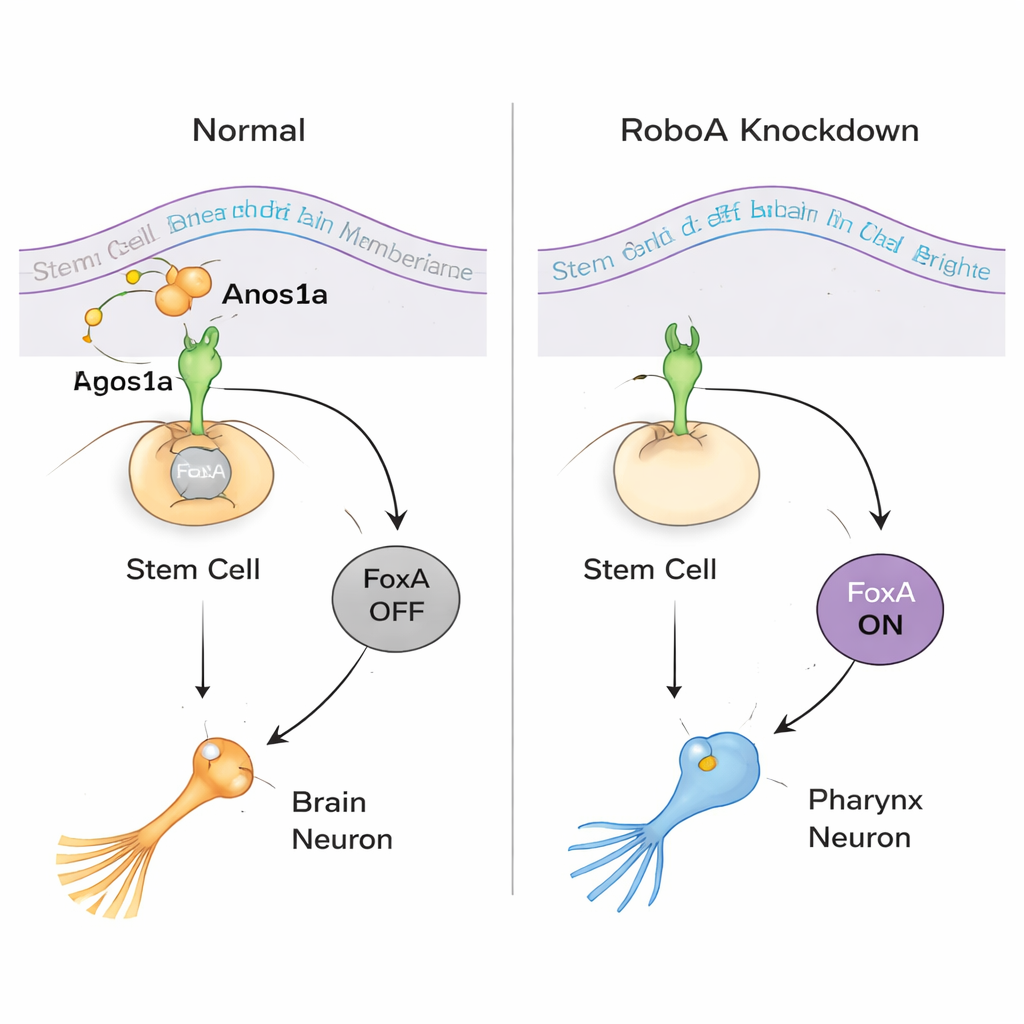
To understand how RoboA works, the team looked for partners outside the cell. Although Robo proteins are best known for binding a ligand called Slit, knocking down Slit did not reproduce the misplaced pharynx cells. A large RNAi screen of secreted and membrane proteins instead pinpointed Anosmin1a (Anos1a), a secreted protein related to a human factor involved in Kallmann syndrome. Reducing Anos1a caused similar extra pharynx neurons, and combined RoboA–Anos1a knockdowns behaved as if they acted in the same pathway. At the same time, molecular profiling showed that the transcription factor FoxA sits at the heart of the decision: when RoboA is present, it keeps FoxA off in head stem cells; when RoboA signaling is removed, FoxA switches on and those same stem cells can choose a pharynx neuron fate even while sitting in the brain.
Revealing a Two-Way Choice in Stem Cells
The team then asked whether this flexibility runs in both directions. In normal animals, stem cells around the pharynx rely on FoxA to become pharynx neurons and epithelial cells, while pharynx muscles follow a different route. When FoxA was knocked down for longer periods, worms lost their pharynxes entirely and grew abnormal outgrowths in the middle of the body. Single-cell RNA sequencing and marker analysis revealed that these outgrowths contained many cell types normally restricted to the head, including eye cells and brain-specific neurons. In other words, when FoxA is missing where the pharynx should form, local stem cells default toward a brain-like fate. This finding shows that the same stem cells can be pushed toward either “pharynx” or “brain” identities depending on which signals they receive.
Fine-Tuning Regeneration, Not Redrawing the Map
Putting all the evidence together, the authors propose that planarian regeneration is guided in two layers. Broad “position control” signals, such as Wnt and other patterning molecules, set up the rough map of head, trunk, and tail. On top of that, local “fate-reinforcing” genes like RoboA and Anos1a act as safety checks, blocking inappropriate options. In the head, RoboA–Anos1a signaling keeps FoxA off so stem cells produce brain neurons instead of pharynx cells; near the pharynx, FoxA is allowed to turn on and drive pharynx-specific fates. This layered control lets planarian stem cells remain extraordinarily plastic while still rebuilding organs in the right place, offering a blueprint for how robust regeneration can coexist with strict anatomical order.
Citation: Wang, KT., Tsai, FY., Chen, YC. et al. RoboA reinforces planarian stem cell fate through FoxA and Anosmin1a. Nat Commun 17, 1971 (2026). https://doi.org/10.1038/s41467-026-68656-1
Keywords: planarian regeneration, stem cell plasticity, organ patterning, RoboA signaling, FoxA transcription factor