Clear Sky Science · en
Atypical pericapillary Ly6G⁺Nur77⁺ macrophages initiate type-2 immune responses to allergens in the mouse lung
Why dust mites matter to your lungs
For millions of people with asthma or hay fever, everyday dust can trigger coughing, wheezing, and itchy eyes—but how the body first senses these harmless-looking particles has been a mystery. This study uses mice to uncover a previously unknown group of "sentinel" cells in the lung that detect house dust mite allergens and kick-start the kind of immune reaction that drives allergies. Understanding this early alarm system could point to new ways to prevent or calm allergic inflammation before it spirals into full-blown disease. 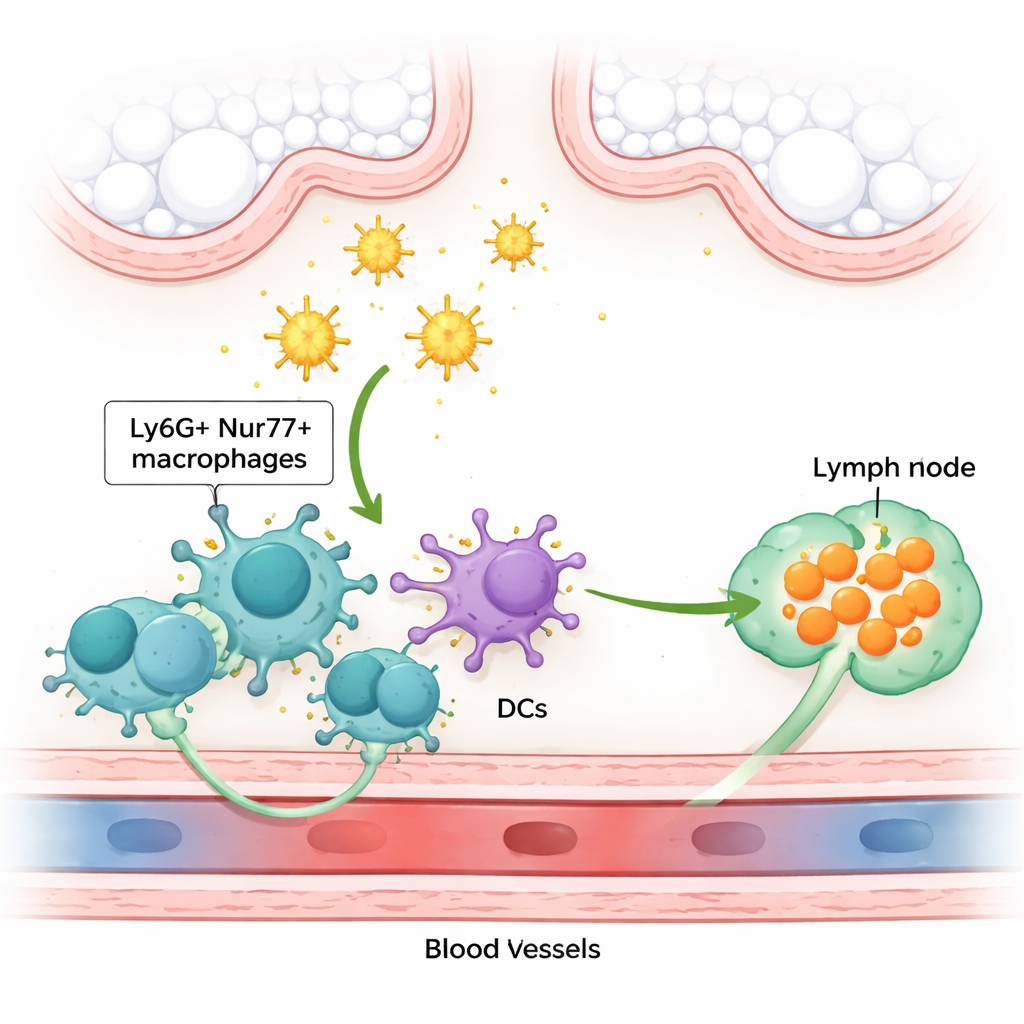
A hidden alarm system in lung blood vessels
The researchers focused on house dust mite extract, one of the most common indoor allergy triggers worldwide. Rather than treating the allergen as a simple foreign invader, the team asked a different question: does the immune system react to what the allergen does to tissues? Many mite proteins behave like scissors, cutting other proteins. The scientists showed that a specific cutting activity—called cysteine protease activity—from dust mites and related model allergens is absolutely required to spark a classic type-2 response, the pathway associated with asthma and other allergic diseases. When this cutting activity was blocked, mice failed to develop allergy-driving T helper 2 (Th2) cells and eosinophils in their lungs, even though the allergen itself was still present.
Meet the perivascular “sentinel” macrophages
Diving deeper, the team discovered an unusual set of immune cells sitting right next to tiny lung blood vessels. These cells look like macrophages—professional eaters that engulf particles—but they carry an unexpected surface marker (Ly6G) more often seen on neutrophils, and a nuclear protein called Nur77 (also known as Nr4a1), which helps control gene activity. Because of their position wrapped around capillaries, the authors call them pericapillary Ly6G⁺Nur77⁺ macrophages. These sentinels are especially good at grabbing inhaled allergens: although they make up only about 1% of lung cells, they account for a large share of the cells that actually internalize dust mite or papain allergens. They also form a stable, self-renewing population seeded early in life, distinct from the better-known alveolar and interstitial macrophages.
How allergen sensing gets translated into an allergic response
Pericapillary Ly6G⁺Nur77⁺ macrophages detect allergen activity using a surface receptor called PAR2, which is switched on when proteases cut it. Once activated by protease-rich allergens, these macrophages multiply in the lung and launch a chemical signal that reshapes the next steps of the immune response. They do not themselves travel to lymph nodes; instead, they control the movement of conventional dendritic cells, the cells that carry allergen fragments to nearby mediastinal lymph nodes where T cells are primed. The macrophages achieve this by producing cysteinyl leukotrienes—lipid mediators better known in asthma treatment—especially LTC₄. These molecules boost the ability of dendritic cells, via their CCR7 receptor, to migrate toward a guiding signal called CCL21 and reach the lymph node efficiently. 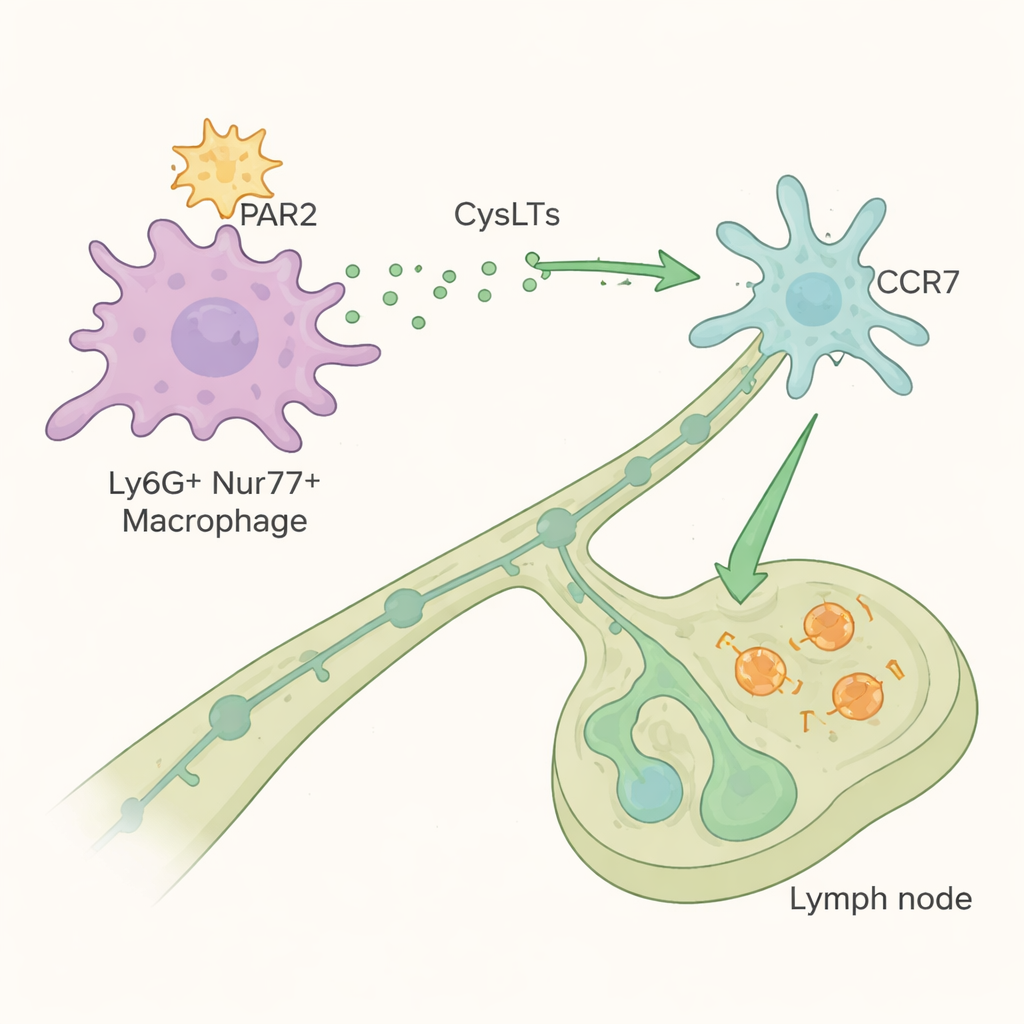
Switching off the signal without shutting down all immunity
Using genetic models and bone marrow chimeras, the authors showed that both PAR2 and Nur77 are intrinsically required in these atypical macrophages for them to expand, make leukotrienes, and drive dendritic cell migration. Mice lacking functional Ly6G⁺Nur77⁺ macrophages or their leukotriene-synthesizing enzymes showed poor dendritic cell arrival in lymph nodes, weak expansion of allergen-specific T cells, and blunted Th2 inflammation in the lung. Crucially, this pathway seemed selective for protease-rich allergens: responses to viral infection or a bacterial signal (LPS) were preserved, suggesting that blocking it would not cripple overall immunity. When the team pharmacologically blocked the enzyme LTC₄ synthase—the step that generates cysteinyl leukotrienes—they could sharply reduce dendritic cell migration, T-cell priming, and allergic lung inflammation even when the drug was given only during sensitization or only during later allergen challenge.
What this means for people with allergies
In plain terms, this work identifies a specialized group of lung macrophages that sit by the capillaries, sense the “scissor” activity of dust mite allergens via PAR2, and then call in other immune cells using leukotrienes to launch a type-2 allergic response. By targeting leukotriene production at its source, rather than just blocking one leukotriene receptor as current asthma drugs do, it may be possible to more precisely turn down allergy-specific inflammation while leaving healthy immune defenses intact. This perivascular macrophage–leukotriene axis offers a new conceptual handle on why some environmental proteins become allergens and highlights a promising route for future therapies aimed at preventing or dampening allergic airway disease.
Citation: Meloun, A., Bachus, H., Lewis, C. et al. Atypical pericapillary Ly6G⁺Nur77⁺ macrophages initiate type-2 immune responses to allergens in the mouse lung. Nat Commun 17, 1946 (2026). https://doi.org/10.1038/s41467-026-68652-5
Keywords: allergic asthma, house dust mite, lung macrophages, leukotrienes, type 2 immunity