Clear Sky Science · en
Stenotrophomonas promotes gastrointestinal tumor progression via STING degradation in tumour cells and mitigated immune response
Hidden Germs Inside Tumors
Most people have heard that the microbes in our gut can affect digestion and even mood, but few realize that living bacteria can actually take up residence inside cancer cells themselves. This study uncovers how a common environmental bacterium, Stenotrophomonas, hides within gastrointestinal tumors and quietly helps them grow, while at the same time dulling the body’s immune defenses and making modern cancer immunotherapies less effective.
Bacteria Living in Cancer Cells
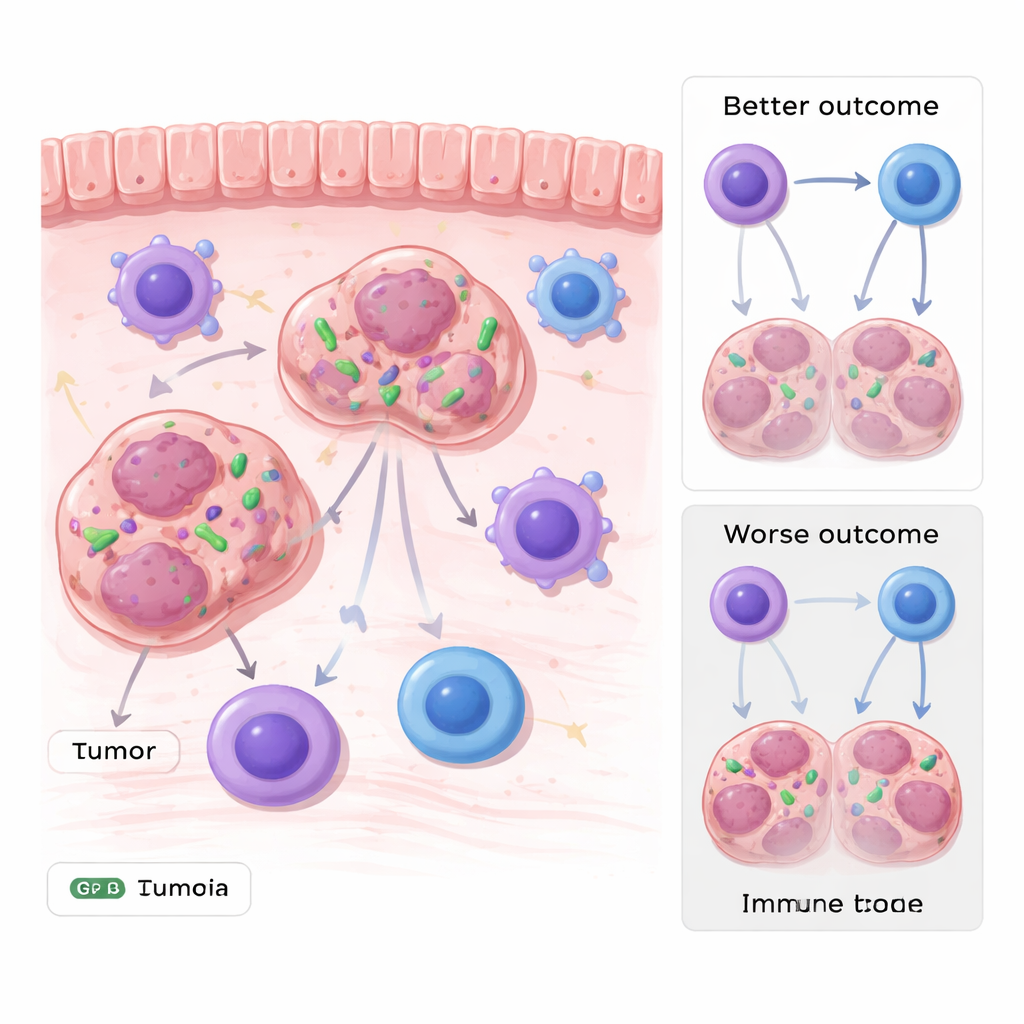
Gastrointestinal tumors, such as stomach and colorectal cancers, grow in constant contact with the rich bacterial communities of the digestive tract. By mining multiple genetic datasets from patients and then validating their findings in tumor samples, the researchers found that DNA from Stenotrophomonas was unusually abundant inside several types of gastrointestinal tumors. Importantly, its presence was not just surface contamination: imaging methods, including fluorescent probes and electron microscopy, revealed whole bacteria sitting inside the cytoplasm of tumor cells. Patients whose tumors contained higher levels of this bacterium tended to do worse over time, linking the microbe to poorer prognosis.
How a Microbe Tips the Immune Balance
The team next asked whether these hidden bacteria merely coincided with aggressive tumors or actively helped drive disease. In mouse models, they infected tumor cells with fluorescently labeled Stenotrophomonas before implanting them into animals. Tumors that carried the bacterium grew faster and larger than those without it, across several cancer cell types. When the scientists examined the surrounding immune landscape, they saw that the total numbers of immune cells, including T cells and macrophages, were similar. The critical difference lay in a specialized subset of macrophages that display a molecule called MHC-II, which helps them present tumor fragments to killer T cells. In tumors harboring Stenotrophomonas, these MHC-II–positive macrophages were markedly reduced, and T cells showed weaker cancer-killing activity.
Short-Circuiting a Cellular Alarm System
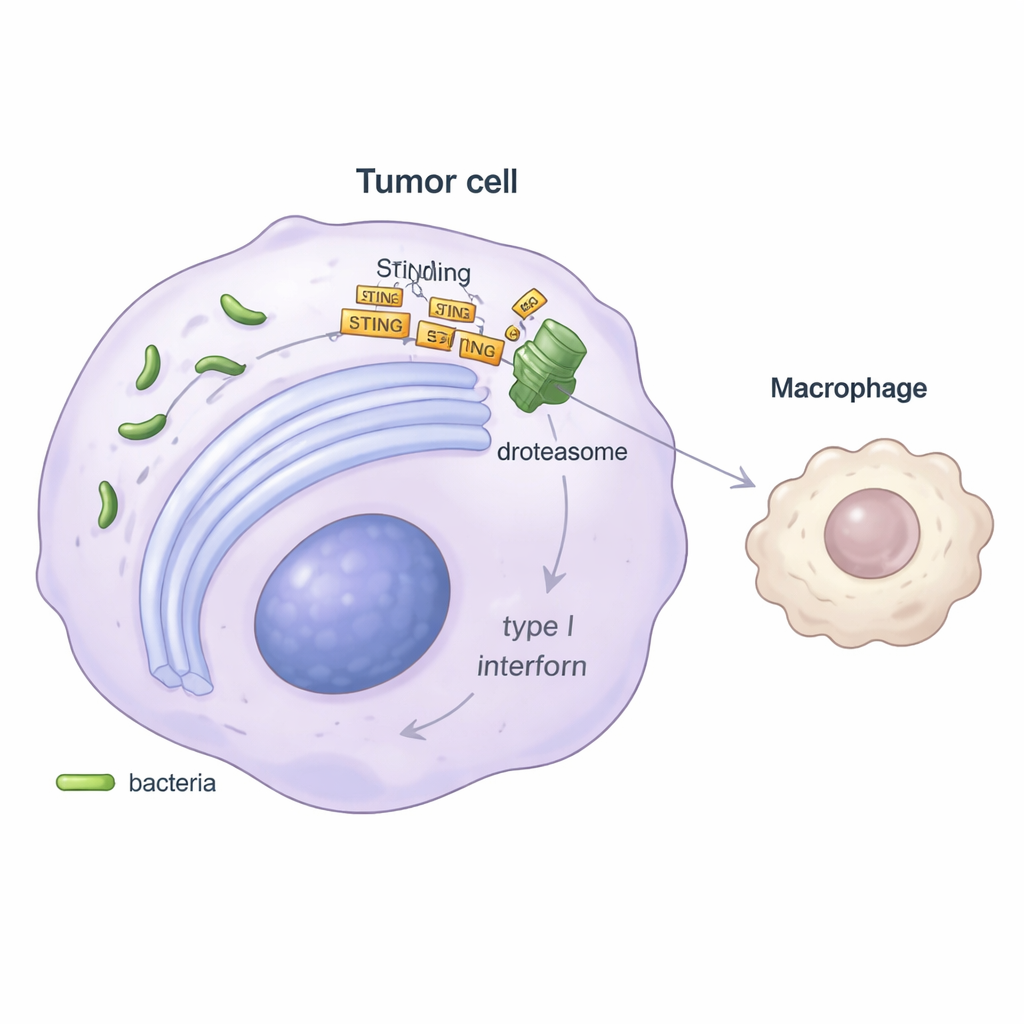
To understand the mechanism, the researchers focused on a key alarm pathway inside cells known as STING, which normally senses foreign DNA and triggers the release of antiviral “danger” signals called type I interferons. In laboratory experiments, tumor cells infected with Stenotrophomonas produced much less interferon beta, and genes normally switched on by this pathway were muted. The study showed that the bacterium induces stress in the cell’s protein-folding factory, the endoplasmic reticulum, and then hijacks a quality-control route called ER-associated degradation. Through a protein helper named SEL1L, this pathway tags the STING protein for destruction in the cell’s proteasome “shredder.” With STING levels depleted, the tumor cell sends out a weaker alarm, leading to less interferon and, downstream, blunted activation of MHC-II–positive macrophages.
Undermining Immunotherapy—and a Possible Fix
Because modern immunotherapies like anti–PD-1 drugs rely on vigorous T cell responses, the team tested how intracellular Stenotrophomonas affected treatment. In mice, tumors carrying the bacterium responded poorly to anti–PD-1 compared with bacteria-free tumors, even when combined with chemotherapy. T cells in these tumors showed reduced markers of activity and division, consistent with poor immune stimulation. Strikingly, when the researchers treated animals with the antibiotic levofloxacin, which effectively kills Stenotrophomonas, tumor growth slowed and responsiveness to anti–PD-1 therapy improved. Restoring interferon signaling with a clinical interferon drug could also rescue the population of helpful MHC-II–positive macrophages and revive T cell killing in some settings, underscoring the central role of this pathway.
What This Means for Patients
For a lay reader, the key message is that some gastrointestinal cancers may be “armed” with their own internal microbiome that helps them escape immune attack. Stenotrophomonas, by degrading the STING alarm system inside tumor cells, dampens immune signals that would normally rally macrophages and killer T cells. This not only speeds tumor growth but may also help explain why certain patients fail to benefit from powerful immunotherapies. Although more work is needed before this can change clinical practice, the findings raise the possibility that testing tumors for such bacteria—and selectively treating them with targeted antibiotics or drugs that stabilize STING—could one day improve outcomes for people with stomach and colorectal cancers.
Citation: Feng, H., Chen, K., Xi, Z. et al. Stenotrophomonas promotes gastrointestinal tumor progression via STING degradation in tumour cells and mitigated immune response. Nat Commun 17, 2042 (2026). https://doi.org/10.1038/s41467-026-68649-0
Keywords: tumor microbiome, gastrointestinal cancer, Stenotrophomonas, STING pathway, cancer immunotherapy