Clear Sky Science · en
NR6A1 is essential for neural crest cell specification, formation and survival
How early cell decisions shape the face and body
Before we are born, a special group of cells called neural crest cells travel through the embryo to help build the face, heart, nerves, and more. When these cells go wrong, babies can be born with serious birth defects. This study uncovers a previously underappreciated molecular switch, a protein called NR6A1, that helps decide when and where neural crest cells form in mammal embryos, and what happens when that switch fails.
The wandering builders of the embryo
Neural crest cells arise very early in development along the border of the forming brain and spinal cord. After breaking away from this tissue, they migrate long distances to create many different structures, from facial bones and cartilage to parts of the peripheral nervous system and pigment cells in the skin. Because they contribute to so many organs, even small disruptions in their formation can cause wide-ranging conditions known as neurocristopathies, including craniofacial syndromes such as Treacher Collins. Understanding how these cells first appear is therefore central to explaining both normal development and birth defects.
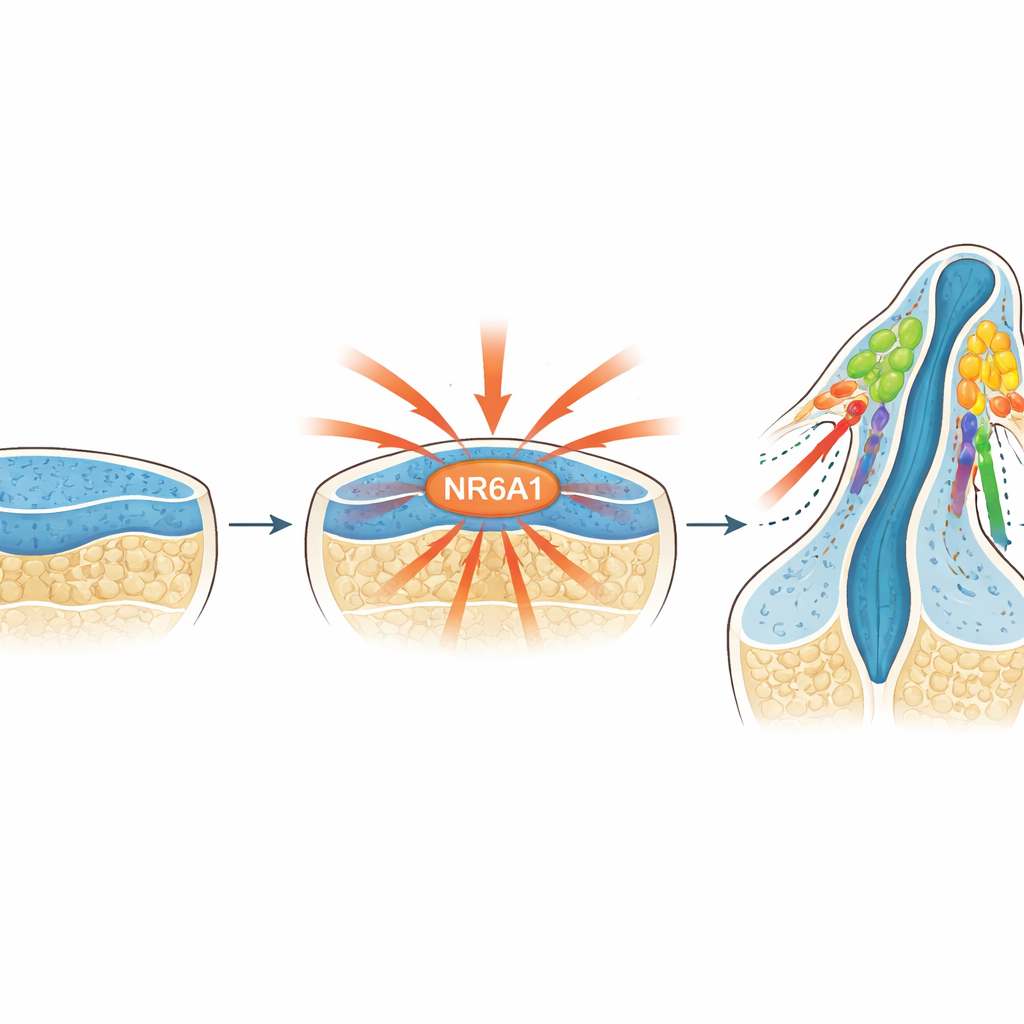
A newly recognized master switch
The researchers focused on NR6A1, a member of the nuclear receptor family of proteins that control gene activity. Earlier work hinted that NR6A1 might be involved in head and face defects, but its role in neural crest cells was unknown. By tracking NR6A1 in mouse embryos from just after implantation onward, the team found that its presence closely matched where and when neural crest cells are normally born: it appears in the early neural tissue, becomes concentrated along the dorsal (back) side of the developing brain and spinal cord, and is also seen in cells just as they leave this tissue to begin migrating.
What happens when the switch is missing
Mice completely lacking NR6A1 die early in development, so the team examined embryos just before this point. They observed a severe shortage of neural crest cells, particularly behind the first arch that will form parts of the jaw and neck, where migratory neural crest cells were almost entirely absent. Key gene programs that normally mark neural crest identity and enable cells to detach and move were sharply reduced. At the same time, genes that keep cells in a stem-like, highly proliferative state remained switched on, and many neural crest–like cells that did form soon died. These findings indicate that without NR6A1, neural stem cells fail to properly transition into migratory neural crest cells and cannot survive.
Flipping off stemness and turning on movement
To understand how NR6A1 exerts this control, the authors combined several powerful molecular techniques, including single-cell RNA sequencing, chromatin accessibility mapping, and targeted DNA–protein binding assays in both mouse and human stem-cell–derived neural crest cells. They found that NR6A1 physically binds to specific DNA motifs near genes that govern two opposing programs: one that maintains pluripotency (the ability to become many cell types) and another that drives neural crest identity and cell movement. When NR6A1 is present, chromatin surrounding stemness genes such as Oct4 and Nanog becomes less accessible and these genes are turned down, while regions near neural crest and epithelial-to-mesenchymal transition genes become more open and active. Overexpressing Oct4 during the critical window of neural crest formation mimicked the loss of NR6A1, further showing that stemness must be silenced for neural crest cells to emerge.
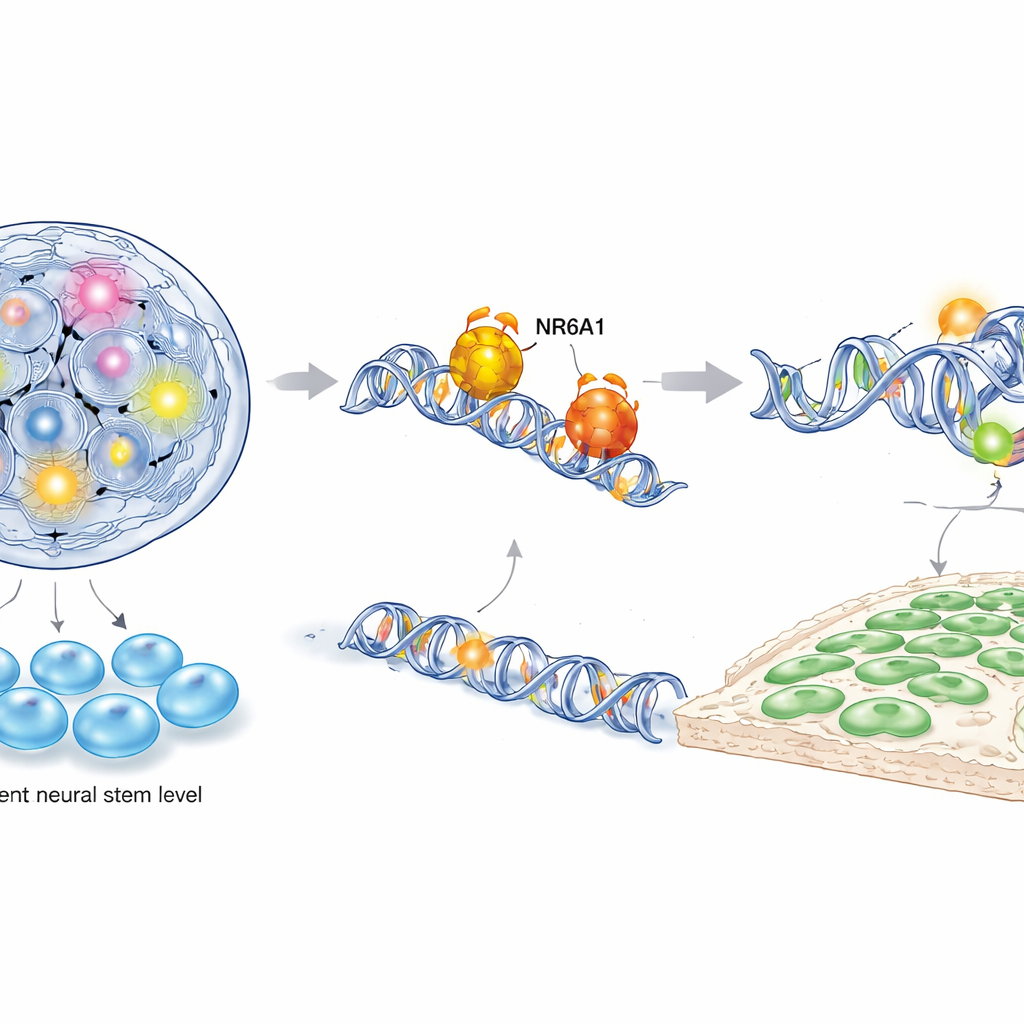
Rewriting the timeline of early development
The study also revises the calendar of when neural crest cells begin to be specified in mammals. Using an inducible genetic system to remove NR6A1 at different times, the team showed that deleting it by mid-gastrulation—a stage earlier than traditionally thought—was enough to block neural crest gene activation. In contrast, deleting NR6A1 later, including with a widely used neural crest–specific driver line, produced no obvious defects. This means that crucial decisions about neural crest fate are made before those standard tools even start working, explaining why some previous mouse studies may have missed early requirements.
Why this matters for birth defects and disease
Altogether, the work reveals NR6A1 as a central coordinator that helps neural stem cells “grow up” into neural crest cells by simultaneously shutting down pluripotency programs and enabling migration and survival programs. This dual action appears to begin during mid-gastrulation, earlier than previously recognized in mammals, and aligns mouse development more closely with that of other vertebrates. By clarifying when and how this switch operates, the study provides a framework for reinterpreting earlier genetic experiments, identifying better tools for studying neural crest biology, and ultimately understanding and potentially preventing neurocristopathies that arise when this early developmental decision goes awry.
Citation: Moore Zajic, E.L., Muñoz, W.A., Dennis, J.F. et al. NR6A1 is essential for neural crest cell specification, formation and survival. Nat Commun 17, 2140 (2026). https://doi.org/10.1038/s41467-026-68647-2
Keywords: neural crest cells, NR6A1, embryonic development, pluripotency switch, neurocristopathy