Clear Sky Science · en
C1q-dependent clearance of alpha-synuclein allows macrophages to transiently limit enteric synucleinopathy in male mice
Why the Gut Matters in Brain Disease
Parkinson’s disease is best known for tremors and movement problems, but many people experience years of severe constipation and other gut troubles before any motor symptoms appear. This study explores what happens in the “second brain” of the body—the nerve network in the gut—when a Parkinson’s-linked protein misfolds there, and how local immune cells first help, then falter. Understanding this early battle in the intestine could open new ways to ease symptoms and possibly slow disease progression long before the brain is heavily damaged.
A Problem Protein in the Body’s Second Brain
In Parkinson’s disease and related disorders, a protein called alpha-synuclein can misfold and clump into toxic aggregates. These clumps are found not only in the brain but also throughout the enteric nervous system, the web of neurons that controls gut movement. Many researchers suspect that, in at least some patients, misfolded alpha-synuclein may first take hold in the gut and then spread toward the brain along nerve pathways. The authors used a mouse model in which pre-formed alpha-synuclein fibrils are injected into the stomach and upper small intestine. Over the next two months, they observed a steady rise in abnormal, chemically tagged alpha-synuclein inside gut neurons. This buildup coincided with slower movement of material through the intestines and reduced stool output, mirroring the constipation seen in many people with Parkinson’s.
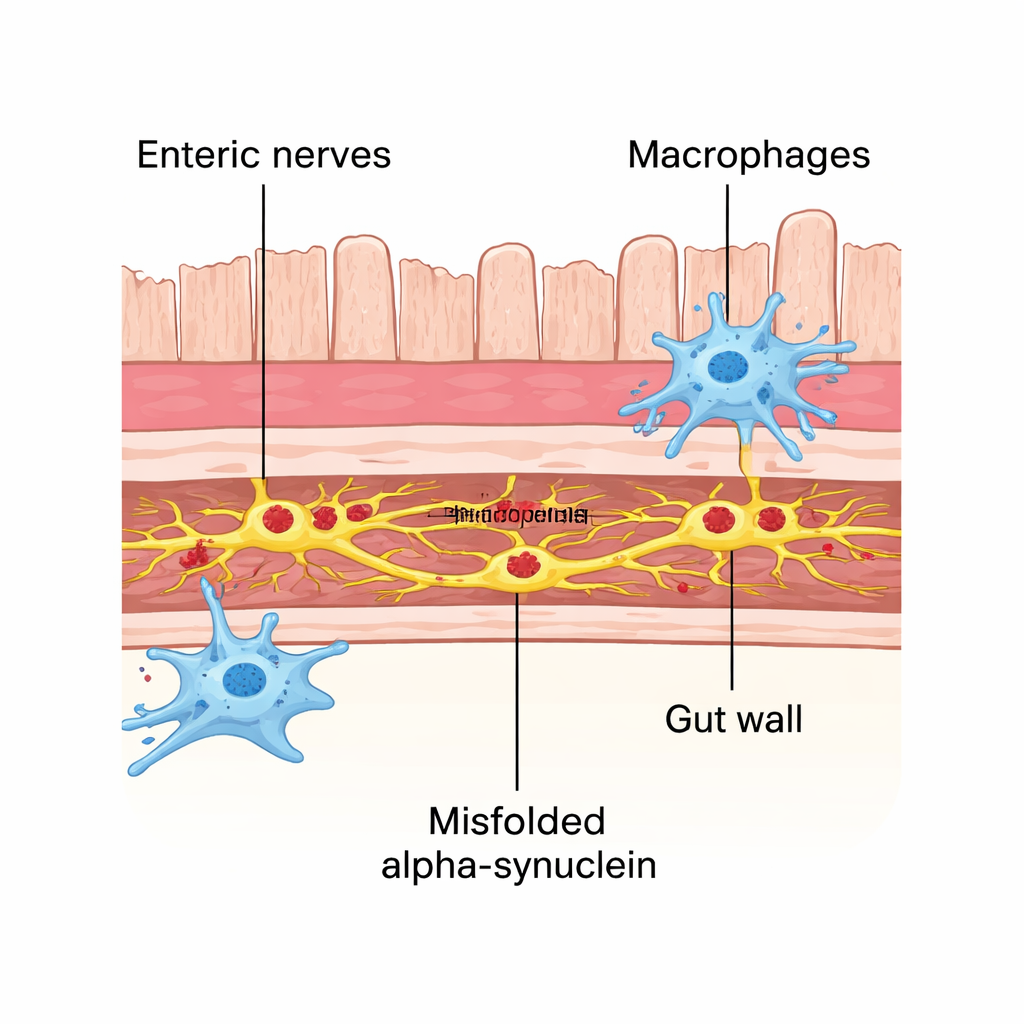
Gut Immune Cells Step In as First Responders
The gut wall contains resident immune cells called macrophages that constantly communicate with neighboring neurons and help keep the tissue healthy. In this model, these macrophages did more than simply react to damage: they physically contacted affected nerve clusters more often and harbored tiny puncta of the misfolded protein inside themselves. When the researchers depleted these macrophages using an antibody treatment, alpha-synuclein pathology in enteric neurons increased significantly. This suggested that gut macrophages play an early protective role, acting as cellular cleaners that engulf and remove toxic protein from nearby nerve cells before it can spread.
A Molecular Tag That Helps—and Hurts
To understand how these immune cells recognize what to remove, the team turned to single-cell RNA sequencing, profiling thousands of individual gut immune cells. They identified specific macrophage subtypes in the muscle layers of the intestine that switched on genes linked to engulfment and protein breakdown, particularly components of the complement system—a molecular tagging pathway better known from the bloodstream. One key player, C1q, was highly active in these gut macrophages. Under the microscope, C1q coated affected enteric neurons, and macrophages contained puncta positive for both C1q and misfolded alpha-synuclein, indicating that C1q helps label toxic protein for removal. When the scientists used mice genetically lacking C1q, neuronal alpha-synuclein pathology worsened and macrophages internalized fewer protein clumps. Yet, surprisingly, these C1q-deficient mice had somewhat better gut motility than normal mice exposed to the same fibrils, implying that the very process that clears toxic protein can also disturb gut function—likely by trimming or weakening nerve connections that control movement.
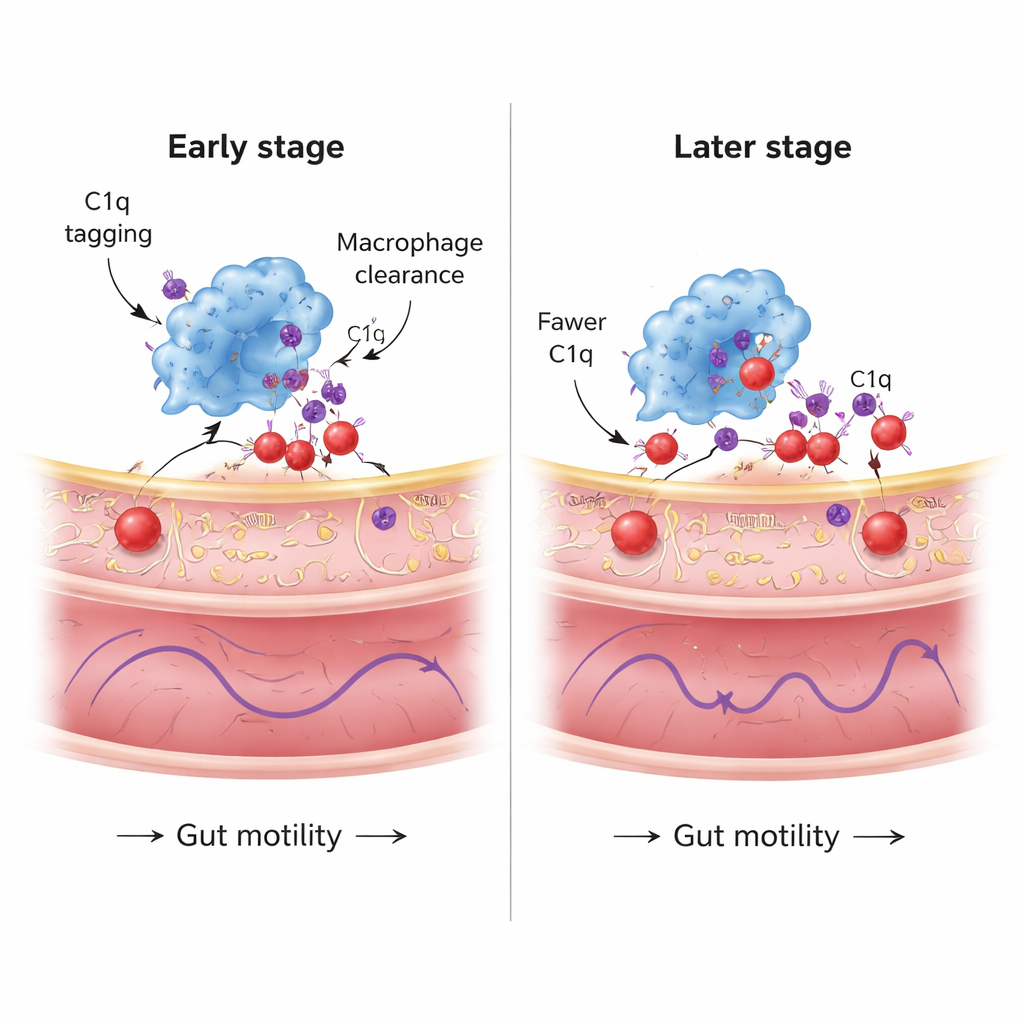
A Protective System That Fades with Time
The study also followed this cleanup response over a longer period. At around one month after alpha-synuclein was introduced, macrophages showed robust C1q expression and many C1q-tagged protein puncta within their cell bodies, and C1q heavily decorated enteric neurons. By two months, however, although overall C1q levels in macrophages remained, the number of internalized C1q/alpha-synuclein puncta and the amount of C1q deposited on neurons had declined. At the same time, neuronal pathology in the gut continued to rise. Gene analyses suggested that ongoing uptake of misfolded protein strains the macrophages’ protein-handling machinery, activating stress pathways related to misfolded proteins, lysosomes, and even cell death. In other words, the macrophages’ protective capacity appears to be finite: they initially restrain pathology, but as stress accumulates, their ability to clear alpha-synuclein wanes.
What This Means for Parkinson’s and the Gut
The work paints a nuanced picture of how gut immune cells shape early Parkinson’s-like changes in the intestine. At first, resident macrophages help by using C1q to tag and swallow misfolded alpha-synuclein from enteric neurons, limiting its spread. But this same complement-driven pruning may impair nerve signaling and slow the gut, contributing to constipation. Over time, the macrophages’ stress-burdened clearance system weakens, allowing more pathology to accumulate even as gut function and brain involvement diverge. For patients, this suggests that targeting macrophage activity or complement signaling in the intestine—aiming to boost safe clearance while avoiding excessive synapse loss—could one day be a strategy to treat or prevent Parkinson’s-related gut symptoms and perhaps influence the broader course of disease.
Citation: Mackie, P.M., Koshy, J.M., Bhogade, M.H. et al. C1q-dependent clearance of alpha-synuclein allows macrophages to transiently limit enteric synucleinopathy in male mice. Nat Commun 17, 1877 (2026). https://doi.org/10.1038/s41467-026-68641-8
Keywords: Parkinson’s disease, enteric nervous system, alpha-synuclein, gut immune cells, complement C1q