Clear Sky Science · en
Camouflaged nanorobots target and regulate macrophage subcellular organelle crosstalk patterns to promote neural regeneration
Why tiny "cloaked" helpers matter for spinal cord injuries
Spinal cord injury often means lifelong paralysis because the damaged nerve tissue is extremely hard to repair. A major reason is that the injury site becomes a hostile zone filled with inflammation and cellular stress that blocks healing. This study describes a new kind of camouflaged nanorobot that travels through the bloodstream, sneaks past the body’s defenses, and reprograms immune cells at the injury site to switch from attack mode to repair mode, helping nerves regrow in animals with spinal cord damage.
The problem: inflammation that won’t turn off
After a spinal cord injury, the first wave of damage comes from the physical trauma itself. But a second, slower wave is driven by inflammation, toxic molecules called reactive oxygen species, and widespread cell death. Immune cells known as macrophages rush in to clean up debris. Early on, they adopt an aggressive “M1” state that kills pathogens and damaged cells but also releases harsh inflammatory signals. For real healing to begin, these same cells must later shift into a calming, “M2” state that supports tissue repair, blood vessel growth, and nerve regeneration. Unfortunately, the intense stress inside injured tissue can trap macrophages in the harmful M1 state, locking in a cycle of chronic inflammation that prevents functional recovery.
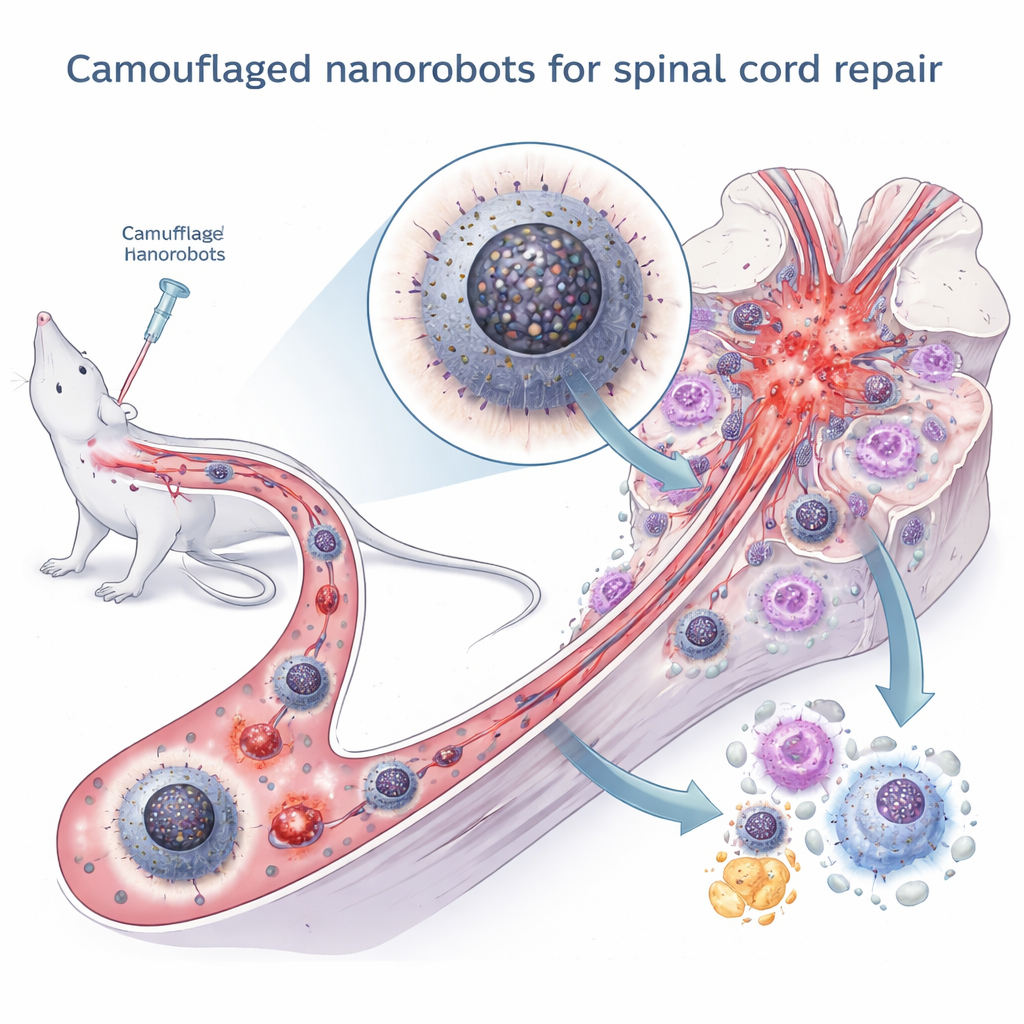
Hidden conversation between cell parts
The authors focused on a subtle but crucial internal “conversation” inside macrophages: the crosstalk between two key compartments, the endoplasmic reticulum (which helps fold proteins and stores calcium) and mitochondria (the cell’s power stations). Under normal conditions, these structures share small amounts of calcium ions through specialized contact zones, helping match energy production to the cell’s needs. After injury, however, excess reactive oxygen species cause protein misfolding and stress in the endoplasmic reticulum. This stress, in turn, drives excessive calcium transfer into mitochondria, leading to energy collapse, more toxic molecules, and leakage of mitochondrial DNA into the cell fluid. That escaped DNA triggers a powerful alarm pathway called cGAS–STING–NFκB, which further locks macrophages into the inflammatory M1 state.
Designing a cloaked nanorobot
To break this vicious cycle, the team built a multi-layered nanorobot called BP@D/N. At its core are black phosphorus quantum dots, tiny particles with strong antioxidant and anti-inflammatory activity but poor stability in the body. These dots are packed into a hollow shell of polydopamine, a biocompatible material that protects them from breaking down too soon and adds its own antioxidant properties. Finally, the entire particle is wrapped in a membrane taken from activated neutrophils, a type of white blood cell naturally drawn to sites of inflammation. This “cloak” lets the nanorobot evade immune clearance, follow inflammatory signals to the injured spinal cord, and stick to macrophages for efficient uptake.
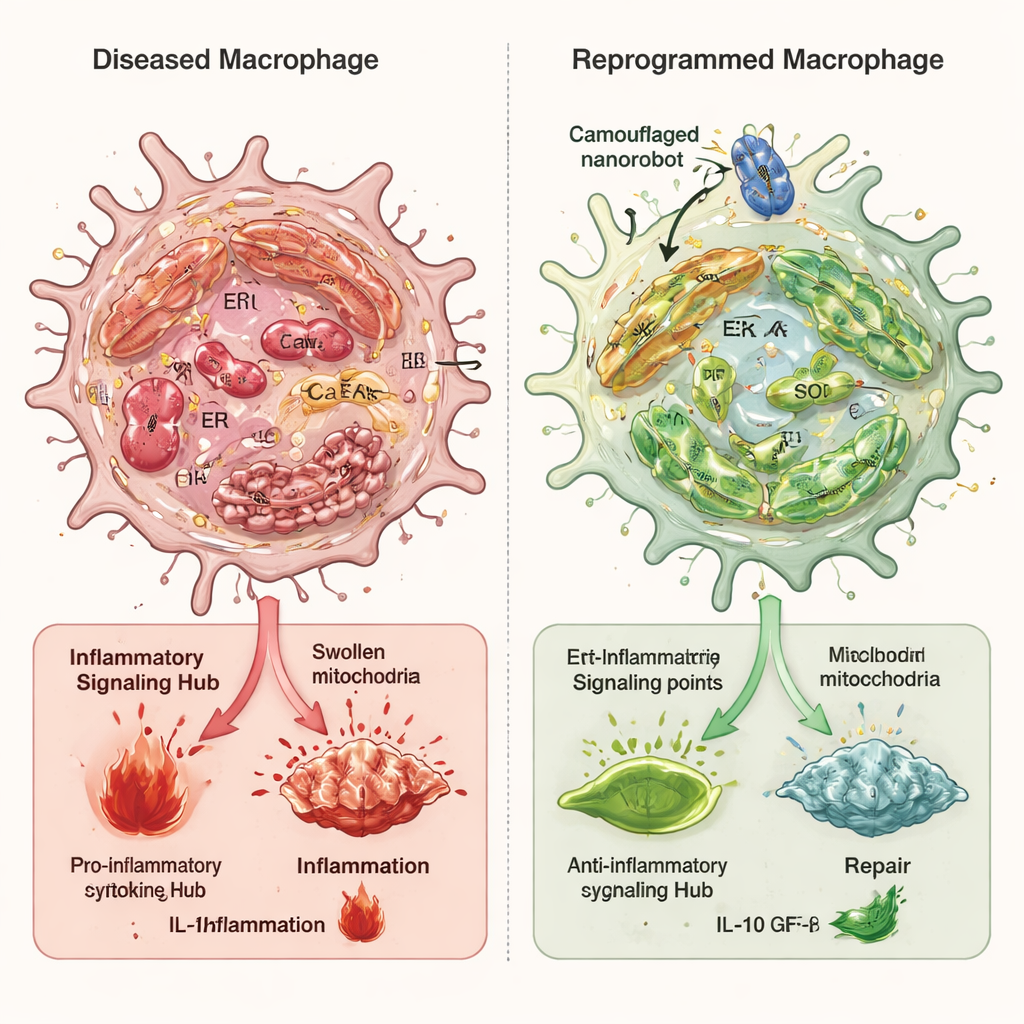
Rewiring stressed immune cells
In cell studies, macrophages exposed to inflammatory triggers showed high levels of endoplasmic reticulum stress, swollen and damaged mitochondria, calcium overload, and strong activation of the cGAS–STING–NFκB alarm pathway. When treated with the camouflaged nanorobots, overall oxidative stress fell sharply, the internal membranes between the endoplasmic reticulum and mitochondria became less overconnected, and mitochondrial calcium returned toward normal. This prevented mitochondrial DNA from leaking into the cell fluid and dialed down the inflammatory signaling cascade. A key player in this process was an enzyme called Ero1α, which helps control the oxidative environment and calcium release from the endoplasmic reticulum. The nanorobots reduced Ero1α activity, and when the researchers artificially boosted Ero1α, it largely canceled the nanorobot’s benefits, confirming its central role.
From calming inflammation to regrowing nerves
These internal changes flipped macrophages from the destructive M1 state to the healing M2 state. In dishes, the M2-skewed cells secreted fewer inflammatory molecules like TNF-α and IL-6 and more growth factors that encourage nerve cells and supporting cells to grow and extend axons. In rat models of spinal cord injury, repeated injection of the nanorobots led to fewer damaging immune signals at the lesion, less scarring, smaller cavity formation, and more regenerating nerve fibers. Animals receiving the camouflaged nanorobots regained better hind-limb movement, stronger muscle responses, and improved bladder structure compared with untreated or less targeted nanoparticle treatments.
What this could mean for future patients
This work shows that precisely tuning the internal stress and communication between cell compartments can flip immune cells from harming to helping damaged nerves. By packaging powerful but fragile antioxidant particles into a cloaked, targeted nanorobot, the researchers restored a healthier balance between inflammation and repair in the injured spinal cord of rats. While much work remains before this approach can be tested in humans, it offers a promising blueprint: instead of just suppressing inflammation broadly, future therapies might steer specific cell machinery inside macrophages to create a more welcoming environment for neural regeneration after spinal cord injury.
Citation: Guo, Q., Wang, W., Jiang, X. et al. Camouflaged nanorobots target and regulate macrophage subcellular organelle crosstalk patterns to promote neural regeneration. Nat Commun 17, 1952 (2026). https://doi.org/10.1038/s41467-026-68636-5
Keywords: spinal cord injury, nanorobots, macrophages, neural regeneration, nanomedicine