Clear Sky Science · en
Hydrogel with cell-cell adhesion cues enhances neural regeneration
Helping the Injured Brain Heal Itself
Traumatic brain injuries can leave lasting problems with movement, memory, and thinking because damaged nerve cells in the brain rarely regrow or reconnect properly. Doctors can remove blood clots or stabilize the skull, but they have few tools to rebuild the brain’s delicate wiring. This study describes a soft, water-rich material called a hydrogel that is engineered to mimic how brain cells naturally stick to each other. By recreating these tiny “handshakes” between cells, the material helps damaged nerve fibers regrow, re-form connections, and restore function in injured rat brains.
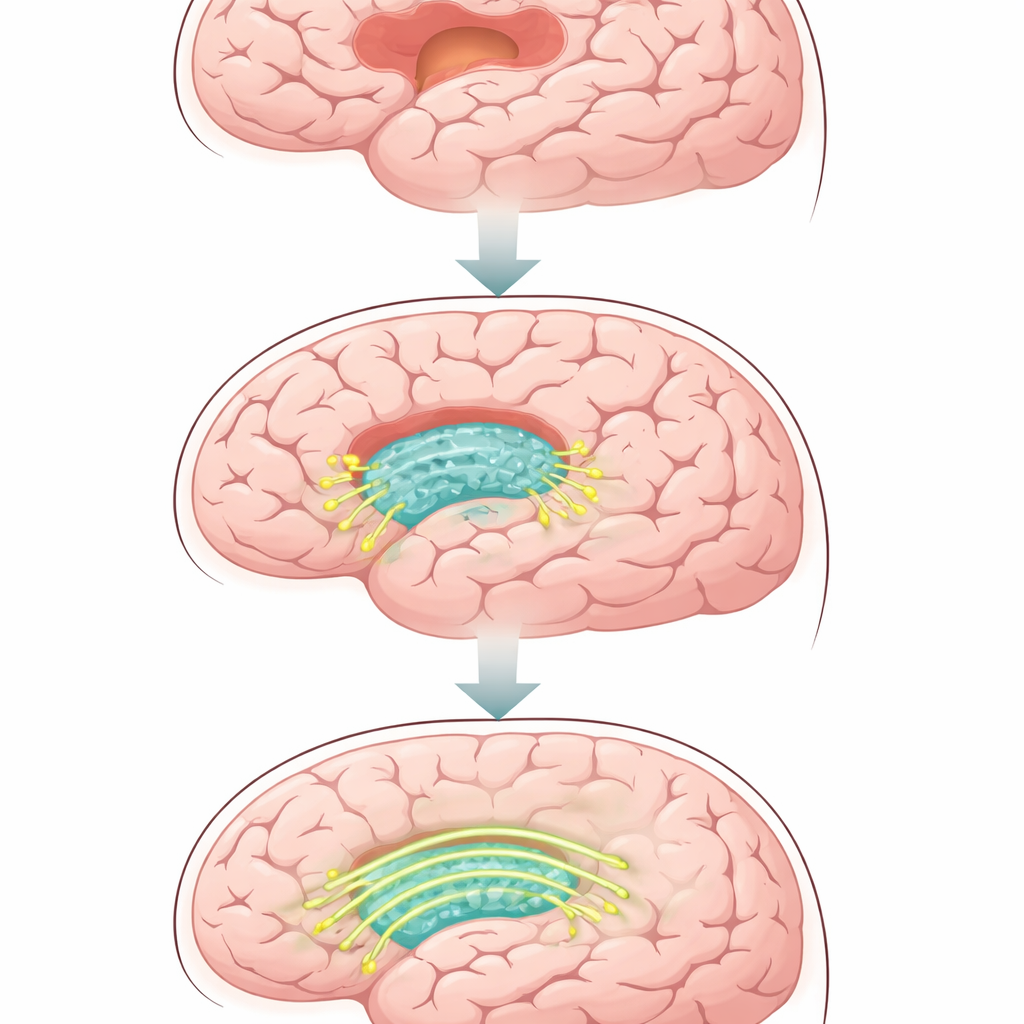
A Soft Scaffold for Broken Brain Tissue
The researchers started with a silk-based hydrogel that has a feel similar to brain tissue—very soft and flexible, with many tiny pores where cells can move and extend their branches. They then coated this gel with a thin, fluid layer of fat molecules that behaves like a cell membrane. This layer can hold special proteins while still allowing them to drift sideways, just as they do on real cells. The result is a three-dimensional, injectable material that can fill irregular brain wounds and present moving signals to nearby nerve cells, instead of being a passive filler.
Borrowing Nature’s Cell-to-Cell Handshake
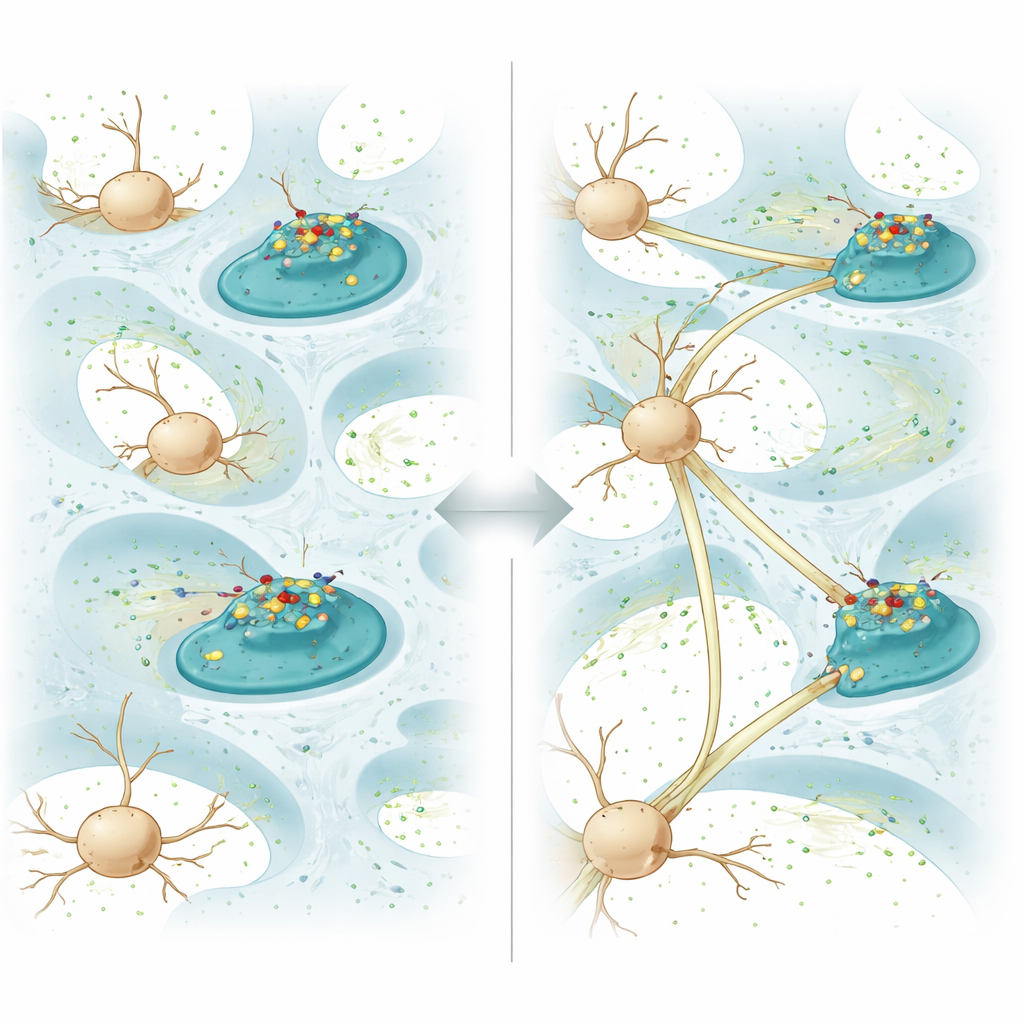
To turn the hydrogel into an active partner in healing, the team attached a protein called N-cadherin to the fatty coating. In the brain, N-cadherin sits on the surface of neurons and helps neighboring cells recognize and grip each other, forming stable contacts and, eventually, synapses where signals pass from cell to cell. In this gel, N-cadherin is not locked in place; it can diffuse freely across the coating. When neurons grow against the gel, the protein clusters at points where their membranes touch the material, reshaping the cells’ small protrusions and triggering the formation of cell-to-cell junctions. In dishes, neurons growing on this “diffusive N-cadherin” gel extended longer fibers, formed more connections, and showed stronger electrical communication than on similar gels where the protein was fixed and immobile.
Waking Up the Brain’s Repair Programs
Beyond simple contact, the hydrogel also nudges neurons to switch on internal repair pathways. Gene and protein analyses showed that cells on the diffusive N-cadherin gel reduced signals that drive programmed cell death and boosted pathways known to support growth and survival. In particular, the material increased levels of a protein called thrombospondin-1, which is released by support cells in the brain and can both promote synapse formation and activate growth-friendly signaling routes. Two of these routes, often called the TGF-β/Smad and AKT/mTOR pathways, were strongly turned on, leading to healthier mitochondria, more energy production, and enhanced regrowth of severed nerve fibers in a microfluidic model that mimics cut axons.
From Petri Dish to Living Brain
To test whether these effects mattered in a living brain, the team injected the hydrogel into rats with a controlled form of traumatic brain injury. Over several weeks, animals treated with the diffusive N-cadherin gel performed better on movement tests and on a water-maze task that measures learning and memory, compared with animals that received plain gel, lipid-only gel, or just saline. Brain scans and tissue slices revealed smaller cavities at the injury site and more newly formed nerve fibers and synapses within the repaired region. At the same time, the treated brains showed fewer overactive immune cells and less dense scar tissue, creating a more welcoming environment for regrowth. Markers of cell death were reduced, while proteins linked to healthy communication between neurons were increased.
Why This Matters for Future Brain Repair
In simple terms, this work shows that giving injured brain tissue the right kind of “social cues” can make a major difference in how well it heals. Instead of only supporting cells mechanically, the hydrogel recreates the moving contact signals that real brain cells use to find and hold on to one another. In rats, this strategy not only filled holes in the brain but also helped rebuild working neural circuits and improved behavior after injury. Although much testing remains before such materials could be used in people, the approach—embedding freely moving cell-adhesion proteins into soft, injectable gels—could be extended beyond N-cadherin and beyond the brain, offering a general recipe for materials that actively guide tissue regeneration.
Citation: Tang, X., Zhang, S., Liu, M. et al. Hydrogel with cell-cell adhesion cues enhances neural regeneration. Nat Commun 17, 2178 (2026). https://doi.org/10.1038/s41467-026-68632-9
Keywords: neural regeneration, traumatic brain injury, hydrogel scaffold, cell adhesion, N-cadherin