Clear Sky Science · en
G9a-mediated H3K9me2 orchestrates intestinal epithelial regeneration through epigenetic silencing of cell cycle-related genes
Why Gut Self-Repair Matters
Every day, the lining of your intestine faces harsh conditions: stomach acid, food particles, and trillions of microbes. Yet this tissue not only survives, it constantly repairs itself. When that repair system fails—after radiation therapy, in inflammatory bowel disease, or during severe infections—people can suffer pain, bleeding, and life‑threatening complications. This study uncovers a hidden “switch” inside intestinal cells that helps them decide when to stop resting and start rebuilding, offering clues to new treatments that could boost the gut’s natural repair crew.
A Delicate Architecture Under Constant Stress
The inner surface of the intestine looks like a shag carpet under the microscope, with finger‑like villi that absorb nutrients and pockets called crypts where stem cells live. These stem cells, and their rapidly dividing daughters, must strike a balance between steady renewal and emergency repair. The authors focused on how chemical tags on DNA-packaging proteins, called histones, influence this balance. These tags do not change genes themselves, but act more like dimmer switches, turning clusters of genes up or down as the intestine moves from normal life to injury and back to health.
An Epigenetic Mark That Tracks Damage and Healing
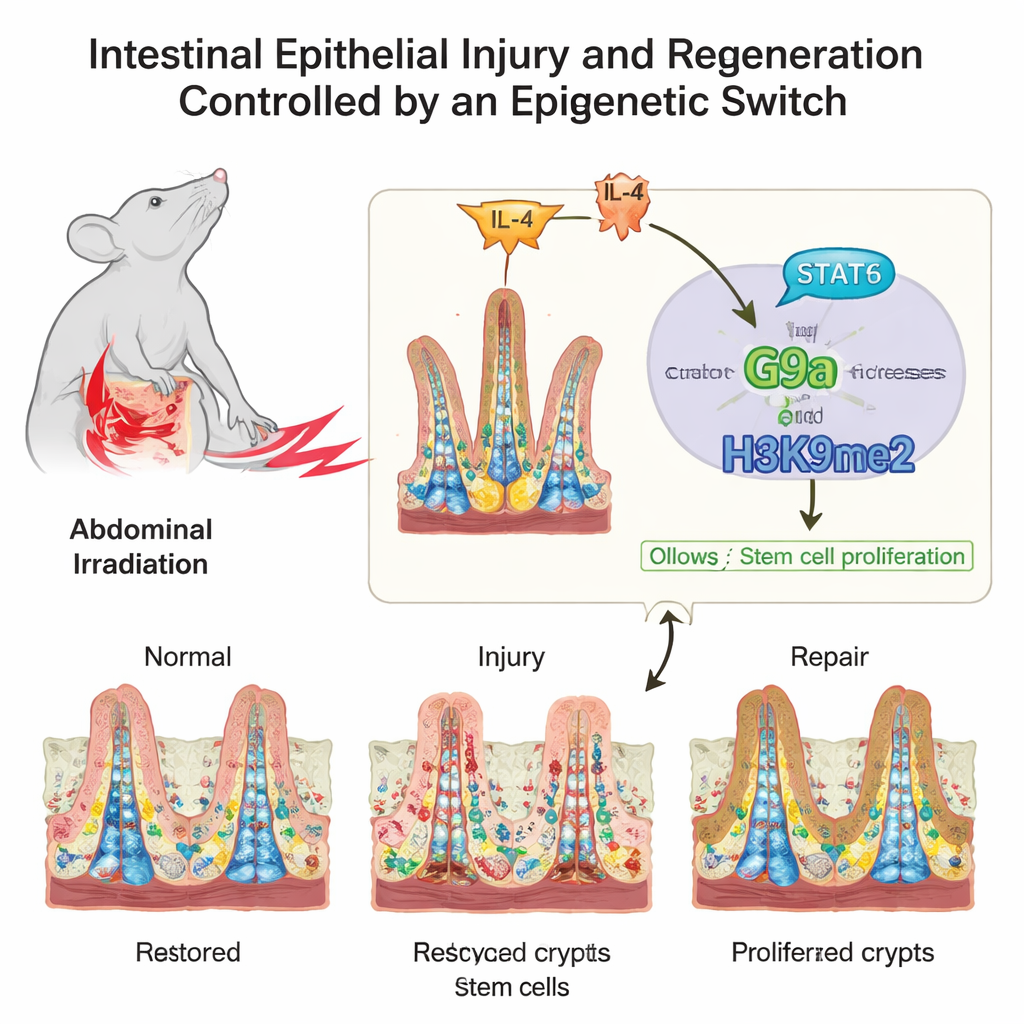
Using mice exposed to radiation—a common cause of intestinal injury during cancer treatment—the researchers scanned many different histone marks in the stem-cell–rich crypts. One mark, known as H3K9me2, rose sharply as the intestine shifted from injury into repair. Its writer enzyme, a protein called G9a, followed the same pattern. The team then examined human samples from patients who had received pelvic radiotherapy, as well as people with Crohn’s disease. In both settings, higher levels of H3K9me2 and G9a in intestinal cells went hand-in-hand with better signs of tissue repair, suggesting this system is conserved between mice and humans.
When the Repair Switch Is Broken
To test whether this mark is merely a bystander or truly drives healing, the scientists removed G9a specifically from the intestinal lining of mice, or blocked its activity with a drug. In both cases, the levels of H3K9me2 fell. After radiation or chemical injury, these mice lost more weight, had shorter, more damaged intestines, and showed fewer regenerating crypts and stem cells than normal animals. Even without any deliberate injury, the gut lining of G9a‑deficient mice looked stunted: crypts were shallower, villi were shorter, and the numbers of stem cells and their specialized descendants all dropped. Tiny “mini‑intestines” grown from these cells in a dish also grew poorly, underscoring that this molecular system is central to the intestine’s day‑to‑day renewal.
Releasing the Brakes on Cell Division
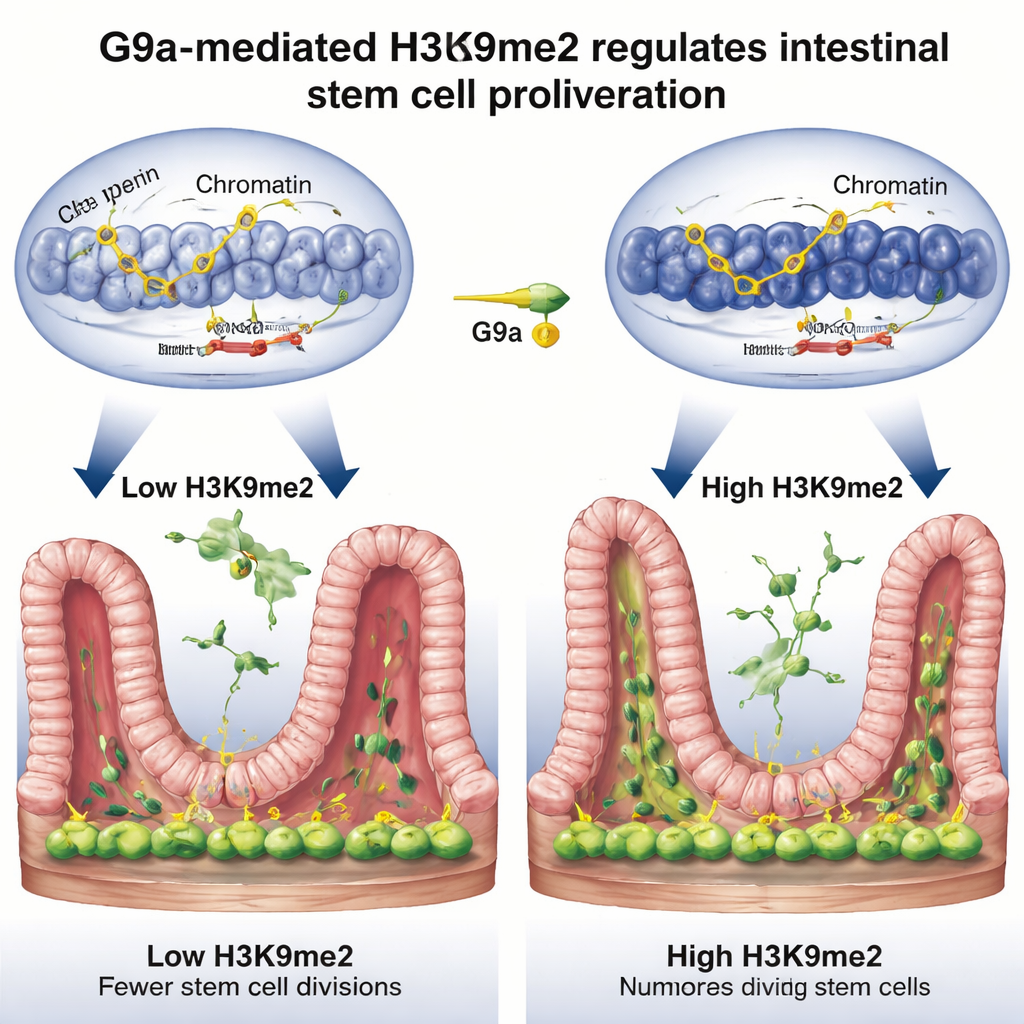
Digging deeper, the team asked which genes are controlled by this repair switch. By combining three powerful genome‑wide methods, they found that H3K9me2, placed by G9a, tends to sit on stretches of DNA that normally act as brakes on the cell cycle—genes like Rb1cc1, Rb1, Cdkn1a, and Pten, which slow or stop cell division. When G9a was missing, the chemical mark vanished from these regions, the surrounding DNA opened up, and these brake genes switched on more strongly, leading to sluggish stem-cell proliferation. During normal repair after injury, H3K9me2 levels at these genes rose, their activity dropped, and stem cells were freer to divide and rebuild the lining. In essence, G9a and H3K9me2 temporarily quiet the “stop” signals so the tissue can regrow.
A Signaling Chain From Immune Cues to Repair
The intestine does not act alone; it continuously listens to signals from the immune system. The researchers traced one such signal upstream of G9a. After injury, levels of the immune messenger IL‑4 increased, triggering activation of a protein called STAT6 inside intestinal cells. Activated STAT6 bound directly to the control region of the G9a gene, boosting its production. This created a chain of command: IL‑4 activates STAT6, STAT6 turns up G9a, and G9a adds H3K9me2 to silence cell‑cycle brake genes, allowing stem cells to expand and restore the damaged lining.
What This Means for Future Treatments
To a non‑specialist, these findings show that gut repair is guided not just by genes, but by reversible chemical marks that fine‑tune when those genes are used. The IL‑4–STAT6–G9a–H3K9me2 pathway acts like an internal control panel that loosens the brakes on stem-cell division just long enough to heal, then can be reset. In the future, therapies that gently enhance this pathway—such as drugs or gene‑based tools that raise G9a levels at the right time and place—might help patients recover faster from radiation injury, flare‑ups of inflammatory bowel disease, or other conditions that damage the intestinal lining, while minimizing the risks of uncontrolled cell growth.
Citation: Chen, J., Shi, X., Zhou, X. et al. G9a-mediated H3K9me2 orchestrates intestinal epithelial regeneration through epigenetic silencing of cell cycle-related genes. Nat Commun 17, 1874 (2026). https://doi.org/10.1038/s41467-026-68626-7
Keywords: intestinal regeneration, epigenetics, stem cells, histone methylation, inflammatory bowel disease