Clear Sky Science · en
WDR5 remodels NANOG condensates to drive transcriptional programs and sustain stem cell identity
Why This Matters for Stem Cells and Cancer
Stem cells have the remarkable ability to become many different cell types, a property that powers early development, tissue repair, and, unfortunately, some cancers. This study uncovers how two key proteins, NANOG and WDR5, cooperate to keep stem cells in this flexible state. By showing that WDR5 can physically reshape how NANOG clusters on DNA, the work links the physics of protein droplets inside the nucleus to the control of genes in both healthy stem cells and leukemia.
Proteins That Decide a Cell’s Future
Embryonic stem cells stay pluripotent—that is, able to become almost any cell—thanks to master regulators such as NANOG, OCT4, and SOX2. These proteins turn whole networks of genes on and off. NANOG is especially central: it not only binds DNA but also recruits large enzyme complexes that add activating chemical tags to histones, the proteins that package DNA. WDR5 is another crucial player. It helps histone-modifying machines place marks that signal active genes. Although both NANOG and WDR5 are known to support stem cell identity, how they talk to each other at the molecular level, and whether that affects the way they form tiny droplets—or “condensates”—in the nucleus, has been unclear.
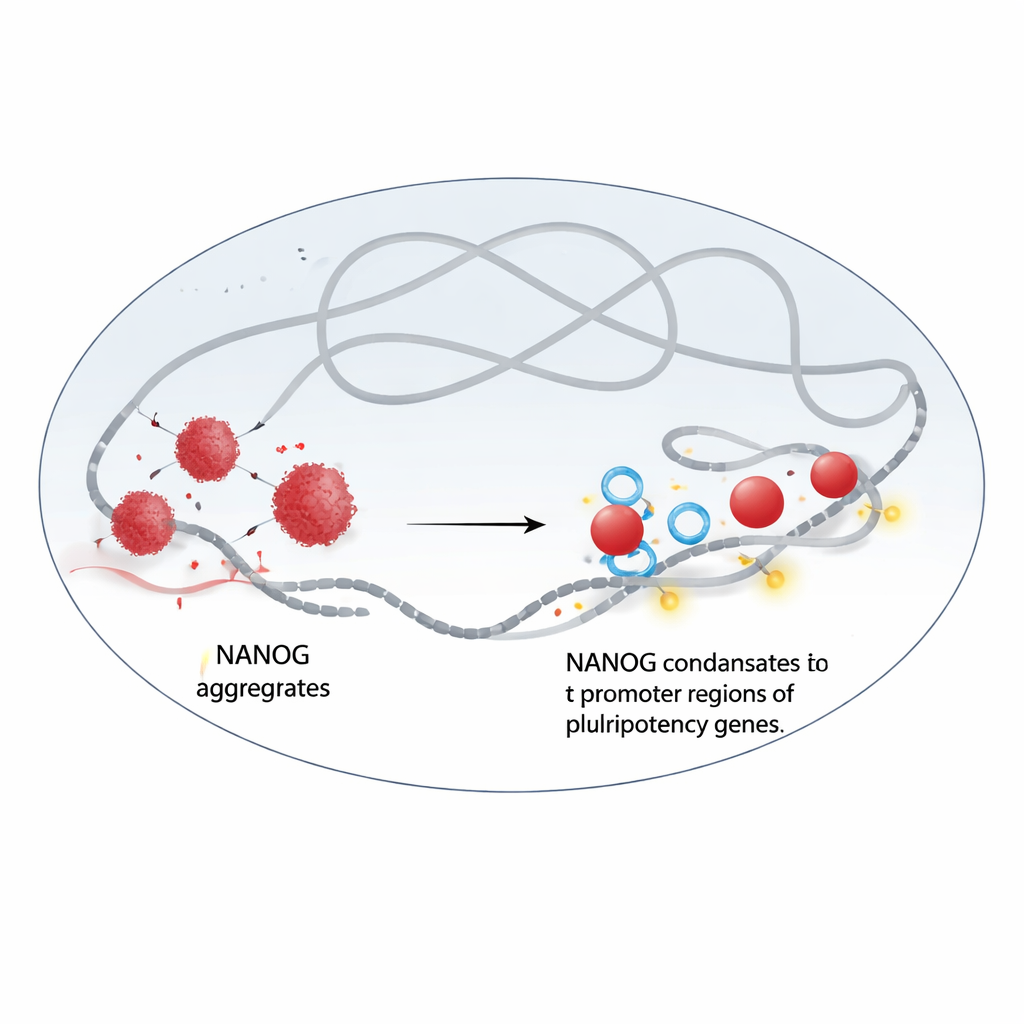
From Clumps to Liquid Droplets
The authors discovered that NANOG and WDR5 interact directly. When NANOG is purified by itself, it tends to clump into irregular, solid-like aggregates. Using electron microscopy and fluorescence experiments, the team showed that adding WDR5 reorganizes these clumps into round, liquid-like droplets that behave as dynamic condensates. In live cells, NANOG and WDR5 gather together in bright nuclear puncta that rapidly dissolve when treated with a chemical known to disrupt liquid–liquid phase separation. When this happens, both proteins lose much of their grip on chromatin, the DNA–protein complex that stores our genetic information, and their presence at key pluripotency genes drops sharply.
A Special Molecular Handshake
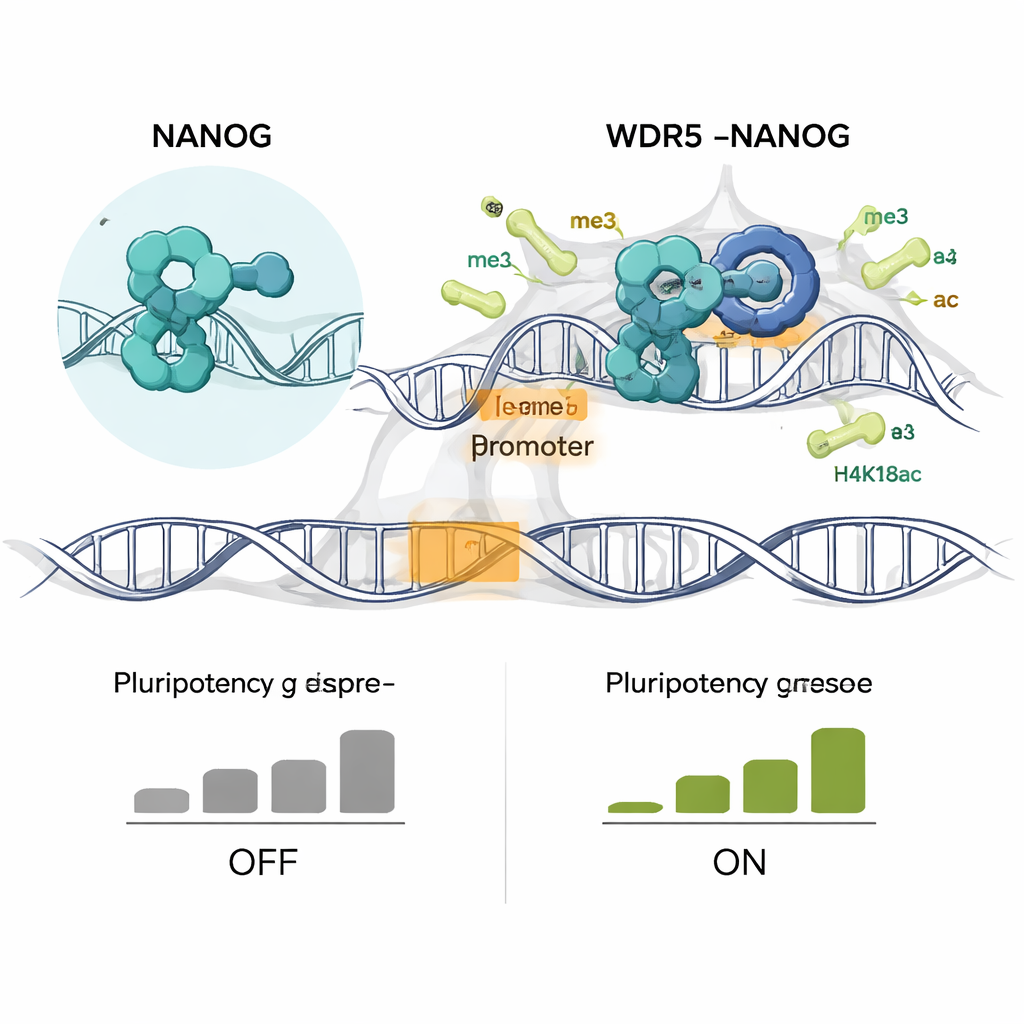
To understand the partnership in atomic detail, the researchers solved the crystal structure of NANOG’s DNA-binding region bound to WDR5. Unlike many other WDR5 partners, which use short flexible loops, NANOG engages WDR5 through an extended surface that includes both an unstructured arm and nearby helices. A single arginine amino acid in NANOG, at position 153, inserts deep into a central channel in WDR5, forming a key part of the “lock-and-key” interaction. Changing this residue to alanine (the R153A mutation) largely abolishes binding. Importantly, the mutant NANOG can still bind DNA, but WDR5 can no longer efficiently remodel its aggregates into fluid droplets, indicating that this specific contact is essential for forming functional condensates.
Keeping Stem Cells Young—and What Happens When It Fails
When the authors engineered mouse embryonic stem cells so that they expressed only the NANOG R153A mutant, the consequences were dramatic. The mutant cells lost the tight, dome-shaped colonies typical of pluripotent cells and flattened out, a visual sign of differentiation. They formed fewer alkaline-phosphatase–positive colonies, another hallmark of stemness, and turned down many core pluripotency genes such as Nanog itself, Sox2, Esrrb, and Klf4. At the same time, genes associated with specific lineages, including bone and developmental regulators, were switched on. Genome-wide profiling revealed that NANOG and WDR5 no longer co-occupied thousands of promoters, and two key activating histone marks, H3K4me3 and H4K16ac, were specifically lost at these shared sites. Early during differentiation, these chemical tags and newly made RNA transcripts declined before bulk mRNA levels dropped, pointing to a primary failure in active chromatin maintenance.
Turning a Fundamental Mechanism into a Cancer Strategy
Because similar gene networks drive some cancer stem cells, the team tested whether disrupting the NANOG–WDR5 axis could curb leukemia stem cells in a mouse model of acute myeloid leukemia. A small-molecule drug, C16, which binds the same WDR5 pocket used by NANOG, selectively weakened the WDR5–NANOG interaction. In leukemia cells, C16 sharply reduced colony formation, depleted the stem-like population, and pushed cells toward mature myeloid fates, while sparing normal blood stem cells far more than an existing leukemia drug that targets a different WDR5 partner. RNA sequencing showed that C16 shut down gene programs for stemness and self-renewal and boosted differentiation pathways. In mice, C16 treatment reduced leukemia stem and progenitor populations, underscoring the therapeutic promise of targeting this protein partnership.
Big Picture: How Droplets Help Decide Cell Identity
This work reveals that WDR5 does more than sit on chromatin and recruit enzymes: it can physically remodel NANOG from inert aggregates into flexible liquid condensates that sit on pluripotency gene promoters and attract activating histone marks. When this remodeling is blocked—either by a precise mutation in NANOG or by a small molecule that disrupts their contact—stem cells lose their identity and cancer stem cells lose their grip on self-renewal. For a lay reader, the key message is that tiny shifts in how proteins assemble into droplets on DNA can have enormous consequences for whether a cell stays young and flexible, matures into a specific fate, or fuels disease.
Citation: Wang, D., Shi, X., Xie, J. et al. WDR5 remodels NANOG condensates to drive transcriptional programs and sustain stem cell identity. Nat Commun 17, 1907 (2026). https://doi.org/10.1038/s41467-026-68623-w
Keywords: stem cell pluripotency, NANOG, WDR5, phase separation, leukemia stem cells