Clear Sky Science · en
Dual-functional quorum sensing signal synthases DspII and DspI coordinate virulence switch in Pseudomonas aeruginosa
How a Hospital Superbug Changes Its Attack Strategy
Pseudomonas aeruginosa is a notorious hospital germ that can cause stubborn lung and wound infections, especially in vulnerable patients. It survives by switching between two lifestyles: a protected, slow-growing community called a biofilm and a fast-moving, aggressive form that invades tissues. This study uncovers how the bacterium itself decides when to break out of a long-term biofilm and launch a sudden acute attack, revealing potential new weak points for future treatments.
A Microbial Life Between Hiding and Attacking
Many bacterial infections unfold in stages. Early on, free-swimming cells spread through the body and deploy powerful weapons that damage host tissues. Later, they often settle down into biofilms—thick, slimy layers stuck to surfaces such as catheters, lung tissue, or wounds. In these communities, cells are wrapped in a protective matrix that makes them hard to kill with antibiotics or immune defenses. Yet biofilms are not a dead end. Cells can leave, regain motility, and spark new rounds of acute infection elsewhere. Until now, it has been unclear how Pseudomonas aeruginosa actively coordinates this shift from chronic persistence back to aggressive disease.
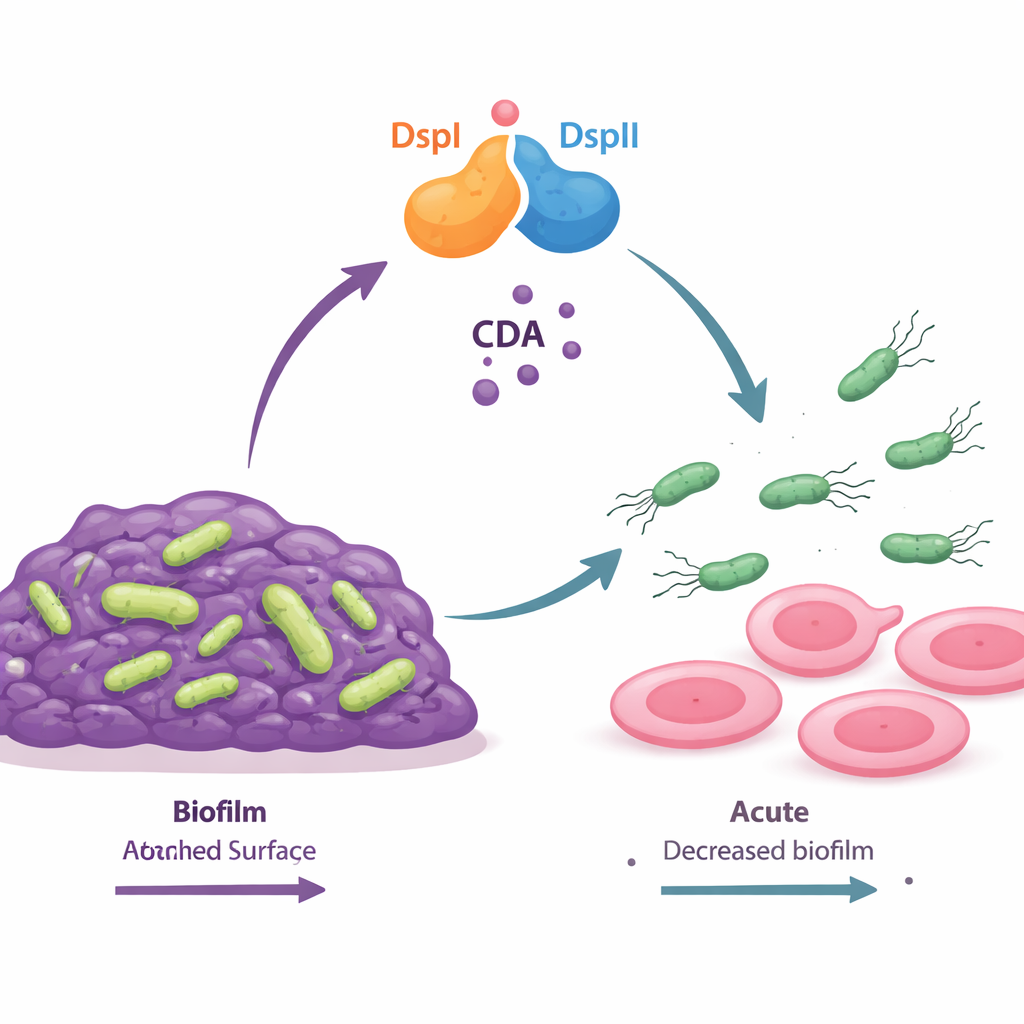
A Two-Protein Switch That Makes a Chemical "Go" Signal
The authors discovered that two bacterial proteins, named DspI and DspII, work together as a kind of molecular switch. Both are enzymes of the same general type and sit next to each other in the bacterial genome, turning on together as the population grows denser. When they form a pair, they manufacture a small fatty acid signal called cis-2-decenoic acid (CDA), part of a broader group of communication molecules known as DSF signals. Mutant bacteria lacking either DspI or DspII could no longer make CDA, formed unusually thick biofilms, and lost their ability to swarm across surfaces. Only when both proteins were restored together did the cells recover normal signal production, biofilm breakup, and motility, showing that the two enzymes function as an interdependent team.
From Chemical Signal to Motion and Escape
CDA does not act alone; it plugs into the bacterium’s internal wiring. The team showed that CDA lowers levels of another messenger molecule, cyclic di-GMP, by boosting the activity of a specific enzyme called RbdA that breaks this messenger down. High cyclic di-GMP normally encourages biofilm building by stimulating sugary matrix production and suppressing flagella, the tiny propellers that drive swimming. When CDA is present, cyclic di-GMP levels fall, a regulator named FleQ shifts gears, sticky sugar production drops, and flagella are extended and strengthened. As a result, biofilms loosen and cells regain the ability to move away, seeding new territories.
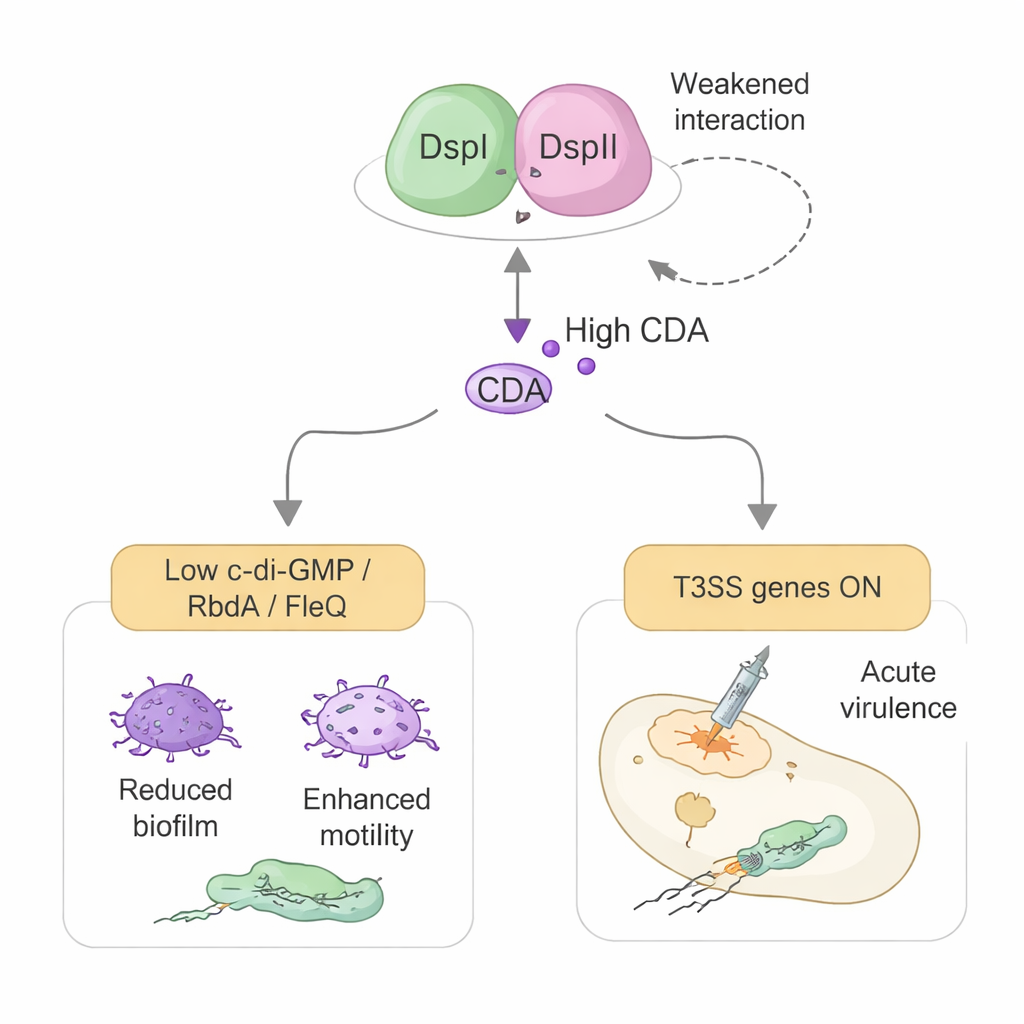
Rewiring the Bacterial Weapons System
The study also reveals a second, unexpected role for DspI and DspII that goes beyond chemistry. The same protein pair helps turn on the bacterium’s type III secretion system (T3SS), a syringe-like apparatus used to inject toxins directly into host cells. Rather than acting through CDA, this effect works through direct control of genetic switches. DspI and DspII dampen the production of two small regulatory RNAs, RsmY and RsmZ, which normally shut down T3SS protein manufacture. They do this by binding to the DNA control region of one RNA and physically interacting with a master regulator protein called GacA. When RsmY and RsmZ are held in check, a downstream activator named RsmA is freed, and the genes for the T3SS and its master controller ExsA are turned on, heightening cytotoxicity in cell culture and killing in an insect infection model.
A Self-Tuning System with Drug Targets
Cleverly, the strength of the DspI–DspII partnership and its control over virulence is itself tuned by CDA levels. At low concentrations—like those expected in cells just leaving a biofilm—CDA tightens the interaction of the two proteins and promotes more signal production and T3SS activation. At high concentrations inside dense biofilms, CDA weakens their interaction and reduces their grip on DNA, limiting further signal synthesis and expensive weapon production in cells that are staying put. This dual role makes the DspI–DspII complex a central hub that links population size, biofilm escape, and acute virulence. Because disrupting the complex can block both signal production and toxin system activation, it offers an attractive target for future drugs that could keep Pseudomonas locked in a less harmful, chronic state and prevent dangerous flare-ups.
Citation: Huang, J., Zhou, T., Zhou, X. et al. Dual-functional quorum sensing signal synthases DspII and DspI coordinate virulence switch in Pseudomonas aeruginosa. Nat Commun 17, 1926 (2026). https://doi.org/10.1038/s41467-026-68622-x
Keywords: Pseudomonas aeruginosa, biofilm dispersion, quorum sensing, virulence switch, cis-2-decenoic acid