Clear Sky Science · en
Dynamic signature of activity-stability tradeoff in lactamase evolution
Why this matters for antibiotic resistance
Antibiotic resistance often seems like a black box: bacteria "somehow" change and drugs stop working. This study opens that box for one of the world’s best‑known resistance enzymes, TEM‑1 β‑lactamase, which helps bacteria destroy penicillin‑like antibiotics. By watching how this enzyme’s shape and motions change as it evolves to hit newer drugs, the authors reveal why mutations that boost activity often weaken stability, and how evolution finds clever ways to balance both.
From penicillin fighter to broader drug breaker
TEM‑1 originally excels at breaking down older penicillin antibiotics but is poor at tackling newer, bulkier drugs like cefotaxime. In many clinical strains, a single key mutation called G238S appears near the enzyme’s active site, the pocket where antibiotics are cut. This mutation dramatically improves the enzyme’s ability to destroy cefotaxime, while only modestly harming its original penicillin‑cleaving job. The authors show that G238S does not simply widen the pocket; it reorganizes how several surrounding loops and helices move, creating a new functional conformation that better accommodates bulky drugs.
Protein motion as an evolutionary tuning knob
Using advanced nuclear magnetic resonance (NMR) techniques, the researchers measured how different parts of TEM‑1 move over timescales ranging from trillionths to thousandths of a second. Wild‑type TEM‑1 is quite rigid, which helps it efficiently process its original substrates. G238S leaves most of this fast rigidity intact but introduces slower, carefully tuned motions in many walls of the active site. These motions are fast enough to keep up with the enzyme’s chemistry yet not so wild that they disrupt critical catalytic residues. The result is an “optimized window” of flexibility: just enough movement to open the pocket for cefotaxime, while keeping the core chemistry machinery properly aligned.
Balancing two shapes instead of choosing one
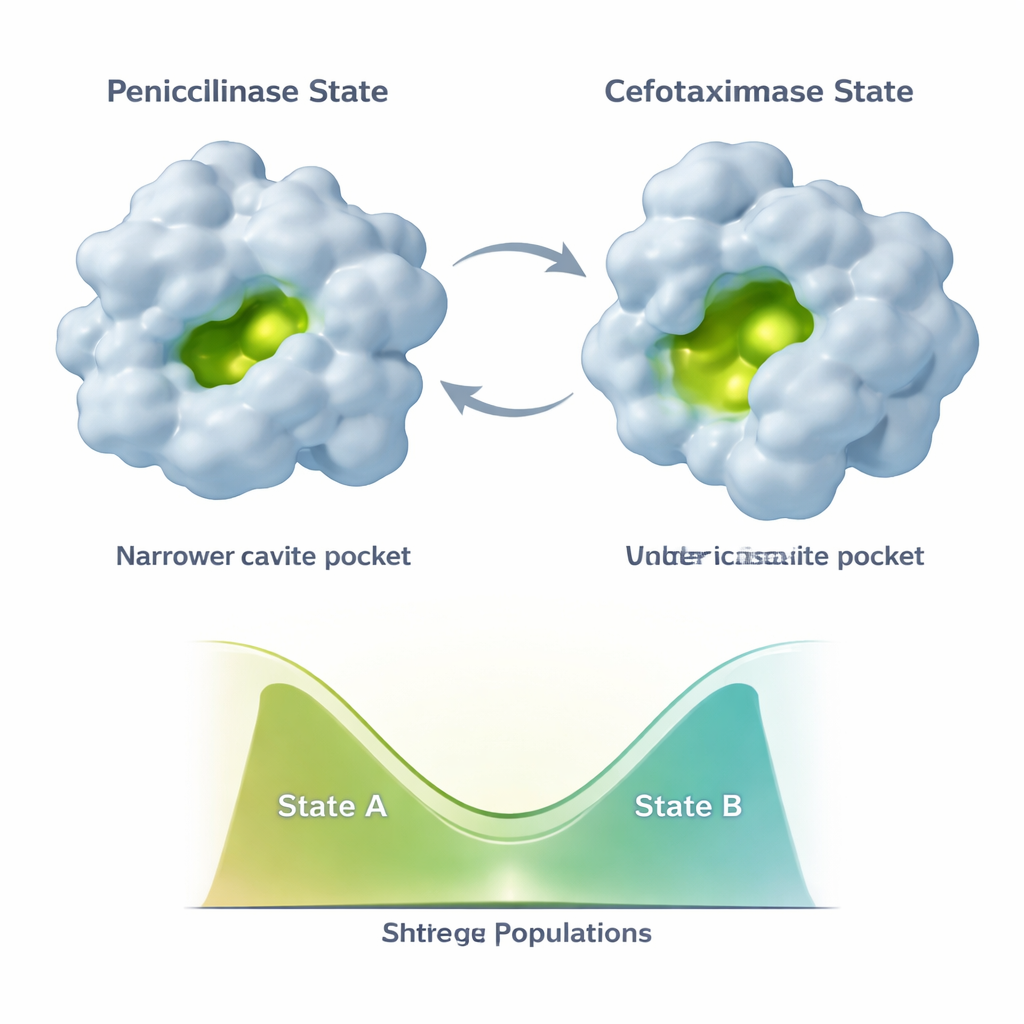
Evolution does not freeze TEM‑1 into a single new shape. Instead, the enzyme samples at least two main conformations: a “penicillinase” state resembling the original structure and a more open “cefotaximase” state better suited for newer drugs. Additional mutations that appear later, such as E104K and A42G, do something subtle. Rather than simply making the new, cefotaximase‑friendly shape more stable, they rebalance the mixture of the two states. NMR data show that different parts of the active site and the supporting scaffold can shift their populations independently along this two‑state continuum. This leads to a combinatorial set of enzyme variants, each with a different blend of penicillin‑like and cefotaxime‑like conformations, and therefore different catalytic profiles.
Hidden weak spots and long‑distance repairs
Mutations that improve activity often carry a hidden cost: they make the protein less stable. Instead of looking only at complete unfolding, the team mapped local stability at the level of short segments using hydrogen–deuterium exchange with mass spectrometry. G238S was found to destabilize not only nearby loops but also distant helices and sheet regions that form a structural backbone. Some of these regions overlap with a “cryptic” allosteric pocket—an infrequently visited opening in the protein’s core that can bind small molecules and dampen activity. G238S makes this pocket easier to open, effectively building a mild self‑inhibitory feature into the enzyme. Later mutations, especially A42G, reinforce this weakened backbone network, improving local stability around three interacting helices without removing the beneficial active‑site dynamics. In other words, evolution patches distant structural weak spots instead of undoing the original innovation.
What the study reveals about evolution’s strategy
To a lay reader, the central message is that proteins like TEM‑1 do not evolve resistance by flipping a simple on–off switch. Each mutation slightly reshapes how the enzyme breathes, flexes, and divides its time between different working poses. G238S opens the door to a new job—breaking newer antibiotics—but also creates local fragilities and a partially self‑inhibiting state. Secondary mutations act like careful reinforcements, stabilizing the scaffold and fine‑tuning the balance between old and new conformations so the enzyme can remain both active and durable. This dynamic view of evolution, where motions and local weaknesses matter as much as static structure, can help guide the design of future antibiotics and enzyme‑targeting drugs that are harder for bacteria to outsmart.
Citation: Arcia, E., Keramisanou, D., Jacobs, L.M.C. et al. Dynamic signature of activity-stability tradeoff in lactamase evolution. Nat Commun 17, 1884 (2026). https://doi.org/10.1038/s41467-026-68620-z
Keywords: antibiotic resistance, beta-lactamase, protein evolution, enzyme dynamics, protein stability