Clear Sky Science · en
A fully degradable triboelectric vagus nerve stimulator for attenuating cardiac remodeling and heart failure at different stages
Why a dissolving heart device matters
Heart failure is a common and deadly condition, affecting tens of millions of people and often leading to years of fatigue, breathlessness, and repeated hospital stays. Doctors know that gently stimulating a major nerve called the vagus nerve can help the failing heart, but current implants are bulky, battery-powered, and must be surgically removed or replaced. This study introduces a tiny, flexible stimulator that powers itself from body motion and then safely dissolves inside the body after treatment, opening the door to short-term, personalized therapies without extra surgeries.
A gentler way to tap into the body’s wiring
The vagus nerve runs from the brain to many organs and helps balance the body’s stress and relaxation systems. In heart failure, this balance tips toward constant “fight-or-flight” activation, which worsens damage to the heart. Low-level vagus nerve stimulation (LL-VNS) delivers signals that are too weak to slow the heart or disturb its rhythm, but strong enough to restore some of this balance and reduce harmful inflammation. Earlier clinical trials using standard, battery-powered stimulators showed that LL-VNS could improve symptoms and quality of life, yet the hardware itself posed problems: limited battery life, the need for replacement operations, and the long-term presence of foreign material in the body.
A self-powered device that disappears
To solve these issues, the researchers built a fully biodegradable triboelectric nanogenerator, or BTENG, that converts gentle mechanical motion into electricity. The device is made from layers of common biodegradable polymers and thin metal films, sealed within a protective coating. As the layers touch and separate—driven by small movements—electrical charges build up and are directed to a cuff that wraps around the vagus nerve. In laboratory tests, the BTENG reliably produced voltages of about 9 volts and currents around 4 microamps, enough to match the safe stimulation level established in mice. The output remained stable over thousands of cycles and for at least four weeks inside a fluid-filled environment similar to the body. After treatment, a burst of medical ultrasound can crack the outer coating and speed up the device’s breakdown, turning it into harmless fragments that the body gradually clears. 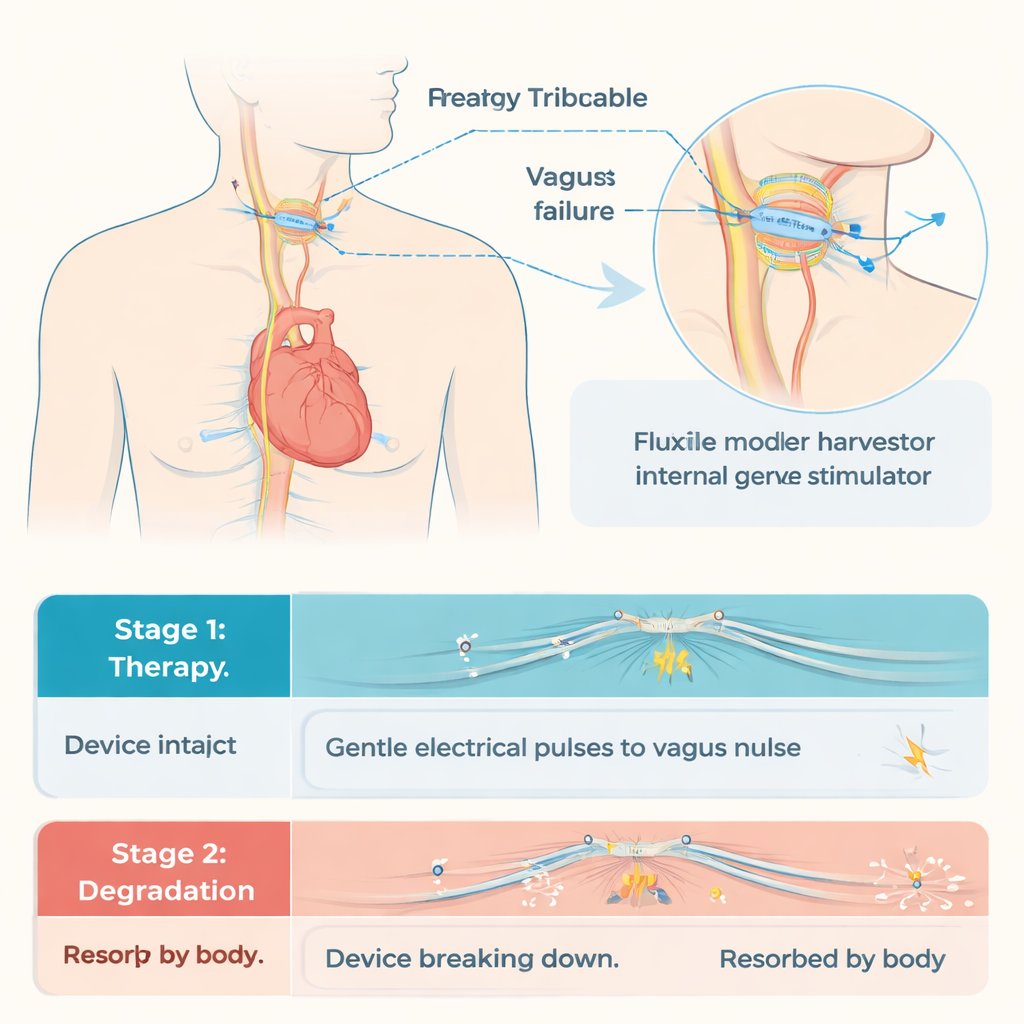
Protecting and repairing the failing heart
The team tested the system in a mouse model of heart failure caused by long-term pressure overload on the heart, similar to what happens in severe high blood pressure or narrowed heart valves. They started LL-VNS at three different stages: very early (before major damage), after early thickening of the heart muscle, and after full heart failure had developed. Across all three scenarios, mice that received daily low-level stimulation through the BTENG had better pumping function, less enlargement of the heart, and less scar tissue than untreated animals or those that received surgery and implants without stimulation. Measures such as ejection fraction, lung congestion, and the ratio of heart weight to body size all improved, showing that the therapy could not only slow damage but also partially reverse it, even when started late.
How nerve signals reshape heart biology
To understand how a small electrical signal could have such wide effects, the researchers examined heart tissue at the genetic level. They found that LL-VNS shifted the activity of hundreds of genes involved in the heart’s supporting scaffold (the extracellular matrix), muscle structure, cell death, growth, and inflammation. These changes lined up with the observed drop in tissue scarring and improvement in muscle health. Because the vagus nerve communicates using the chemical messenger acetylcholine, the team tested whether this molecule was essential by blocking its main receptors with the drug atropine. When atropine was given, the benefits of LL-VNS disappeared: heart function worsened, scarring increased, and disease-related genes stayed elevated. This showed that acetylcholine release triggered by vagus nerve signals is a key driver of the heart-protective effects. 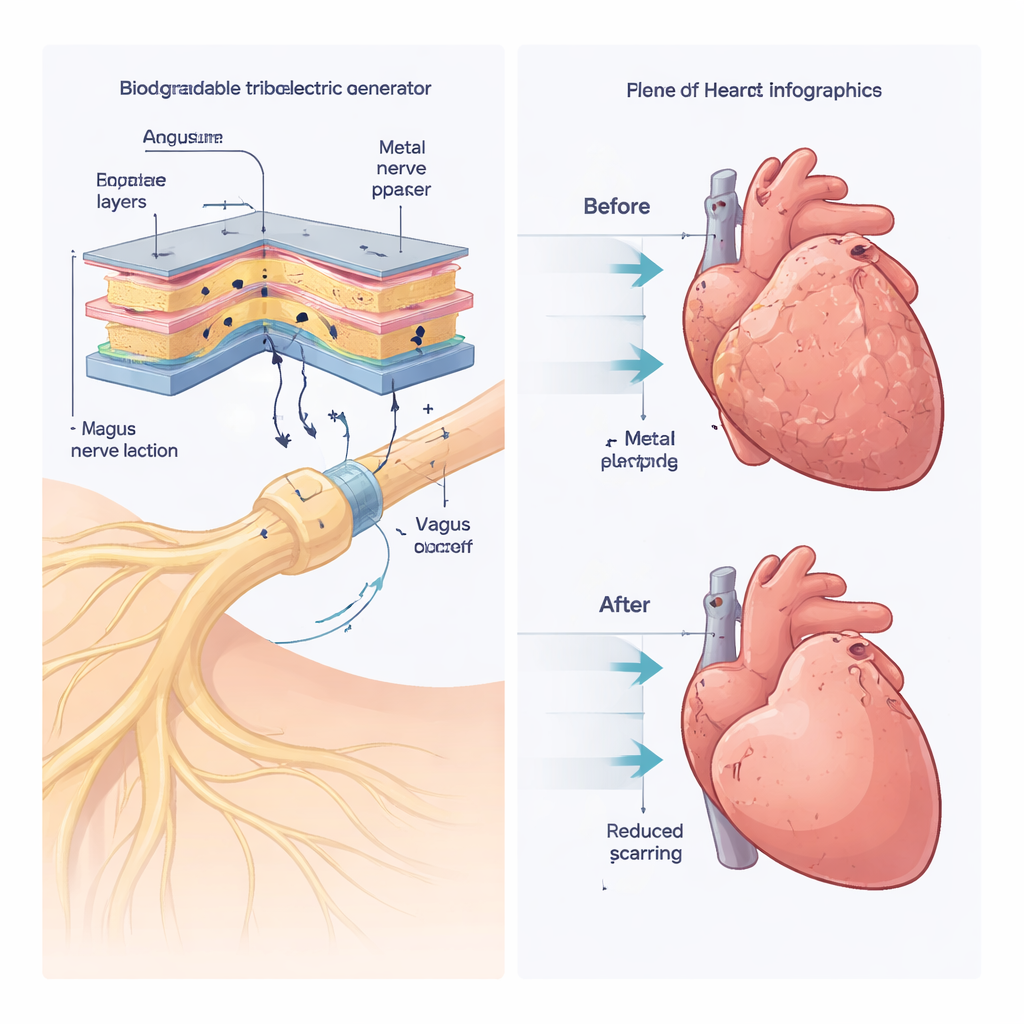
From dissolving implants to personalized heart care
Taken together, the findings demonstrate that a small, self-powered, fully degradable stimulator can safely deliver nerve signals that prevent or reverse harmful heart remodeling in mice. For patients, a future version of this technology could be implanted for only as long as needed, powered by their own body movements, and then erased from the body using a simple ultrasound session—no battery changes or removal surgery required. By combining precise nerve stimulation with smart materials that vanish on command, this approach points toward a new class of temporary, personalized heart failure treatments that may reduce complications, lower costs, and improve quality of life.
Citation: Guo, Z., Chao, SY., Kong, CY. et al. A fully degradable triboelectric vagus nerve stimulator for attenuating cardiac remodeling and heart failure at different stages. Nat Commun 17, 1893 (2026). https://doi.org/10.1038/s41467-026-68619-6
Keywords: heart failure, vagus nerve stimulation, biodegradable implant, triboelectric generator, cardiac remodeling