Clear Sky Science · en
A spatially resolved atlas of gastric cancer characterises a lymphocyte-aggregated region
Why the Neighborhood Around a Tumor Matters
Cancer is not just a clump of rogue cells; it lives in a busy neighborhood of immune and structural cells that can either help fight the tumor or let it grow unchecked. This study maps that neighborhood in gastric (stomach) cancer with unprecedented detail, showing how tiny immune hot spots and cold spots inside the same tumor may influence whether a patient responds to modern immunotherapies.
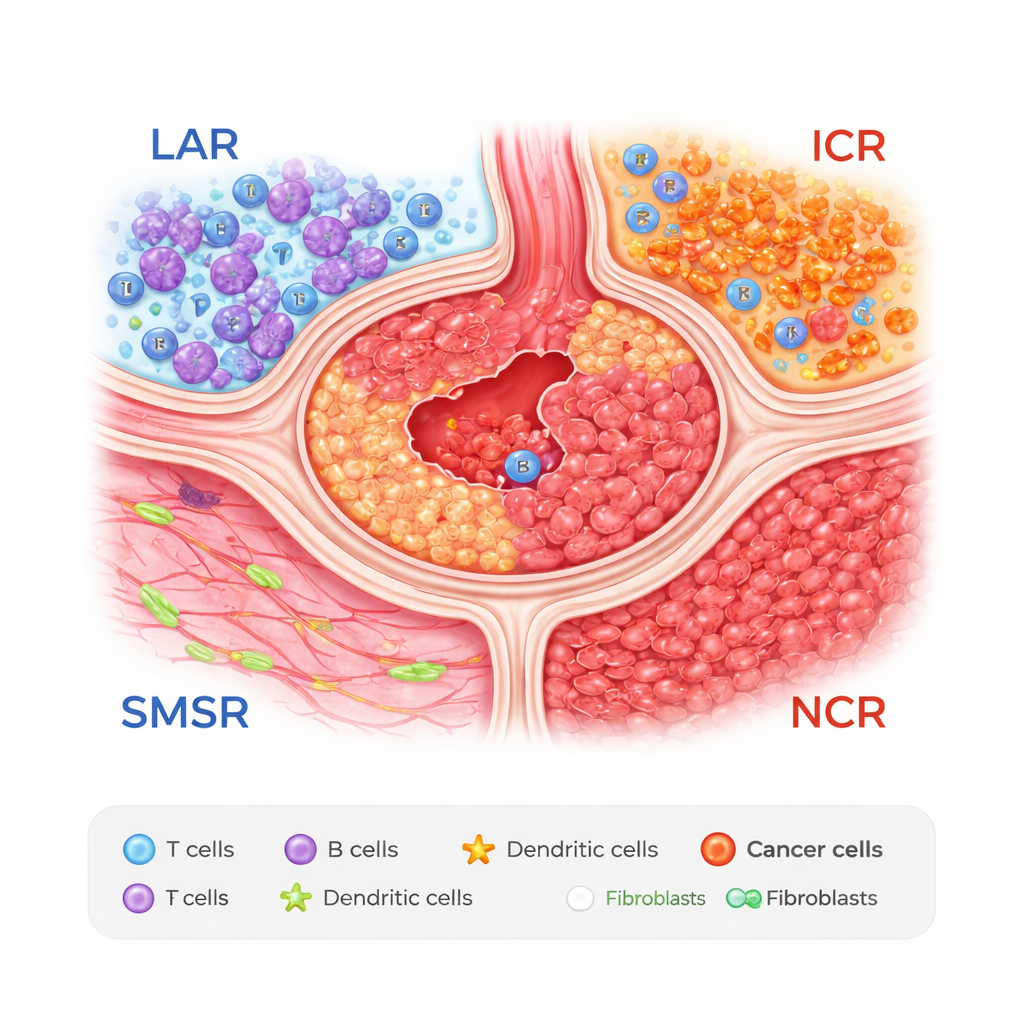
Four Zones Inside a Stomach Tumor
The researchers combined two powerful tools: single-cell RNA sequencing, which reads gene activity in individual cells, and spatial transcriptomics, which records where those cells sit in tissue slices. Studying samples from 27 patients, they discovered that stomach tumors can be divided into four recurring zones. One is a lymphocyte-aggregated region, or LAR, rich in clusters of T and B cells that resemble miniature lymph nodes. Another is a smooth muscle and stromal region (SMSR), dominated by muscle and connective tissue. The remaining two zones are cancer-dense areas: an immunogenic cancer region (ICR), where tumor cells mingle with some protective stomach cell types, and a negative-immunogenic cancer region (NCR), packed with malignant cells and relatively few defensive immune cells.
Immune Hot Spots Linked to Better Outcomes
The team found that the LAR zone is enriched for genes normally seen in lymph nodes, where immune cells are trained and activated. These include signals that help recruit lymphocytes and organize them into structures called tertiary lymphoid structures. When they looked at large public cancer datasets, tumors with higher expression of LAR-linked genes tended to have better patient survival and a somewhat higher likelihood of responding to immune checkpoint therapies that release the brakes on T cells. In contrast, gene signatures from muscle-rich or heavily cancerous regions were usually associated with poorer outcomes across several cancer types.
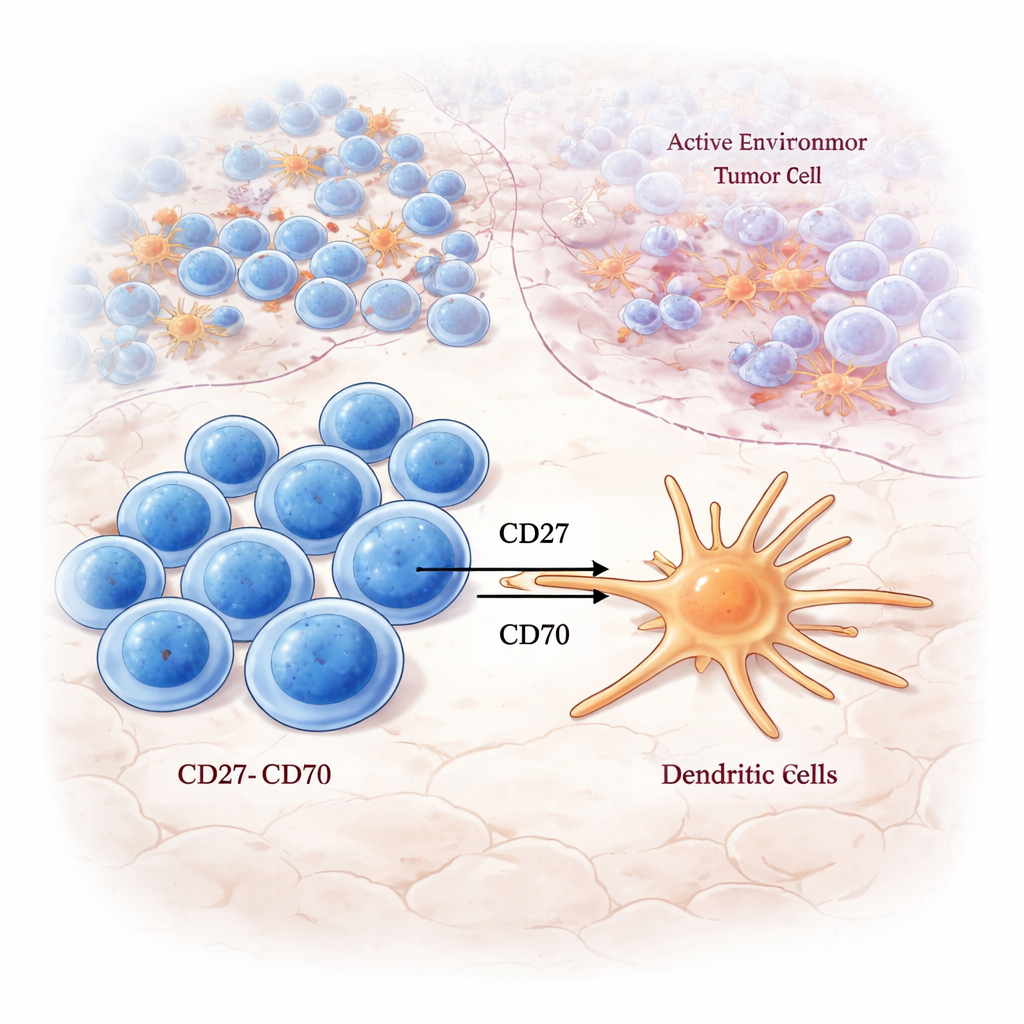
Where Naive T Cells Learn to Fight
Zooming in on the LAR, the authors saw strong activity in signaling pathways that are central to waking up T cells: T cell receptor signaling, costimulation pathways, and antigen processing. Naive CD8 T cells—cells that have not yet fully committed to attacking a specific target—were especially tied to these activation signals inside the LAR, but not elsewhere. Using multicolor staining, they detected CD8 T cells in the LAR that bore markers of both renewal and proliferation, suggesting that this region acts as an on-site training and expansion zone where fresh tumor-fighting cells are primed.
Two Flavors of Immune Neighborhood
Not all LARs were equal. When the researchers grouped patients by the cellular makeup of their LARs, they uncovered two patterns. In “Group A” tumors, LARs closely resembled classic lymph nodes, rich in specialized B cells and dendritic cells, and the cancer regions next to these LARs were filled with activated, battle-ready lymphocytes. In “Group B,” LARs were less developed, and the adjoining cancer tissue contained more resting, less engaged T cells. Advanced computational analysis of tissue images confirmed that activated immune cells physically clustered near these more mature LARs, suggesting that the local architecture can steer how vigorously the immune system attacks the tumor.
Checkpoint Signals in Space
The study also tracked where immune “switches” known as checkpoint molecules are expressed. Stimulatory pairs such as CD27–CD70 were concentrated in the LAR, particularly in Group A tumors, connecting exhausted but tumor-reactive CD8 T cells with dendritic cells that could further boost their activity. In contrast, many inhibitory checkpoint pairs, like TIGIT–NECTIN2 and LAG3–LGALS3, were enriched in the NCR, the most cancer-heavy zone. This suggests that even when T cell receptor signaling is present, the surrounding checkpoint landscape can tilt local immunity toward either attack or suppression, depending on the region of the tumor.
What This Means for Patients
To a non-specialist, the key message is that where immune cells and signals sit inside a tumor may matter as much as how many there are. This work shows that gastric cancers contain distinct immune neighborhoods, and that robust, lymph-node-like regions near the tumor are linked to more active, better-positioned T cells and more favorable outcomes. By learning to recognize and possibly strengthen these lymphocyte-aggregated regions, future therapies could be better tailored—choosing drugs, combinations, or even targeted delivery approaches that exploit the tumor’s own internal immune strongholds.
Citation: Gao, S., Qin, S., Wang, D. et al. A spatially resolved atlas of gastric cancer characterises a lymphocyte-aggregated region. Nat Commun 17, 2059 (2026). https://doi.org/10.1038/s41467-026-68612-z
Keywords: gastric cancer, tumor microenvironment, tertiary lymphoid structures, spatial transcriptomics, cancer immunotherapy