Clear Sky Science · en
Divergent aging of nulliparous and parous mammary glands reveals IL33+ hybrid epithelial cells
How Pregnancy Can Reshape Lifetime Breast Cancer Risk
Why does having a baby early in life lower a woman’s lifetime risk of breast cancer, while simply getting older raises it? This study uses mice to peer into the breast’s cellular “ecosystem” over time, comparing animals that never had a pregnancy with those that did. By following how individual cells change with age and reproduction, the researchers uncover a rare, shape‑shifting cell type that may help explain pregnancy’s long‑term protection against breast cancer.
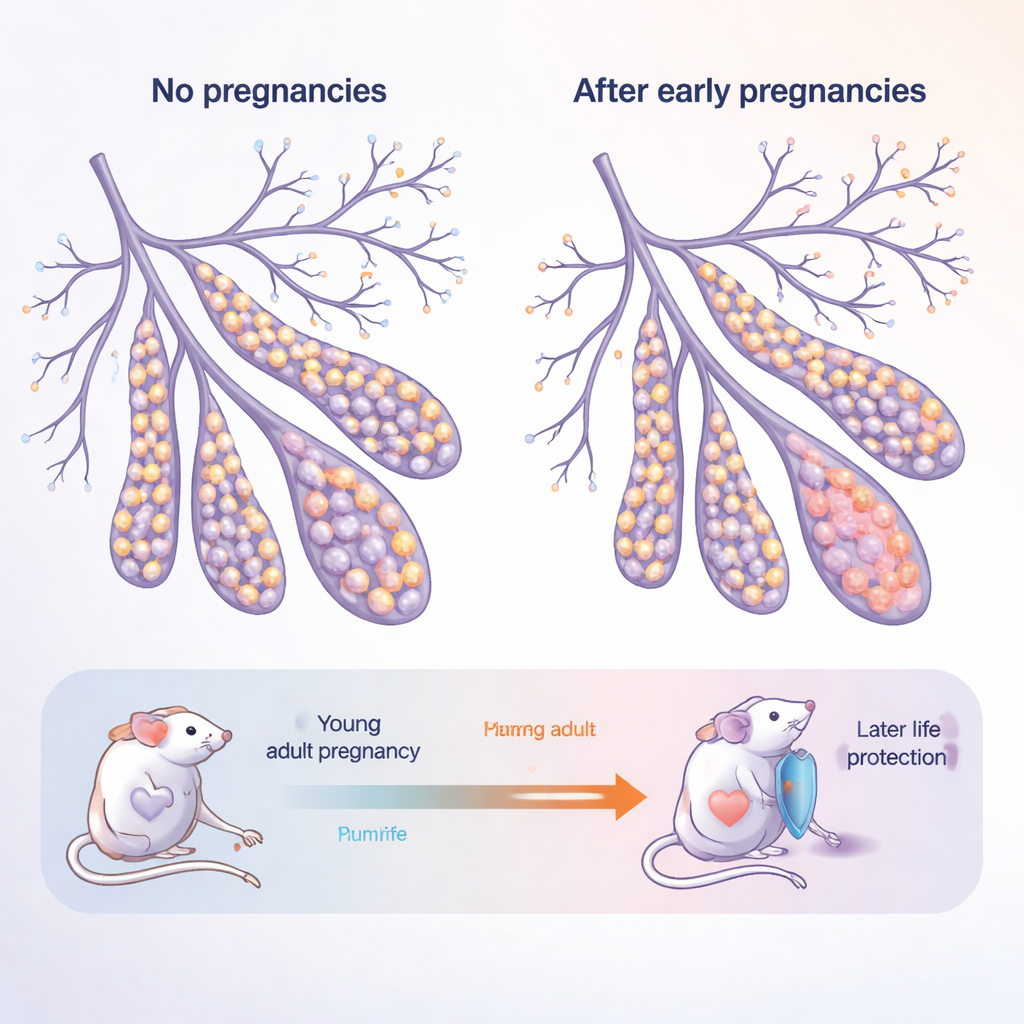
The Breast as a Living, Changing Tissue
The mammary gland is not a static organ: it grows and remodels dramatically at puberty, during pregnancy, and again when milk production ends. It is built from two main layers of epithelial cells that line the ducts: inner “luminal” cells that contact milk and hormones, and outer “basal” cells that provide structure and house stem‑like cells. The authors studied young adult mice and older, “post‑menopausal” mice, each either never pregnant (nulliparous) or having gone through multiple pregnancies (parous). This design mimics a woman who gives birth in her twenties and then ages into her fifties and beyond.
How Aging and Pregnancy Pull the Tissue in Different Directions
As mice aged without ever being pregnant, their mammary glands shifted toward a higher share of basal cells and fewer luminal cells. These older, never‑pregnant glands also produced many more organoids—miniature 3D structures grown from single cells—indicating that their cells were unusually eager to proliferate. In contrast, mice that had experienced pregnancies showed a “normalized” balance: the age‑related surge in basal cells was dampened, and their cells formed fewer organoids. This suggests that pregnancy leaves a lasting mark that both changes how cells choose their fates and reins in their regenerative, and potentially cancer‑prone, potential.
The Discovery of Hybrid, Shape‑Shifting Cells
To look more deeply, the team used single‑cell RNA sequencing to read out which genes were active in thousands of individual mammary cells. Among the expected luminal, basal, immune, and stromal cells, they found a rare group that did not fit cleanly into either main layer. These cells expressed markers of both luminal and basal lineages, earning the name “hybrid” epithelial cells. Strikingly, these hybrids accumulated with age in never‑pregnant mice but were far less common in equally old mice that had been pregnant. The hybrids strongly expressed a gene called Il33, which encodes the signaling molecule IL‑33, along with markers linked to early developmental stages and high plasticity—the ability to switch identity or behavior.
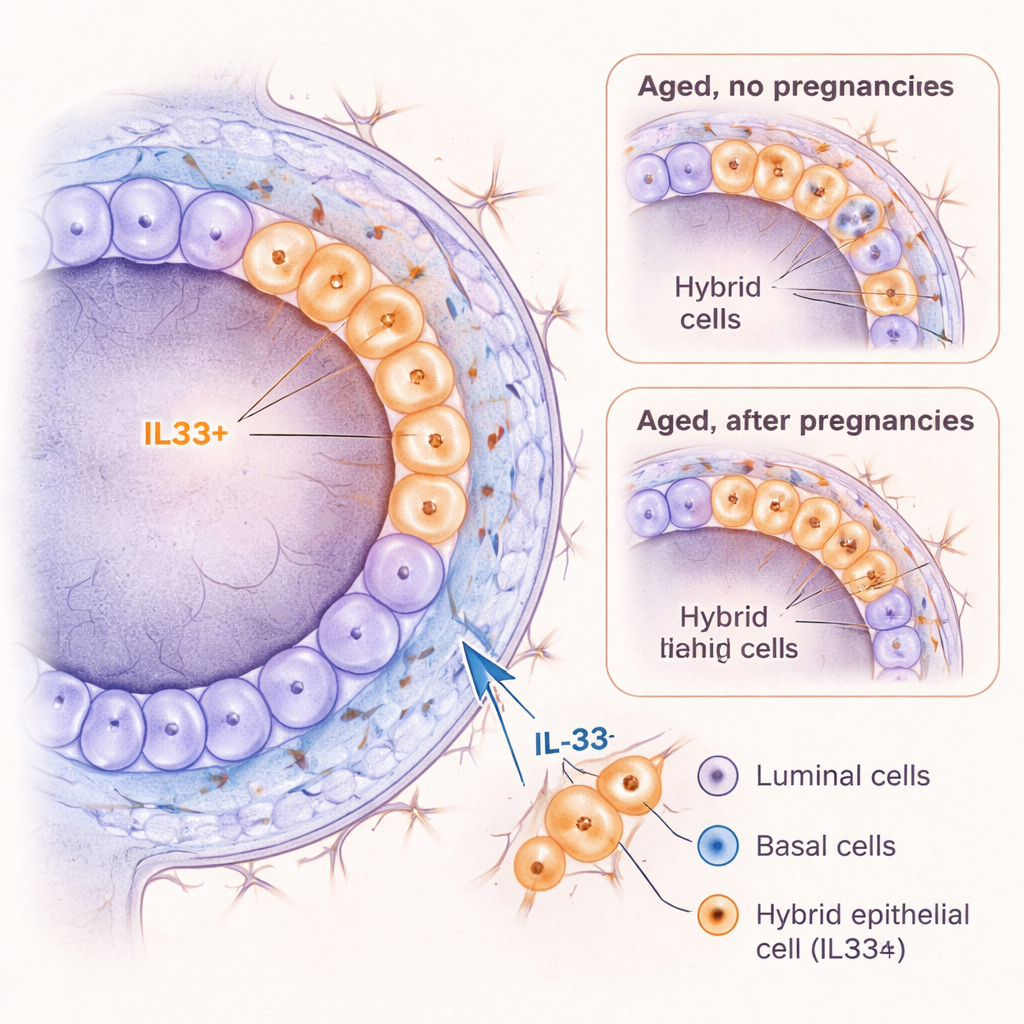
IL‑33 as a Driver of Risky Cell States
Because IL‑33 has been tied to inflammation and cancer in other organs, the researchers asked whether it can actively push mammary cells into a hybrid‑like state. When they treated young mouse mammary cells with IL‑33 in 3D culture, basal‑layer cells formed more organoids, held onto a more primitive identity, and produced more cells with the hybrid marker KRT6A. In live young mice, short‑term IL‑33 injections caused the ducts to widen and triggered a burst of cell division, along with a temporary spike in KRT6A‑positive hybrid cells and a shift toward more basal‑like cells. Importantly, when cells carried a cancer‑related mutation (loss of the tumor‑suppressor gene Trp53), IL‑33 further boosted their growth. Together, these findings suggest that IL‑33 helps create a plastic, proliferative cell state that could be especially vulnerable to becoming cancerous.
Clues from Human Breast Tissue
To test whether similar cells exist in people, the authors re‑analyzed several large single‑cell datasets from normal human breast tissue. They identified a small population of human epithelial cells that, like the mouse hybrids, showed both luminal and basal features and expressed IL33 and related genes. IL33‑positive cells became more common with age across multiple datasets. In primary human mammary cells grown in the lab, IL‑33 treatment increased organoid formation, shifted cells toward a basal‑like profile, and enriched for CD44‑positive cells—a marker often associated with immature, stem‑like cells in breast cancer. Although available human samples did not allow a clean test of how early pregnancy alters this population, the parallels with mice are striking.
What This Means for Understanding Breast Cancer Protection
For a lay reader, the central message is that pregnancy seems to re‑wire how breast tissue ages. In mice, an early pregnancy prevents the buildup of IL‑33‑rich hybrid cells that blur the boundary between the breast’s main cell layers and behave in a highly flexible, growth‑ready way. IL‑33 itself can push cells toward this risky, shape‑shifting state, especially when cancer‑linked mutations are present. By reducing the number or persistence of these hybrid cells, pregnancy may lower the chances that aging breast tissue will contain the “wrong” kind of cell at the “wrong” time—one that is poised to tip over into cancer. This mechanistic insight could eventually guide strategies to mimic the protective effects of early pregnancy without requiring a pregnancy itself.
Citation: Olander, A., Medina, P., Haro Acosta, V. et al. Divergent aging of nulliparous and parous mammary glands reveals IL33+ hybrid epithelial cells. Nat Commun 17, 1898 (2026). https://doi.org/10.1038/s41467-026-68611-0
Keywords: breast aging, pregnancy and breast cancer, mammary stem cells, IL-33 signaling, hybrid epithelial cells