Clear Sky Science · en
Expanding the payload scope in antibody-drug conjugates by delivery of hydroxy-containing drugs through self-immolative phosphoramidates
Smarter Missiles Against Cancer
Cancer drugs can be incredibly powerful, but they often act like carpet bombing: they hit healthy tissues almost as hard as tumors. Antibody–drug conjugates (ADCs) were invented to solve this problem by attaching a potent drug to an antibody that homes in on cancer cells, turning the treatment into a guided missile. This article explains a new chemical “plug” between antibody and drug that makes these missiles both more precise and more flexible, opening the door to many more types of cancer medicines than today’s ADCs can carry.
Why Today’s Targeted Drugs Are Still Limited
ADCs are built from three parts: an antibody that recognizes a marker on cancer cells, a toxic drug (the “payload”), and a chemical linker that ties them together. Most approved ADCs rely on only a few kinds of payloads that damage DNA or block cell division in similar ways. One major reason is the linker: it must hold the drug tightly while the ADC circulates in the bloodstream, then let go cleanly once inside the tumor cell. Existing linkers work well only with certain chemical groups on drugs, especially amines, and often struggle with the many medicines that contain alcohol groups (–OH). This leaves a huge number of promising cancer agents on the shelf because they cannot be safely or efficiently delivered by current ADC designs.
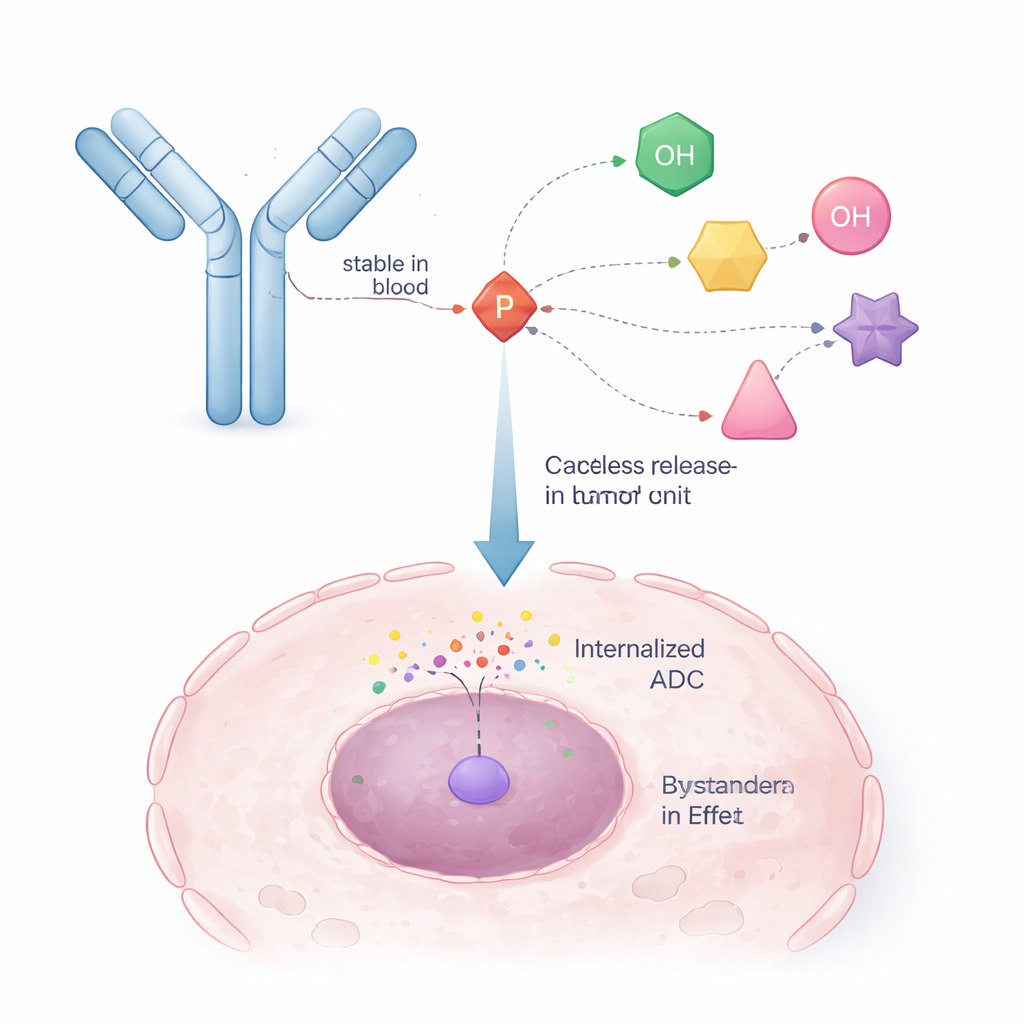
Borrowing a Trick from Antiviral Pills
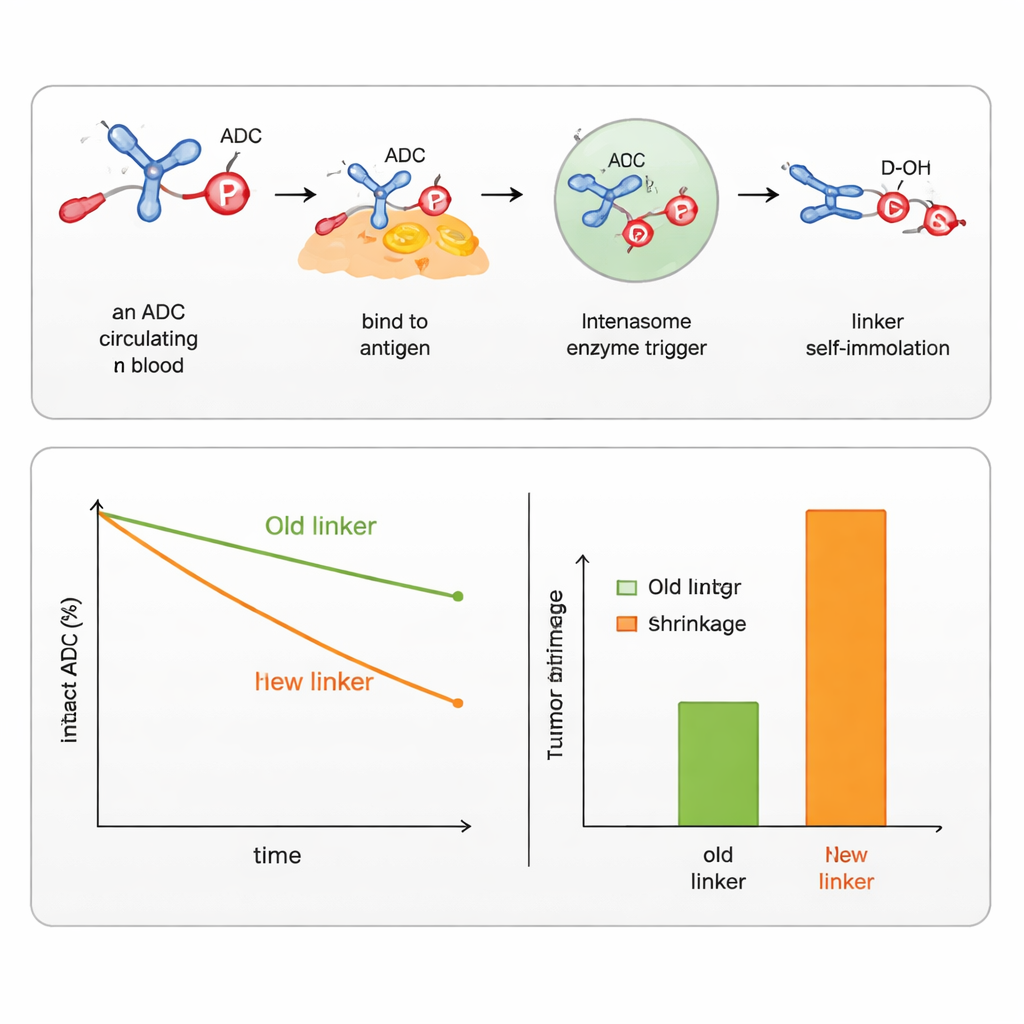
The researchers turned to a strategy already proven in antiviral drugs called ProTides. In those medicines, a phosphorus-based chemical unit temporarily masks a drug’s charged phosphate group so it can enter cells, then falls apart inside to release the active form. The authors re-engineered this concept so that one arm of the phosphorus core is used to attach to an antibody, while the other arms hold the cancer drug. This creates a “self-immolative” linker: once an internal trigger (such as an enzyme in a tumor cell) clips a small part of the structure, the phosphorus unit collapses in a controlled way and releases the original drug molecule with its alcohol group restored and unmodified.
Building a Versatile Chemical Plug
The team showed that by tweaking the groups around the phosphorus atom they could fine-tune how stable the linker is in blood and how quickly it falls apart inside cells. They designed versions that respond to several types of triggers common inside cancer cells, including esterases and proteases (enzymes that cut specific bonds), as well as enzymes that recognize sugar-based groups or reducing conditions in cell compartments. Using these designs, they successfully attached and released both aromatic alcohols (like the chemotherapy SN38) and aliphatic alcohols (like DXd, the payload in an approved breast cancer ADC). Compared head-to-head with existing commercial linkers for SN38 and DXd, the new phosphoramidate linkers kept the drugs attached for longer in serum, delivered more drug to tumors, and produced stronger tumor shrinkage in mice, while showing less impact on healthy cells.
Unlocking Many New Cancer Drug Payloads
To test how broadly the linker could be used, the researchers assembled ADCs carrying ten different drugs that all contain at least one –OH group but act in very different ways inside cancer cells. These included inhibitors of DNA synthesis, protein folding (HSP90), energy metabolism (NAMPT and DHODH), and protein translation, as well as well-known agents like paclitaxel and gemcitabine. Despite the wide range of structures, they were able to make uniform ADCs with high drug loading for all of them. In cell studies across multiple cancer types, most of these ADCs showed nanomolar or even sub-nanomolar potency and clear selectivity for cells that expressed the antibody target, confirming that the linker could consistently release active drug inside the right cells.
Gemcitabine as a Showcase
Gemcitabine is a widely used chemotherapy, but in its conventional form it is cleared from the body so quickly that patients must receive large repeated doses, which limits how much can be given safely. Using the phosphoramidate linker, the authors attached gemcitabine to a HER2-targeting antibody at either of two alcohol positions and showed that both versions regained full activity once inside cancer cells. In mouse models of HER2-positive tumors, a single dose of the gemcitabine ADC—containing about a thousand times less total gemcitabine than typical free-drug regimens—produced strong and selective tumor control. The ADC’s behavior in the bloodstream closely matched that of the naked antibody, and high doses were well tolerated in rats, suggesting a wide safety margin.
What This Means for Future Cancer Treatment
For non-specialists, the main message is that this work delivers a new, highly adaptable chemical connector that lets antibodies carry a far broader set of cancer drugs than before, and do so more safely. By ensuring that drugs stay attached while in circulation and are released cleanly only after the ADC is pulled inside a cancer cell, phosphoramidate linkers improve how much drug reaches the tumor and reduce collateral damage to healthy tissues. Just as important, they allow existing potent small-molecule drugs—with very different shapes and mechanisms—to be repurposed as targeted therapies. This could greatly expand the menu of ADCs available to patients and help tackle tumors that have become resistant to today’s limited set of payloads.
Citation: Ochtrop, P., Jagtap, A.P., Felber, J.G. et al. Expanding the payload scope in antibody-drug conjugates by delivery of hydroxy-containing drugs through self-immolative phosphoramidates. Nat Commun 17, 759 (2026). https://doi.org/10.1038/s41467-026-68605-y
Keywords: antibody–drug conjugates, cancer drug delivery, self-immolative linkers, phosphoramidate chemistry, targeted chemotherapy