Clear Sky Science · en
Operando nuclear magnetic resonance decodes alkali-tuned proton-electron relay boosting CO2-to-formate conversion
Turning a Climate Problem into a Useful Product
Carbon dioxide (CO2) is the main greenhouse gas driving climate change, but it is also a cheap, abundant raw material. Scientists are trying to turn CO2 into useful chemicals using electricity from renewable sources. This study shows how a tiny amount of lithium added to a bismuth-based material makes this CO2-to-chemicals process much more efficient, and uses advanced nuclear magnetic resonance (NMR) techniques to watch the reaction as it happens in real time.
Why Converting CO2 Is So Hard
Transforming CO2 into fuels or feedstocks is not as simple as flipping a chemical switch. CO2 is a very stable molecule, and changing it into something like formate (a liquid that can be used as a fuel or building block) requires carefully coordinated movement of both electrons and protons (hydrogen atoms without their electrons). If these moves are out of sync, the reaction either slows down or makes unwanted by-products such as hydrogen gas. The key scientific challenge is to design catalyst materials that guide electrons and protons along the right pathway at the right speed.
A Tiny Lithium Tweak with a Big Payoff
The team focused on a known CO2-reducing material called bismuth oxycarbonate. By gently introducing a trace amount of lithium into its crystal structure, they produced a new catalyst, BOC-Li. Microscopy and X-ray measurements showed that the overall structure stayed the same, but the lattice became slightly distorted and contained more subtle defects, such as missing oxygen atoms. These changes, caused by lithium sitting in specific positions, alter how the surface interacts with CO2 and water. When tested in a simple lab cell, BOC-Li converted CO2 to formate far more efficiently than the original material, delivering higher currents, less electrical resistance, and a much larger share of the desired product compared with other gases.
Watching Protons and Oxygen Move in Real Time
To understand why lithium made such a difference, the researchers turned to operando NMR, which lets them track atoms while the reaction is running. By using water and CO2 that contained rare isotopes of hydrogen, oxygen, and carbon, they could distinguish where each atom in the final formate came from. The NMR signals showed that BOC-Li produced about 21 times more formate than the undoped material under the same conditions. Crucially, the data revealed that most of the hydrogen in formate came from water near the surface, not from other ions in the solution, and that water-derived oxygen also played an active role. In other words, lithium helps set up a more direct “relay” where water at the catalyst surface delivers protons and oxygen to CO2 in a tightly coupled way.
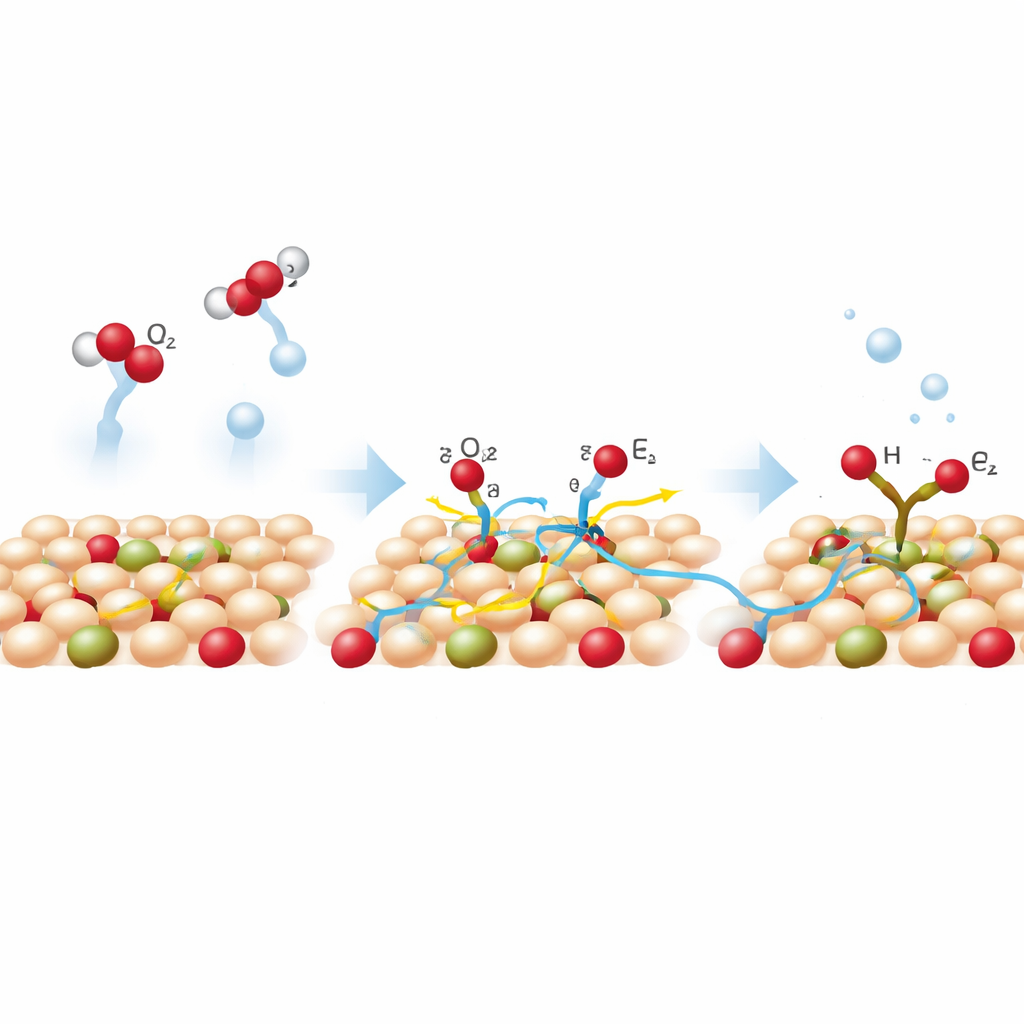
How Lithium Speeds Up the Reaction
Computer simulations helped explain this behavior. On the lithium-doped surface, both CO2 and water stick more strongly, especially near the tiny defects promoted by lithium. The energy needed to break a water O–H bond and generate a reactive hydrogen drops noticeably, meaning protons can be supplied more easily. At the same time, the preferred reaction route involves an intermediate in which CO2 is bound through oxygen on the surface before becoming formate. Lithium shifts the electronic structure of nearby atoms so this intermediate is stabilized and hydrogen is guided toward CO2 instead of pairing up to make hydrogen gas. In practical flow reactors that resemble industrial devices, the BOC-Li catalyst sustains around 90% selectivity to formate at very high current densities, and runs for hundreds of hours with little loss of performance.
From Better Catalysts to Cleaner Energy Cycles
In accessible terms, this work shows that sprinkling in a tiny amount of lithium re-tunes the “wiring” of a bismuth-based catalyst so electrons and protons arrive at CO2 together, taking the most efficient route to formate instead of side products. The combination of real-time NMR tracking and theory reveals not just that the catalyst works better, but how and why: the reaction mainly draws its hydrogen from nearby water, and lithium-created sites make it easier for water and CO2 to cooperate. This strategy could guide the design of next-generation catalysts that convert CO2 into a range of useful chemicals and fuels more efficiently, helping close the carbon loop in a future low-carbon energy system.
Citation: Shi, Y., Liu, Y., Dong, H. et al. Operando nuclear magnetic resonance decodes alkali-tuned proton-electron relay boosting CO2-to-formate conversion. Nat Commun 17, 2136 (2026). https://doi.org/10.1038/s41467-026-68604-z
Keywords: CO2 electroreduction, formate production, lithium-doped catalysts, operando NMR, proton-coupled electron transfer