Clear Sky Science · en
Membrane-free CO2 hydrogenation electrolyzer for salt precipitation management in acidic electrochemical CO2 reduction
Turning Climate Pollution into Useful Liquid Fuel
Carbon dioxide (CO2) from power plants and factories is a major driver of climate change, but it is also a cheap, abundant raw material. Scientists are racing to turn CO2 into useful chemicals using electricity from renewable sources. This study tackles a practical roadblock that has quietly limited these technologies: the buildup of salts inside industrial-style reactors that slowly choke off performance. The researchers present a new, membrane-free reactor design that keeps running smoothly for days while efficiently turning CO2 into formic acid, a liquid that can be used as a chemical feedstock, preservative, or energy carrier.
Why Today’s CO2 Devices Clog Up
Many CO2-to-chemical devices look like compact fuel cells. CO2 is fed to one side (the cathode), where it is converted into products, while water is split on the other side (the anode) to supply the needed positive charges (protons). A thin polymer sheet called a membrane sits between these two sides, allowing certain ions through but keeping the liquids separate. In alkaline or neutral liquids, much of the CO2 reacts with hydroxide to form carbonate salts instead of useful products, wasting carbon and forcing energy-hungry recycling. Acidic liquids can avoid this, but then the system depends on the membrane’s ability to deliver protons quickly and evenly. When proton delivery lags, the local acidity at the cathode drifts upward, carbonates and bicarbonates crystallize, and solid salts gradually block the gas pathways needed for CO2 to reach the catalyst.
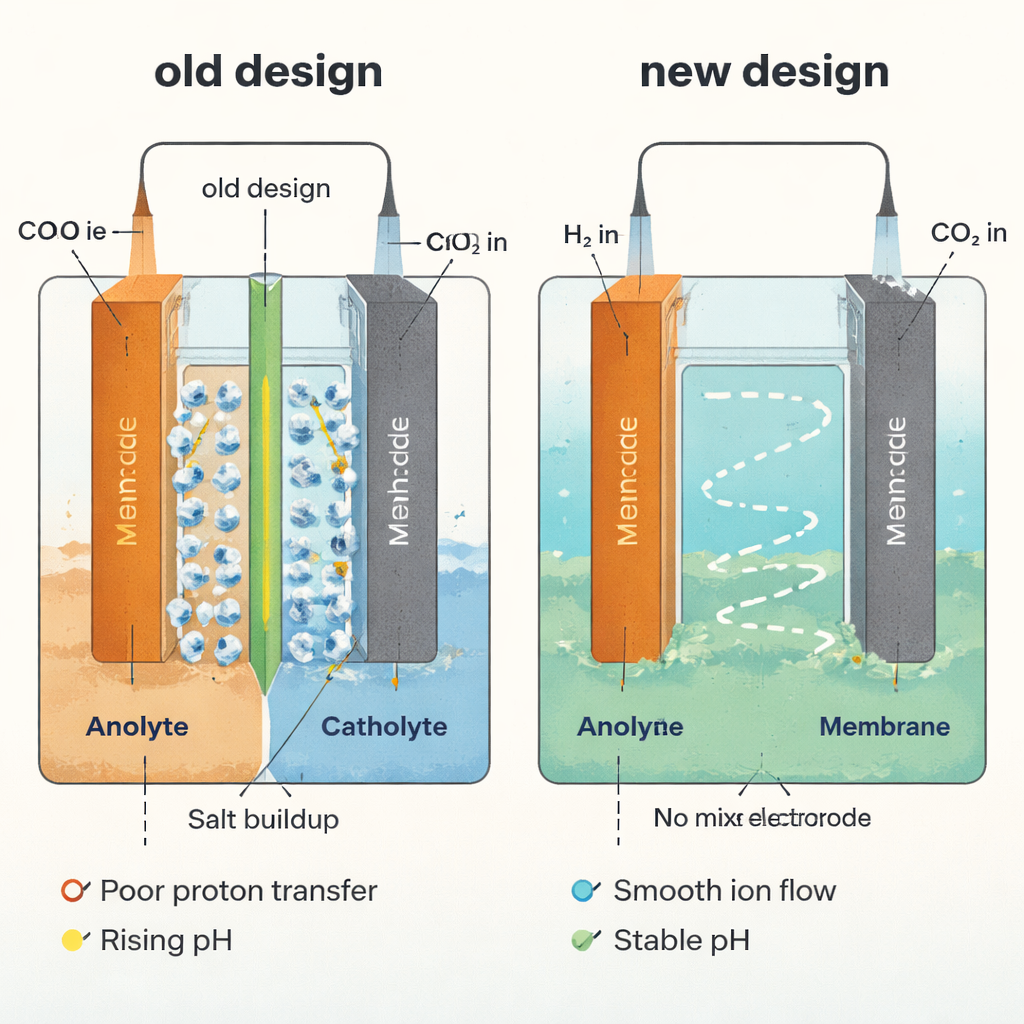
A New Way to Move Protons Without a Barrier
The team first quantified how efficiently protons move across membranes in typical acidic reactors, introducing a simple measure: the number of protons that cross per electron passing through the circuit. Using theory and computer simulations, they showed that real membranes rarely reach ideal proton transfer. Thicker films, lower proton selectivity, and certain ion mixtures all slow proton motion and create uneven acidity between the two sides. Experiments confirmed this: in a standard membrane-based cell, the catholyte (liquid at the cathode) drifted from strongly acidic to nearly neutral within hours, encouraging carbonate formation and salt precipitation deep inside the gas diffusion electrode.
Membrane-Free CO2 Hydrogenation
To escape the membrane bottleneck altogether, the researchers removed it and let a single, shared liquid flow past both electrodes. That alone stabilized the pH, but it created a new issue: valuable formic acid made at the cathode could be destroyed at a conventional anode that performs the oxygen evolution reaction, which runs at a relatively high voltage where many organic molecules are oxidized. The solution was to replace oxygen evolution with the hydrogen oxidation reaction—essentially “burning” hydrogen into protons at very low voltage. In this membrane-free CO2 hydrogenation electrolyzer, hydrogen is supplied to the anode, CO2 to the cathode, and the flowing liquid rapidly mixes the protons and hydroxide produced on each side, preventing any lasting pH gradient and sharply reducing salt buildup.
Smart Catalyst and Long-Term Performance
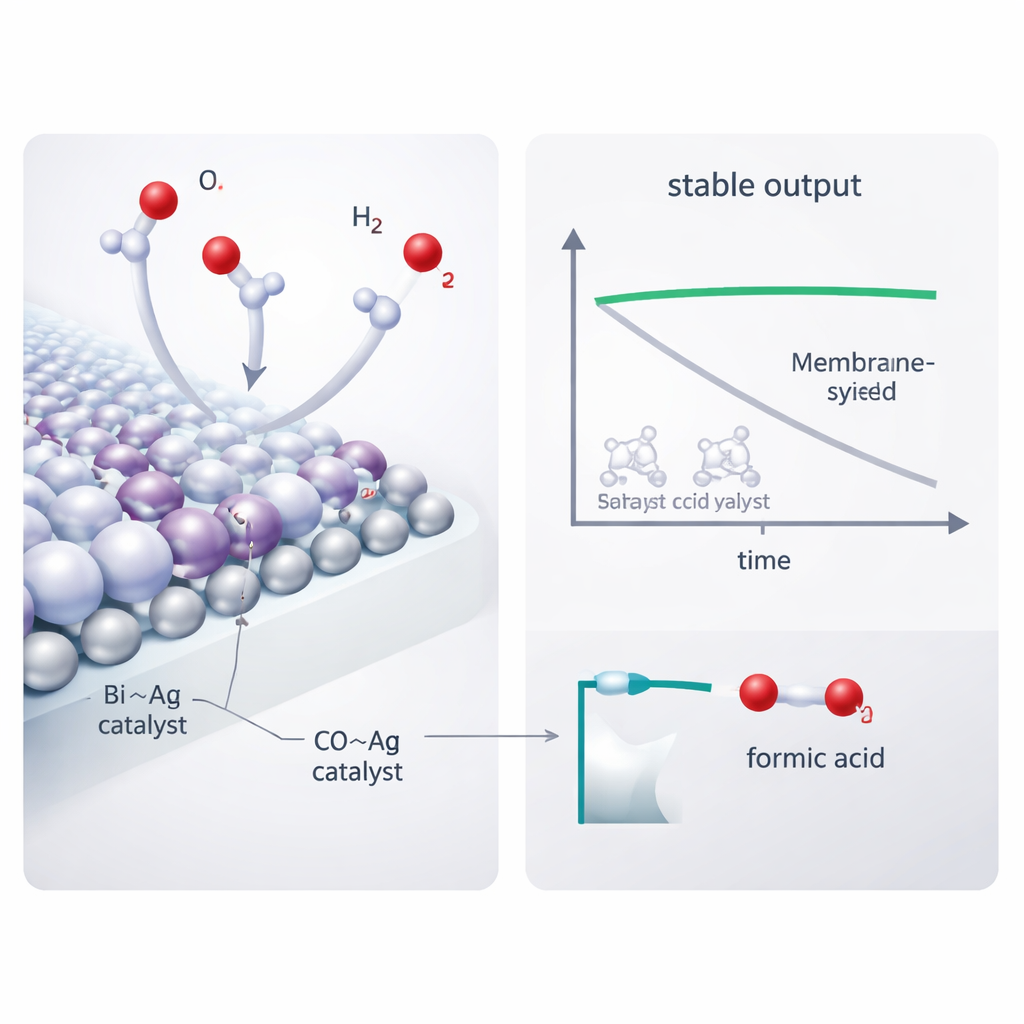
At the heart of the cathode, the authors built a bismuth–silver (Bi–Ag) catalyst that combines bismuth’s knack for making formate with silver’s excellent electrical conductivity. Microscopy and spectroscopy revealed silver nanoparticles decorating bismuth nanosheets and subtle electronic interactions between the two metals that improve CO2 adsorption and activation. In acidic solution, this catalyst converted CO2 to formic acid with more than 90% efficiency over a wide current range. When placed in the membrane-free hydrogen-coupled reactor, it delivered over 90% efficiency at 100 milliamps per square centimeter using only 1.7 volts—significantly lower than competing designs—and ran stably for 208 hours. Only traces of carbonate salt were detected in the electrode after days of operation, showing that the troublesome precipitation problem was largely suppressed.
From Lab Setup to Practical CO2 Upgrading
Beyond showing that the concept works, the team pushed the design toward real-world relevance. By shrinking the liquid channel between electrodes, they lowered electrical resistance and maintained high performance at lower voltages. They achieved a single-pass CO2 conversion efficiency of up to 77%, meaning most of the CO2 entering the cell was turned into product in one go. Economic modeling suggested that removing the membrane, cutting energy use, and achieving high carbon utilization can significantly reduce the cost of making formic acid, even though further savings will depend on cheaper electricity, better separation of the product from the liquid, and higher current operation. Overall, the work demonstrates a practical route to turning waste CO2 into a useful liquid chemical while sidestepping a key durability issue that has plagued earlier reactor designs.
Citation: Da, Y., Fan, L., Wang, W. et al. Membrane-free CO2 hydrogenation electrolyzer for salt precipitation management in acidic electrochemical CO2 reduction. Nat Commun 17, 1872 (2026). https://doi.org/10.1038/s41467-026-68600-3
Keywords: CO2 electroreduction, formic acid, membrane-free electrolyzer, hydrogen oxidation, carbon utilization