Clear Sky Science · en
Efficient solution-processed light-emitting diodes based on organic-inorganic hybrid antimony halides
A new way to make bright, efficient red LEDs
Light‑emitting diodes (LEDs) are everywhere, from phone screens to car headlights, but making them both efficient and cheap to manufacture remains a challenge. This study reports a new class of red‑emitting materials based on antimony compounds that can be processed from simple solutions, much like printing ink. By carefully redesigning the organic part of these hybrid materials, the researchers dramatically boost efficiency and lifetime, pointing toward safer, lead‑free LEDs that could someday power large, low‑cost displays and lighting panels.
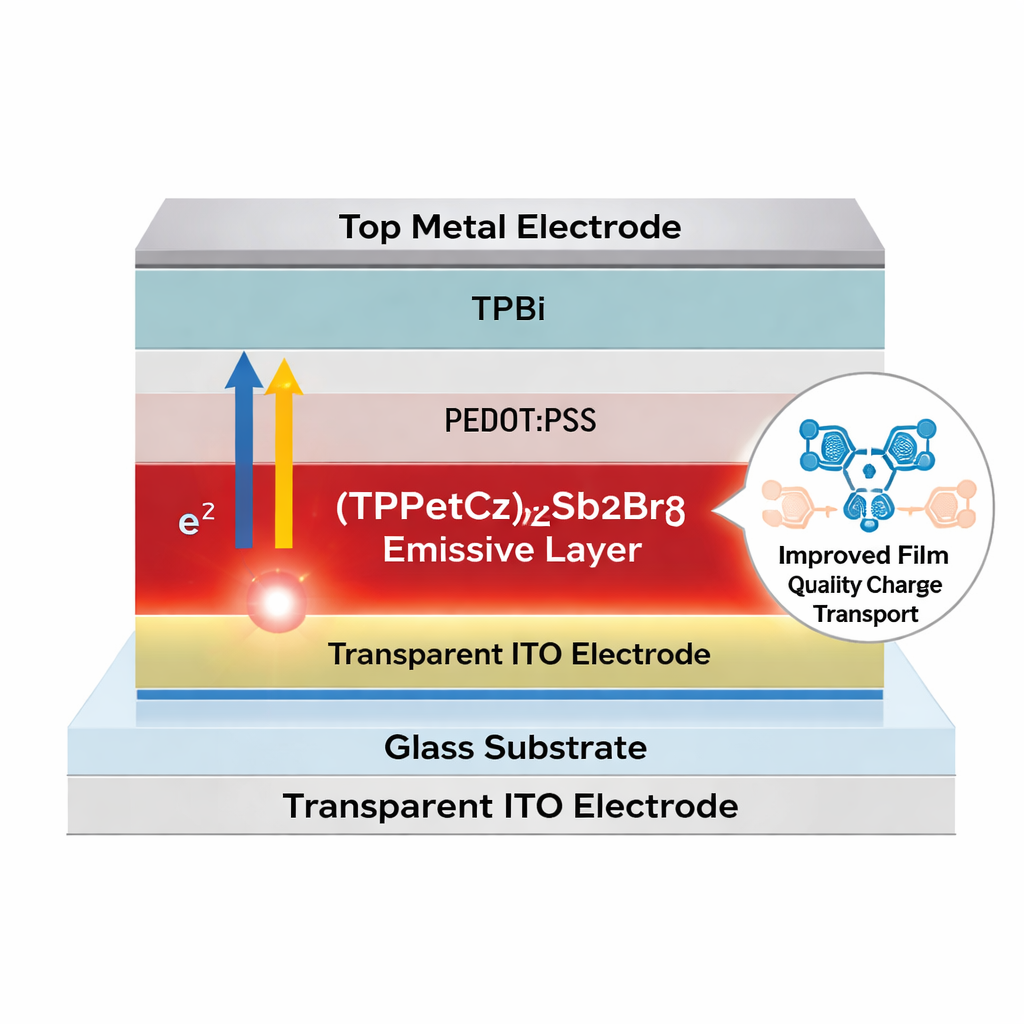
Why hybrid antimony LEDs matter
Most high‑performance LEDs today rely on either organic molecules, quantum dots, or lead‑based perovskites. Each option has drawbacks, including expensive processing, stability concerns, or the presence of toxic lead. Organic–inorganic hybrid antimony halides offer an attractive alternative: they combine the robust light‑emitting behavior of an inorganic semiconductor with the flexibility of organic molecules. In particular, their zero‑dimensional structure behaves like tiny isolated light sources, which can give very bright, stable emission. Yet, until now, devices using these materials have struggled to convert electrical energy into light efficiently, mainly because charges were not transported and recombined effectively inside the device.
Redesigning the building blocks of light
The team tackled this bottleneck by re‑engineering the organic “scaffolding” that surrounds the light‑emitting antimony–bromine units. They designed a new positively charged molecule, called TPPEtCz+, which carries a carbazole group—a flat, ring‑shaped structure that can stack neatly with similar rings in neighboring materials. When combined with antimony and bromine, this molecule forms a hybrid compound named (TPPEtCz)2Sb2Br8. Compared with an earlier control material that lacks the carbazole unit, the new compound melts at a higher temperature, has a purer crystal structure, and forms much smoother, more uniform thin films when spun from solution onto a substrate.
Smoother films and brighter light
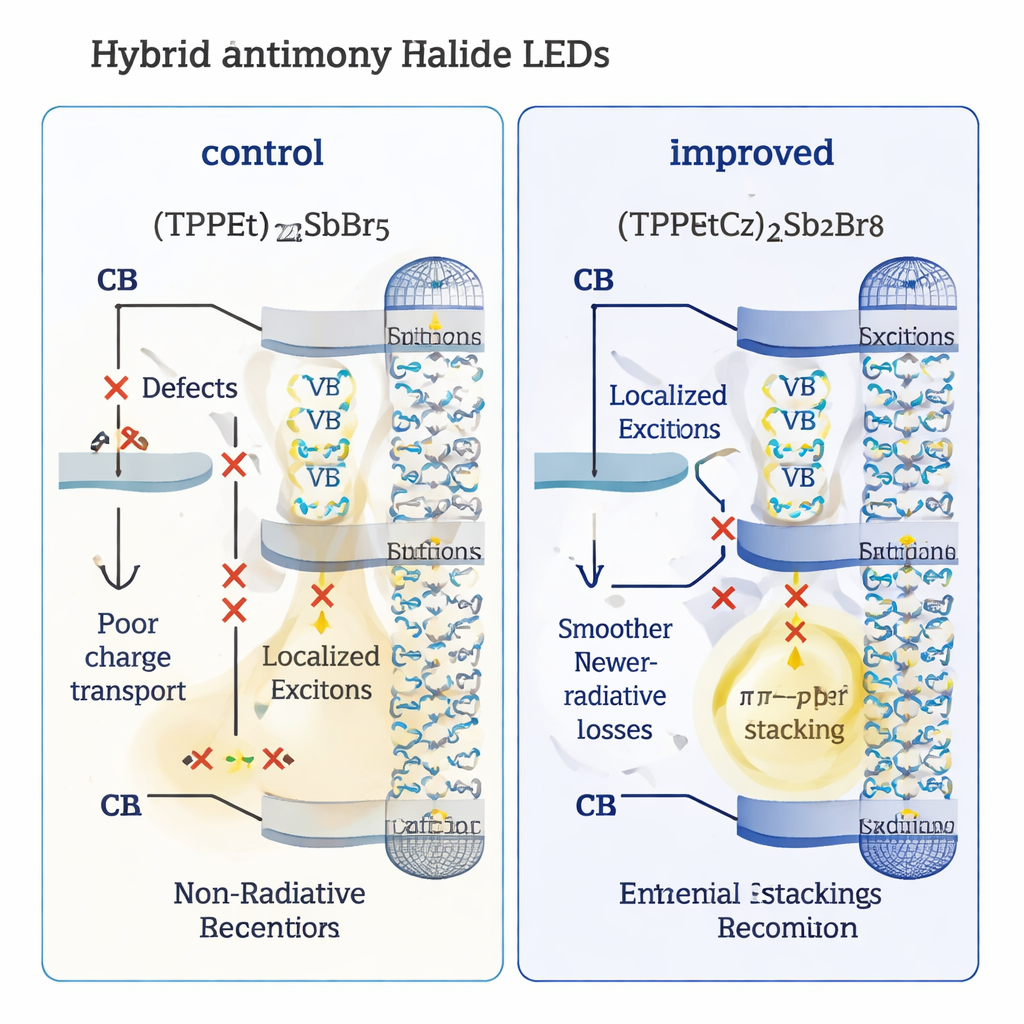
At the microscopic level, the new organic component slows down how the crystals form as the solvent evaporates. Strong hydrogen bonds between TPPEtCz+, the antimony–bromine clusters, and the solvent act like a gentle brake on crystallization, preventing the material from freezing into a rough, defective film. Measurements show that the new films have far fewer “trap” sites where excited states can die without emitting light. As a result, their light‑emitting efficiency under optical excitation (photoluminescence quantum yield) climbs to about 88%, compared with only 20% for the control. Time‑resolved experiments further reveal that the useful, radiative processes dominate, while wasteful non‑radiative pathways are strongly suppressed.
Better charge pathways inside the device
Equally important, the carbazole group helps charges move more easily across the device. The emitting layer sits next to an electron‑transport material called TPBi, which also contains flat aromatic rings. The carbazole rings in (TPPEtCz)2Sb2Br8 and the benzimidazole rings in TPBi can stack face‑to‑face, a weak but highly organized interaction known as π–π stacking. Spectroscopic measurements and computer simulations confirm that this stacking changes the energy levels at the interface and lowers the barriers for electrons to flow into the emitter. Device‑level tests show reduced electrical resistance, more balanced injection of electrons and holes, and faster, cleaner establishment of light emission when the LED is switched on, with fewer charges piling up and being wasted.
Record performance and large‑area devices
Putting these advantages together, the researchers build red LEDs with a record peak external quantum efficiency of 19.4% for lead‑free metal halide emitters—roughly four times higher than the best previous antimony‑based devices. The new LEDs also last far longer: their brightness falls to half only after about 10,000 minutes of operation at a practical luminance level, compared with just minutes for the control. The team goes on to fabricate large‑area devices more than 3 cm on a side that glow uniformly bright red, with only a small drop in efficiency. They further test several related carbazole‑based molecules and find that, while details differ, the overall strategy of using carbazole‑functionalized cations consistently improves performance over older designs.
What this means for future lighting and displays
For non‑specialists, the key message is that clever molecular design on the organic side of a hybrid material can unlock the full potential of its inorganic light emitters. By using a carbazole‑bearing cation, the researchers manage to grow cleaner crystals, reduce internal losses, and create better electrical contact inside the LED stack—all in a solution‑processed, lead‑free system. This combination of high efficiency, long lifetime, and large‑area uniformity suggests that hybrid antimony halide LEDs could become promising candidates for future low‑cost, environmentally friendlier lighting and display technologies.
Citation: Ma, Z., Chu, W., Peng, Q. et al. Efficient solution-processed light-emitting diodes based on organic-inorganic hybrid antimony halides. Nat Commun 17, 1865 (2026). https://doi.org/10.1038/s41467-026-68597-9
Keywords: antimony halide LEDs, hybrid metal halides, solution-processed lighting, lead-free perovskite alternatives, organic cation engineering