Clear Sky Science · en
Reticulating node-linker-modulator chemical spaces for modular design of alkoxide-based glasses and liquids
New Building Blocks for Everyday Glass
Windows, phone screens, and optical fibers are all made from glass, yet most glasses have very limited chemistries and fixed properties. This paper explores a new way to build glassy materials from modular molecular parts, more like snapping together LEGO bricks than melting sand. By doing so, the researchers open a route to tailor‑made glasses that can be liquid, rubbery, or solid, and can even light up in electronic devices.
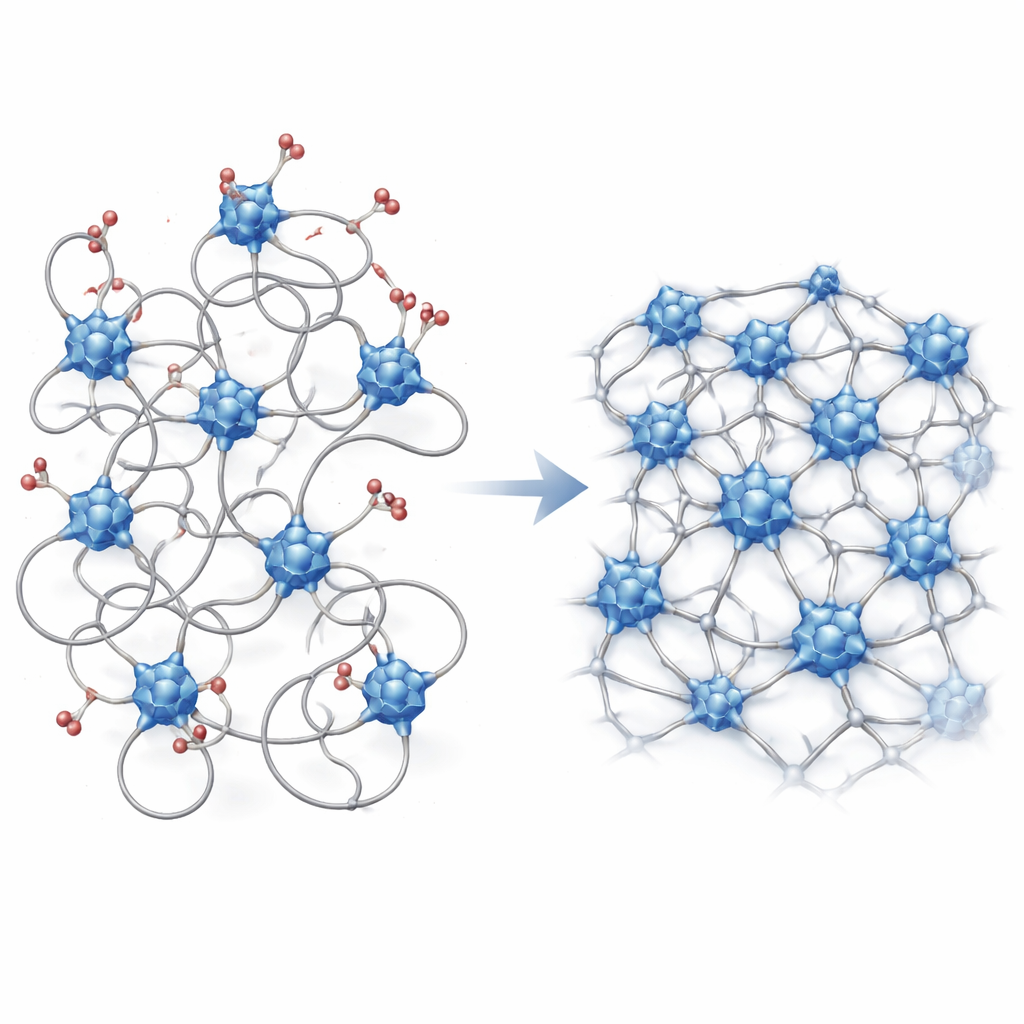
From Crystal Frameworks to Shapeable Glass
Modern materials chemists often design rigid, crystalline frameworks where metal atoms are connected by organic linkers in tidy, repeating patterns. These so‑called reticular materials can be tuned almost at will, but only a few can be melted and quenched into glasses without falling apart. The authors ask whether the same design logic used for crystals can be brought into the messy, non‑crystalline world of glasses. Their key idea is a simple formula: every material is built from nodes (metal‑oxo clusters), linkers (multidentate alcohol molecules that bridge nodes), and modulators (single‑toothed alcohols that compete with linkers for space on the nodes).
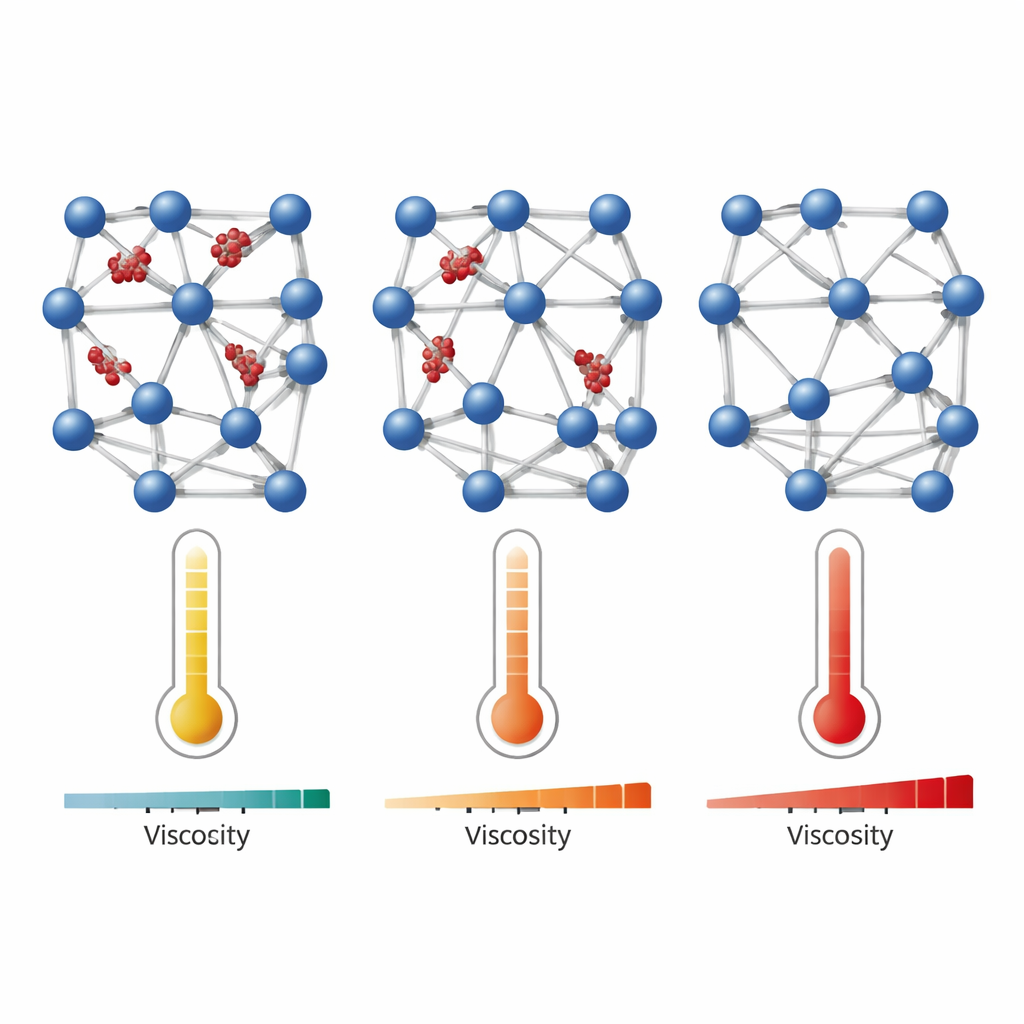
Dialing Connectivity with Molecular Modulators
In these materials, the modulators act like temporary spacers. When many modulators are present, they block linkers from tying nodes together, so the structure resembles a loose molecular soup with low viscosity and an easy, liquid‑like flow. As modulators are removed—by evaporating the alcohol solvent or by choosing lower modulator ratios—more linkers can bridge between nodes. This gradually transforms the system into a tangled polymer‑like network that resists flow and finally becomes a rigid glass. Using rheology (to measure viscosity), calorimetry (to track the glass transition), and X‑ray total scattering (to probe local structure), the team shows that decreasing modulator content steadily increases connectivity, raises the glass transition temperature, and reduces the heat capacity jump, all signatures of a stiffer, more strongly connected network.
Balancing Weak Attractions and Strong Bonds
The glass transition temperature in these systems is not set by a single factor. Instead, it arises from a tug‑of‑war between weak, non‑covalent attractions among molecules and the strong, covalent‑like bonds that knit the network together. By swapping liquid modulators for solid ones, or by changing the flexibility and shape of the linkers, the authors can see when the behavior is dominated by modulator–modulator interactions (much like a concentrated solution) and when the network itself takes over. In some series, adding more node–linker bonds always stiffens the material and raises its glass transition. In others, especially those based on flexible polyether linkers, reducing modulators lowers the glass transition at first—because favorable weak interactions are lost—before the growing network finally wins out and pushes the transition temperature back up.
Swapping Metals and Even Removing Them
To show that their strategy is truly modular, the researchers move beyond titanium clusters to analogous systems built from zirconium, and then to fully organic boron‑based networks with similar alkoxide‑type linkages. Across these families, the same node‑linker‑modulator rules apply: metal or boron clusters serve as hubs, flexible linkers tie them together, and small alcohol‑like molecules fine‑tune the connectivity and motion. X‑ray scattering and composition analysis confirm that all of these materials form non‑crystalline networks with tunable local structures and thermal behaviors, substantially enlarging the “chemical space” of possible glasses.
Lighting Up Modular Glasses
Finally, the team demonstrates a practical payoff of this design freedom. They embed a fluorescent aromatic linker into titanium, zirconium, and boron networks to create bright, blue‑emitting glassy materials. The boron‑based glass, in particular, reaches a high quantum yield and can be cast as a clear plate. As a proof of concept, the authors use this glass as the light‑emitting layer in a simple alternating‑current electroluminescent device, where charges injected from carbon nanotube and metal contacts recombine to produce light. Although the prototype operates at relatively high voltage and is not optimized, it illustrates that these modular network glasses can be processed like polymers yet retain the robustness and design flexibility of reticular frameworks.
Why This Matters for Future Materials
By treating glassy materials as combinations of nodes, linkers, and modulators, this work brings the powerful, mix‑and‑match mindset of reticular chemistry into the realm of non‑crystalline solids. The result is a versatile recipe for designing alkoxide‑based glasses whose flow, stiffness, and optical properties can be tuned by changing a few molecular building blocks and their ratios. Such control could eventually yield customizable, processable glasses for displays, sensors, and other optoelectronic technologies, all built from modular chemical parts rather than a single, fixed glass composition.
Citation: Liu, Y., Geng, Y., Deng, Y. et al. Reticulating node-linker-modulator chemical spaces for modular design of alkoxide-based glasses and liquids. Nat Commun 17, 1863 (2026). https://doi.org/10.1038/s41467-026-68591-1
Keywords: glassy materials, reticular chemistry, alkoxide networks, modular design, electroluminescent glass