Clear Sky Science · en
Multiple hydrogen bonding enables large-area doped phosphorescent glasses with robust stability and high-temperature afterglow
Glowing glass that keeps shining
Imagine a sheet of clear plastic-like glass that you can cut, mold, or print into shapes, charge for a moment with a small UV lamp, and then watch it glow for tens of seconds—even in hot ovens or harsh solvents. This study describes exactly such a material: a new class of organic glowing glasses that combine long-lasting afterglow, toughness, and easy processing, opening doors for safer emergency signs, anti-counterfeiting labels, and futuristic displays. 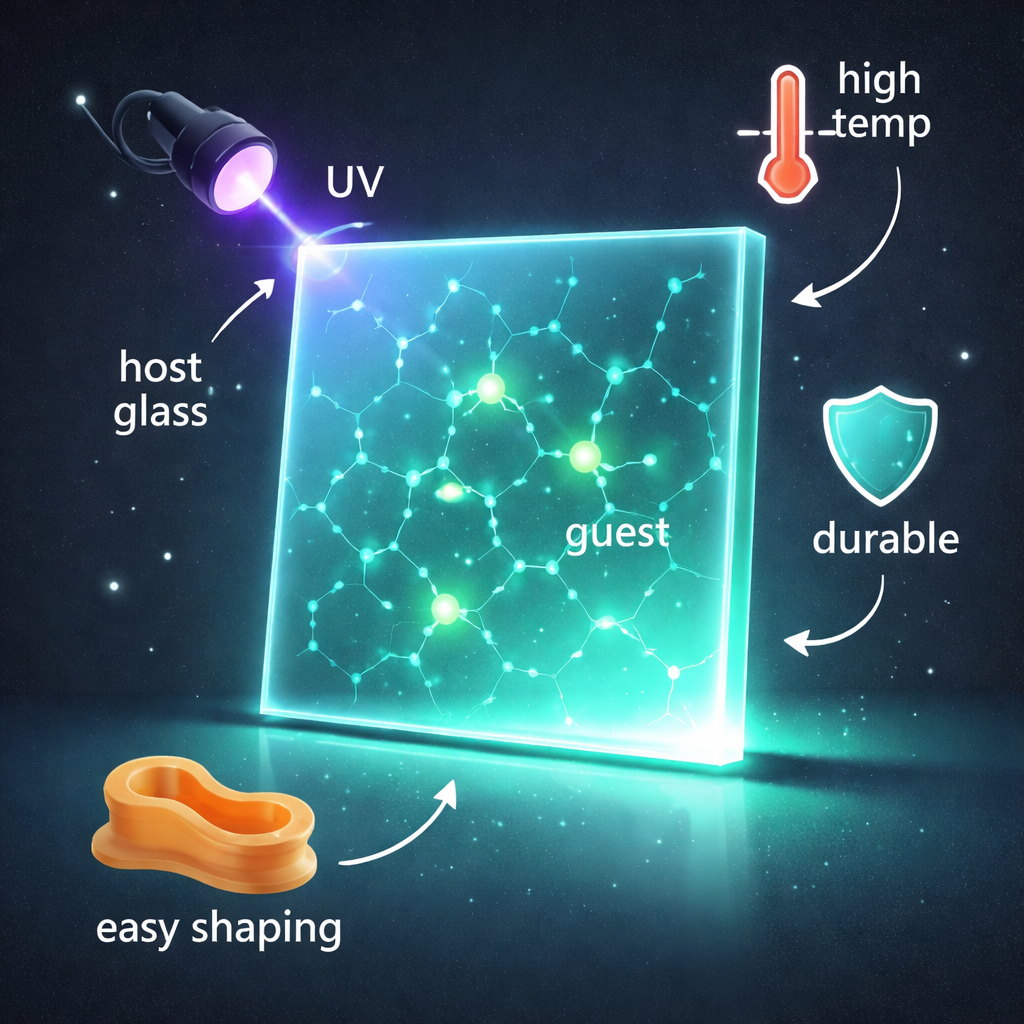
Why long‑lasting glow matters
Most everyday glow-in-the-dark items rely on inorganic crystals that are hard, brittle, and require high temperatures to make. Organic glow materials, built from carbon-based molecules, promise lighter, more flexible, and more easily tunable alternatives. However, getting organic materials to store light efficiently and release it slowly (a behavior called persistent phosphorescence, or afterglow) at room temperature is difficult. The excited states that hold the stored light are easily drained by tiny molecular motions or by oxygen in the air, so the glow usually fades quickly or is too weak for practical use.
Building a better glowing glass
The research team tackled this challenge by designing a special host–guest system. The host is a small, non-conjugated molecule called 1,2,3,4-butane tetracarboxylic acid (BTA), which carries several acid groups able to form many hydrogen bonds. When a concentrated solution of BTA in ethanol is slowly dried, the molecules do not crystallize into a rigid, ordered lattice. Instead, they form a clear, amorphous glass—essentially a molecular “frozen liquid” with no long-range order but high local density. Into this host glass, the team doped tiny amounts of rigid aromatic anhydride “guest” molecules, which are good light emitters but on their own do not show strong afterglow at room temperature.
How hydrogen bonds lock in the light
Careful experiments and computer simulations revealed why this combination works so well. In the glass, BTA molecules arrange themselves in a disordered yet tightly linked network, held together by many hydrogen bonds between their acid groups. These connections create a rigid micro-environment that cages the guest molecules, restricting their vibrations and rotations that would otherwise waste energy as heat. At the same time, the multiple carbonyl and oxygen atoms in both host and guest help funnel excited electrons into long-lived triplet states, where the stored energy can be released slowly as phosphorescent afterglow. The result is a transparent glass that glows for up to 40 seconds, with a phosphorescence efficiency as high as 56.8%, among the best reported for purely organic materials.
Staying bright in tough conditions
Unlike traditional crystals, the BTA-based glass keeps its performance under demanding environments. The afterglow remains visible up to 200 °C, a temperature at which many organic emitters would fail, and the material tolerates repeated heating and cooling cycles with little loss of emission. It also remains amorphous and glowing after months in air and after soaking in many different organic solvents, from nonpolar liquids like hexane to polar ones like dimethyl sulfoxide. Because the glass is formed from solution at modest temperatures and has a relatively low glass transition temperature, it can be molded thermoplastically into bulk objects or large-area panels without cracking or crystallizing. 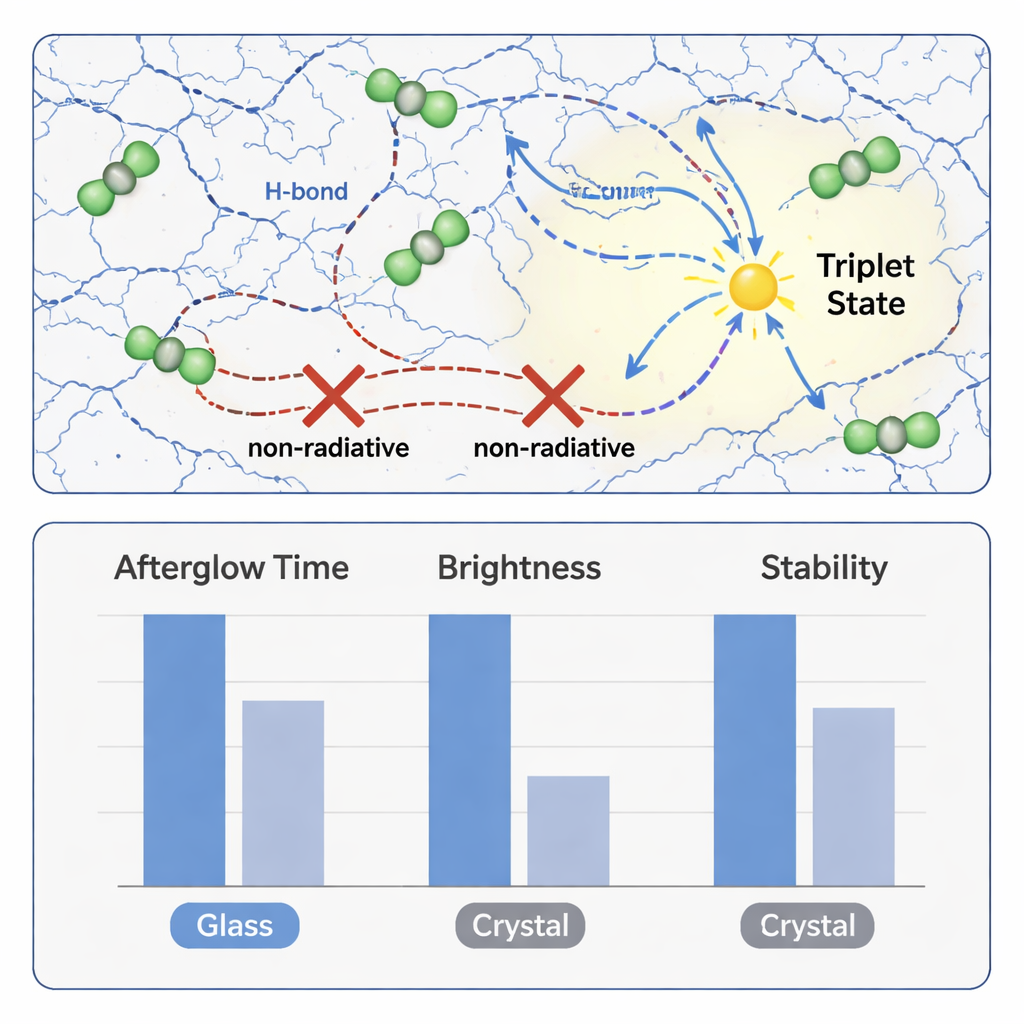
From lab glass to practical gadgets
These properties make the material highly practical. The authors demonstrate a 25 cm × 25 cm glowing panel that can act as a self-powered emergency map: a short UV exposure charges the panel, which then emits enough light to reveal printed details in the dark. They also show 3D-shaped glowing objects and multicolor bulk glasses formed by gently fusing pieces doped with different guests. Finally, by coating a UV LED array with different versions of the glass, they create time-staggered glowing number patterns that appear only after the power is switched off, hinting at uses in information encryption and security labels.
What this means for future glowing materials
In simple terms, the study shows that many small hydrogen bonds, arranged in a disordered glass rather than a perfect crystal, can stabilize light-storing states extremely well. The BTA host acts like a tough, transparent scaffold that both protects and activates the glow of the guest molecules. Because the approach is chemically flexible and works with different guests to produce various colors, it offers a general recipe for crafting large-area, shapeable, long-afterglow glasses for advanced displays, smart lighting, and anti-counterfeiting technologies.
Citation: Chen, C., Yang, Y., Zhang, L. et al. Multiple hydrogen bonding enables large-area doped phosphorescent glasses with robust stability and high-temperature afterglow. Nat Commun 17, 1870 (2026). https://doi.org/10.1038/s41467-026-68590-2
Keywords: afterglow glass, room temperature phosphorescence, hydrogen bonding, organic luminescent materials, anti-counterfeiting