Clear Sky Science · en
Disruption of iron homeostasis sensitizes pancreatic cancer to irreversible electroporation
Turning Electricity and Iron Against a Deadly Cancer
Pancreatic cancer is one of the hardest cancers to treat, and even aggressive local therapies often fail to stop it from coming back. This study explores a new way to make an existing procedure—irreversible electroporation, which kills tumors using short, powerful electric pulses—work better and more safely. By cleverly disturbing how cancer cells handle iron, the researchers show they can push surviving cells into a destructive form of death, potentially lowering the chance that the tumor will return.
Why Electric Pulses Alone Are Not Enough
Irreversible electroporation (IRE) treats tumors by inserting thin electrodes in or around the cancer and firing rapid, high-voltage pulses. These pulses punch permanent holes in cell membranes, killing many cancer cells and stirring up an immune response. But the electric field is not perfectly even. Areas that receive slightly weaker pulses can leave cancer cells alive, and these survivors can seed a new tumor. When the team looked closely at pancreatic cancer cells and mouse tumors exposed to a sub-lethal electric field, they found that instead of dying by an iron-driven process called ferroptosis, these cells switched on a broad antioxidant defense program. Key protective genes, including those that help neutralize iron-related damage, were turned up, allowing the cells to weather the storm and recover.
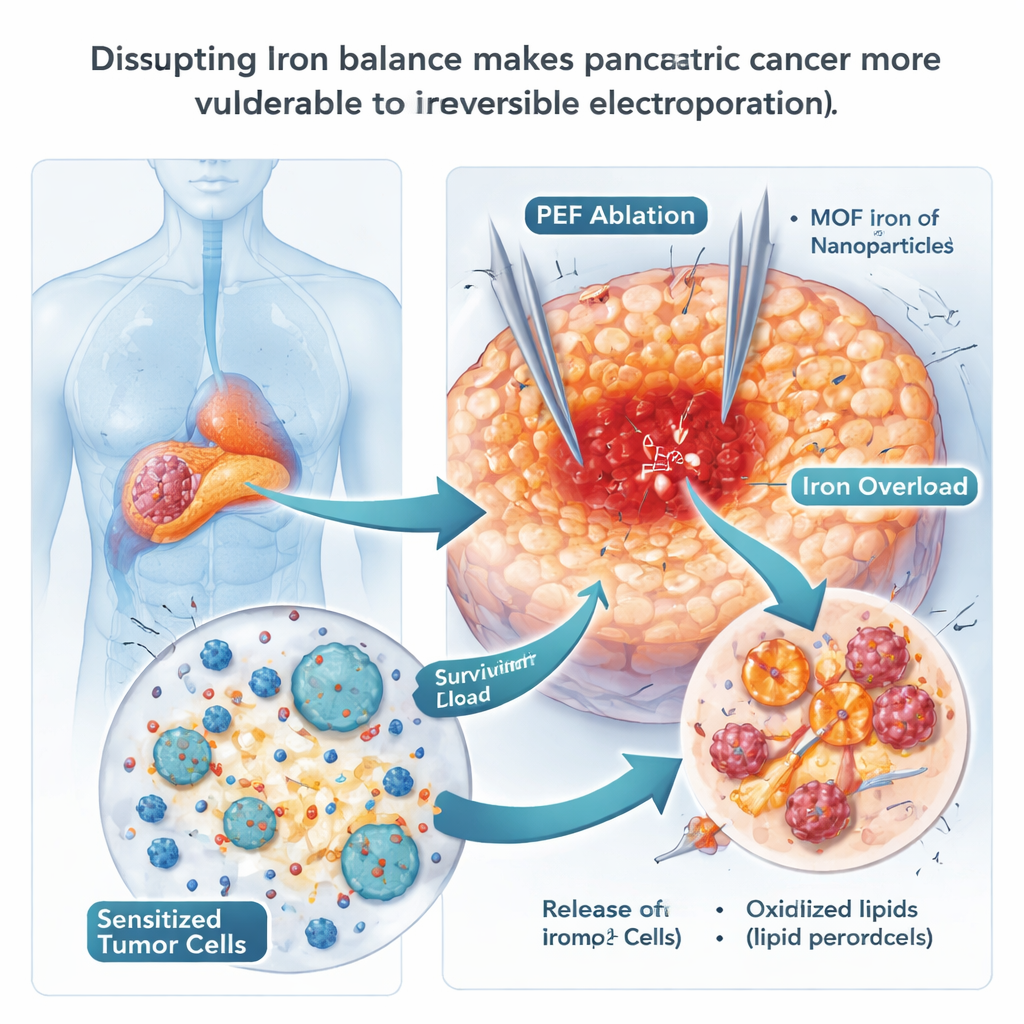
Using Iron Nanoparticles to Push Cells Toward Destruction
Because ferroptosis depends on iron, the scientists tested whether iron-rich nanoparticles could force cancer cells over the edge. They used specially designed metal–organic framework particles containing iron (MOF-Fe), which quickly release iron inside the acidic compartments of cells. In dishes, pancreatic cancer cells that took up these particles accumulated free iron and oxidized fats in their membranes, classic hallmarks of ferroptosis, and formed far fewer colonies. In mice, combining MOF-Fe with IRE delayed tumor regrowth longer than either treatment alone, and tumors showed more signs of oxidative damage. However, this benefit was temporary: after about three weeks, tumors began to rebound, suggesting that the cancer had found a way to adapt to the iron overload.
Targeting the Cell’s Iron “Safe”
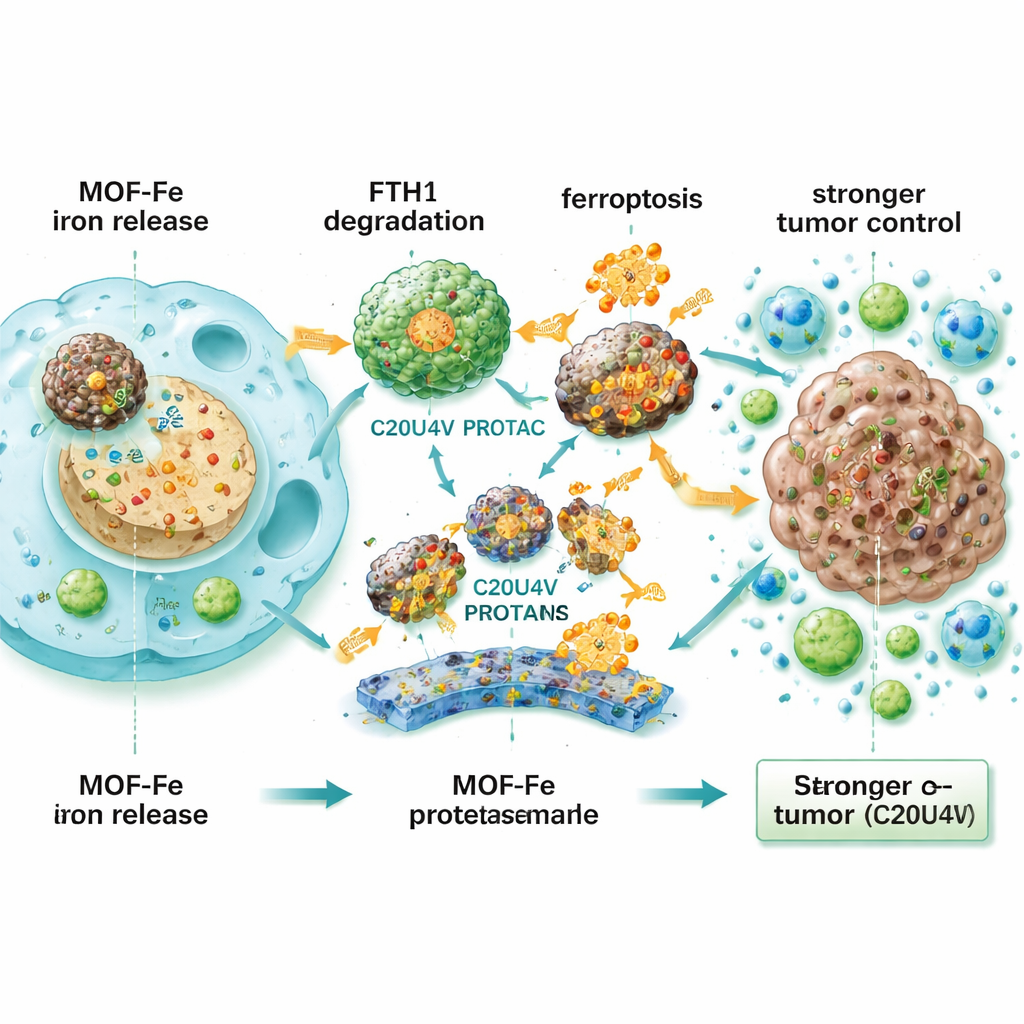
To understand this resistance, the team analyzed which proteins changed after MOF-Fe treatment. A standout was ferritin heavy chain 1 (FTH1), part of the cell’s iron storage complex—the molecular equivalent of a fireproof safe for excess iron. When FTH1 levels were experimentally raised, cancer cells became more resistant to ferroptosis; when FTH1 was lowered, they became more vulnerable. This pointed to FTH1 as a central shield against iron-driven death. The researchers then created a designer molecule, C20U4V, built from arachidonic acid (a highly oxidizable fat) linked to a “PROTAC” scaffold that directs specific proteins to the cell’s waste-disposal machinery. In the presence of MOF-Fe, C20U4V bound ferritin and recruited it to be tagged and destroyed, sharply reducing FTH1 levels and preventing the cell from safely locking away iron.
From Cell Dishes to Mini-Tumors and Mice
When MOF-Fe and C20U4V were combined, pancreatic cancer cells showed higher oxidative stress, more damaged membrane fats, sicker mitochondria, and greatly reduced ability to regrow. This synergy also appeared in three-dimensional patient-derived organoids—tiny tumor-like structures grown from real patients—where the drug pair collapsed organoid size and number. Because C20U4V is oily and does not dissolve in water, the team packaged it into tiny, reactive-oxygen-sensitive micelles (M-C20U4V) that circulate in the bloodstream and can release the drug in stressed, damaged tumor tissue. In mouse models with pancreatic tumors placed under the skin and in the pancreas itself, a triple combination of IRE, MOF-Fe, and M-C20U4V shrank tumors far more effectively and prolonged survival compared with any single or double treatment. Treated tumors showed fewer dividing cells, more markers of ferroptosis, and heavier infiltration by T cells and other immune cells, indicating that the strategy not only kills cancer cells directly but also helps the immune system recognize and attack the tumor.
What This Could Mean for Patients
To a non-specialist, the key idea is simple: pancreatic tumors often escape after electric-pulse ablation because some cancer cells survive and quickly adapt. This work shows that loading those cells with iron and simultaneously disabling their iron “safety vault” can force them into a self-destructive state they cannot easily resist. While this approach is still at the experimental stage and would need extensive testing for safety and practicality in humans, it suggests that fine-tuning how tumors handle a basic element like iron could turn an imperfect local treatment into a more decisive blow against one of the deadliest cancers.
Citation: Li, L., Su, S., Wang, Z. et al. Disruption of iron homeostasis sensitizes pancreatic cancer to irreversible electroporation. Nat Commun 17, 1866 (2026). https://doi.org/10.1038/s41467-026-68585-z
Keywords: pancreatic cancer, irreversible electroporation, ferroptosis, iron nanoparticles, PROTAC therapy