Clear Sky Science · en
Additive-specific modulation of non-classical nucleation pathways
Why tiny additives matter for everyday materials
From sturdy concrete to simple wallboard and even our own bones, many materials form when minerals crystallize out of water. Industry routinely adds tiny amounts of helper molecules to steer this crystallization, but exactly how these additives work at the earliest stages has remained murky. This study peeks inside that “black box,” revealing how different additives reshape the birth of crystals in two common minerals—portlandite (linked to cement) and gypsum (used in plaster and wallboard)—and how this knowledge could guide greener, more efficient formulations.
Crystals that do not form in one leap
Textbooks often portray crystal formation as a single leap: once a solution is supersaturated, atoms or ions assemble directly into a tiny crystal that then grows. Here, the authors show that both portlandite and gypsum follow a more complex, stepwise route. First, dissolved ions (like calcium, hydroxide, or sulfate) associate into small, dynamic clusters called prenucleation species. These then aggregate into larger, disordered blobs that are more like dense liquids or amorphous solids than proper crystals. Only after this intermediate stage do well-ordered crystals emerge. Crucially, the timing and sharpness of these transitions differ: portlandite shifts gradually from disorder to order, whereas gypsum stays disordered for longer and then transforms abruptly into crystals, more like flipping a switch than turning a dimmer.
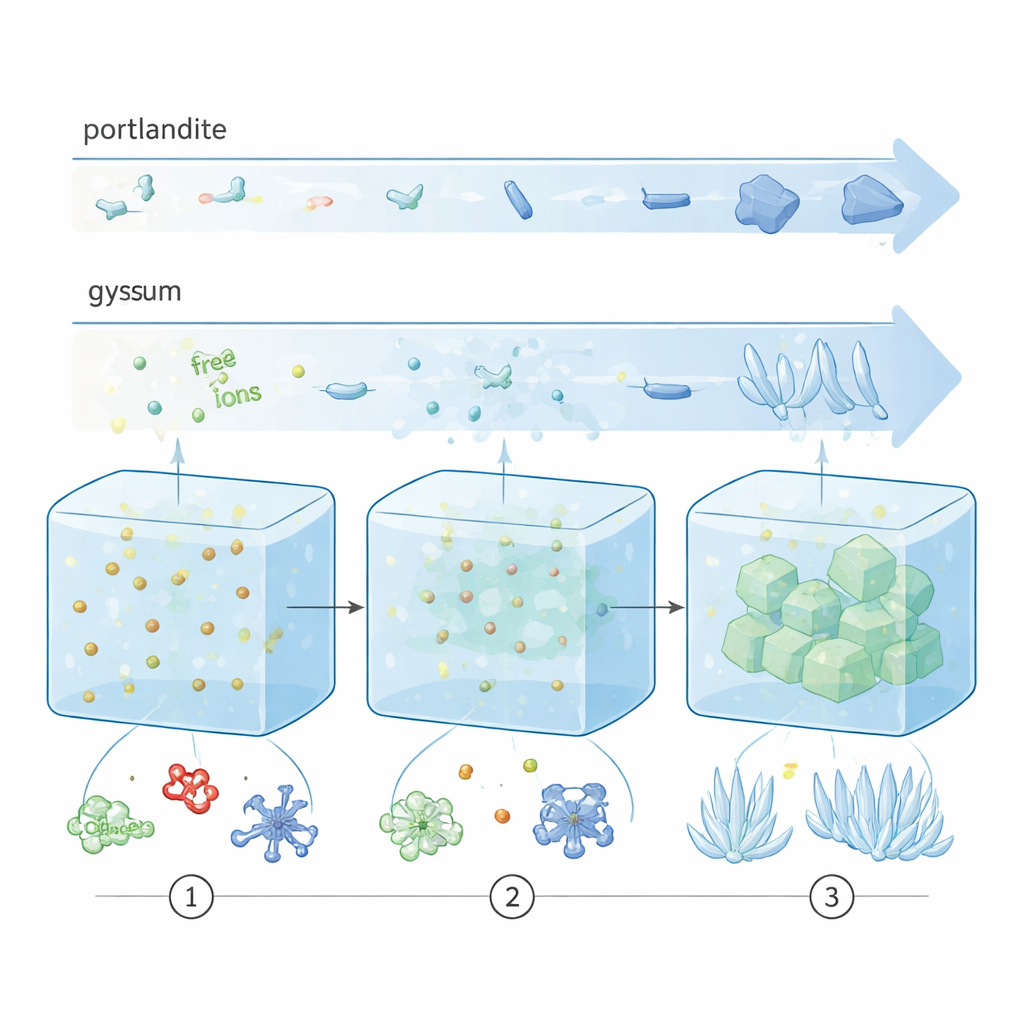
Watching nucleation unfold in real time
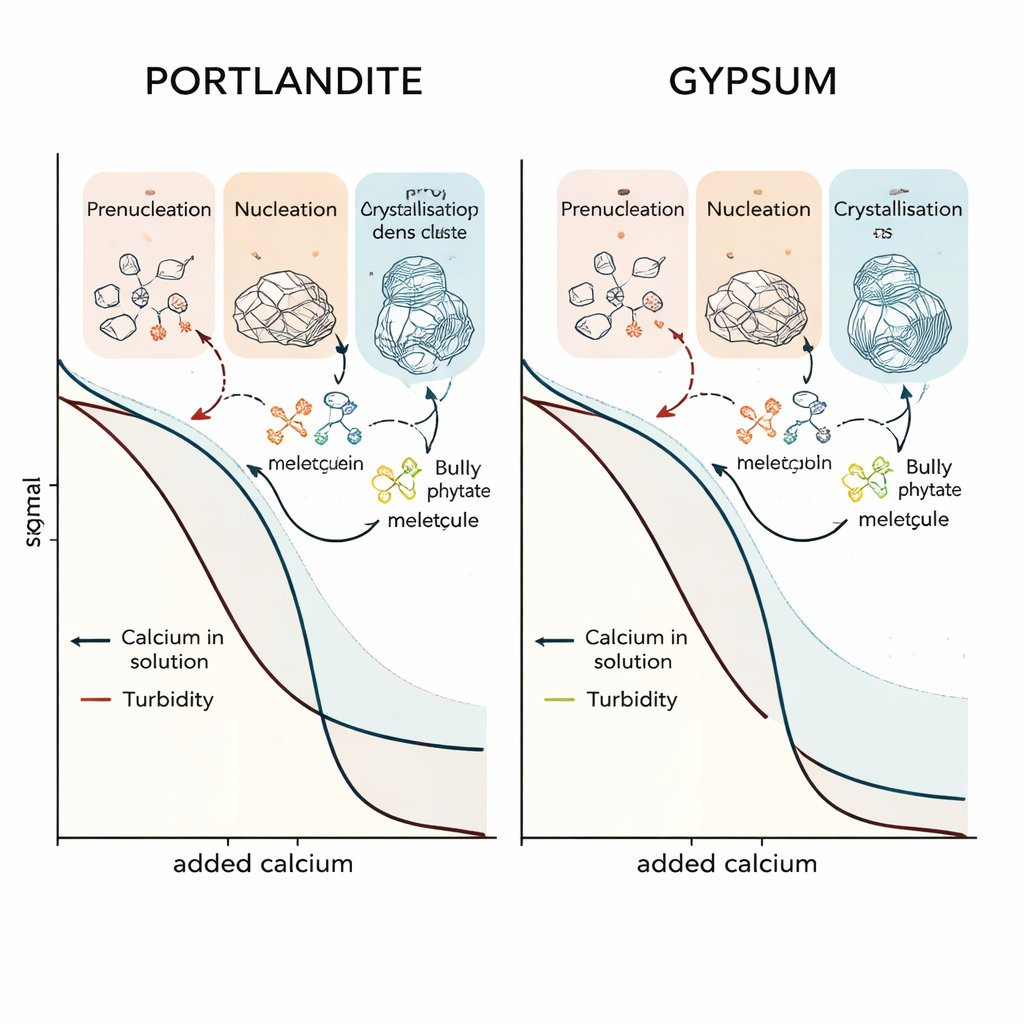
To follow these hidden steps, the team combined a specialized titration setup with high-energy X-ray scattering at a synchrotron. They slowly dosed calcium solutions into water containing the other needed ions and, in some runs, different organic additives. Probes tracked pH, conductivity, free calcium, and turbidity (how cloudy the solution became), while the X-rays revealed how atomic arrangements evolved from fully dissolved ions to partially ordered structures. By analyzing how the scattering patterns changed, the researchers could distinguish three stages: ion association in clear solution, the appearance of phase-separated but still disordered particles, and finally the growth of crystalline domains. Molecular dynamics simulations helped interpret what these early clusters look like at the atomic scale.
Additives that push and pull on different steps
The authors then added three industrially relevant or “green” molecules: a small phosphate ring (STMP), a short-chain polymer (polyacrylic acid, PAA), and a plant-derived molecule rich in phosphate groups (phytic acid). They discovered that these additives do far more than simply bind calcium. Instead, they act in a stage- and mineral-specific way, sometimes delaying one step while accelerating another. For portlandite, PAA encourages the formation of a liquid-like, amorphous calcium-rich phase, stabilizing it and slowing its conversion into crystals, yet it also triggers tiny crystalline domains unusually early at the nanoscale. STMP, in contrast, helps prenucleation clusters aggregate into an intermediate but then slightly delays the final crystallization, apparently stabilizing clusters of a particular size that must reorganize before crystals can grow. Phytic acid forms large complexes in alkaline portlandite solutions but barely changes the overall nucleation timing.
Same additive, different mineral, different outcome
A striking lesson is that the same additive can behave very differently in gypsum than in portlandite. Gypsum forms near neutral pH and includes water as part of its crystal structure, contributing to its abrupt, cliff-like crystallization step. In this system, phytic acid strongly promotes the build-up of stable, non-crystalline calcium–sulfate clusters, greatly delaying when gypsum finally appears. PAA, meanwhile, mainly stretches out the time between the first appearance of particles and the onset of rapid crystal growth, acting as an effective crystallization retarder without forming a polymer-induced liquid phase. STMP, which has a pronounced effect on portlandite, barely alters gypsum’s overall behavior. These differences stem from both pH (which changes how additives are charged) and the nature of the early clusters: charged precursors can be bridged into dense amorphous networks, whereas neutral precursors are more easily trapped as stable, nanoscale particles.
From generic additives to mineral-specific design
For non-specialists, the key takeaway is that crystal formation is less like freezing water and more like a multi-act play, with additives nudging different scenes in different directions. The study shows that additives exert much of their influence before any crystal is visible, during the formation and aggregation of tiny clusters and amorphous phases. Because portlandite and gypsum follow distinct non-classical pathways, an additive that works well for one may fail—or even act in an opposite way—for the other. Understanding these subtleties opens the door to designing smarter, more sustainable additives that are tailored to a specific mineral and operating condition, improving everything from cement strength and workability to scale prevention and biomimetic materials.
Citation: Baken, A., Fernandez-Martinez, A., Lanson, M. et al. Additive-specific modulation of non-classical nucleation pathways. Nat Commun 17, 1925 (2026). https://doi.org/10.1038/s41467-026-68583-1
Keywords: crystallization additives, non-classical nucleation, portlandite, gypsum, prenucleation clusters