Clear Sky Science · en
Identification of Chlamydia pneumoniae and NLRP3 inflammasome activation in Alzheimer’s disease retina
Why germs in the eye matter for memory
Alzheimer’s disease is usually thought of as a problem of plaques and tangles in the brain. This study suggests that a common pneumonia-causing bacterium, Chlamydia pneumoniae, may quietly settle in the light-sensing tissue at the back of the eye—the retina—and help drive the inflammation and nerve damage seen in Alzheimer’s. Because the retina can be imaged non‑invasively, this work hints that eye exams might one day help detect infection‑linked brain disease and guide early treatment.
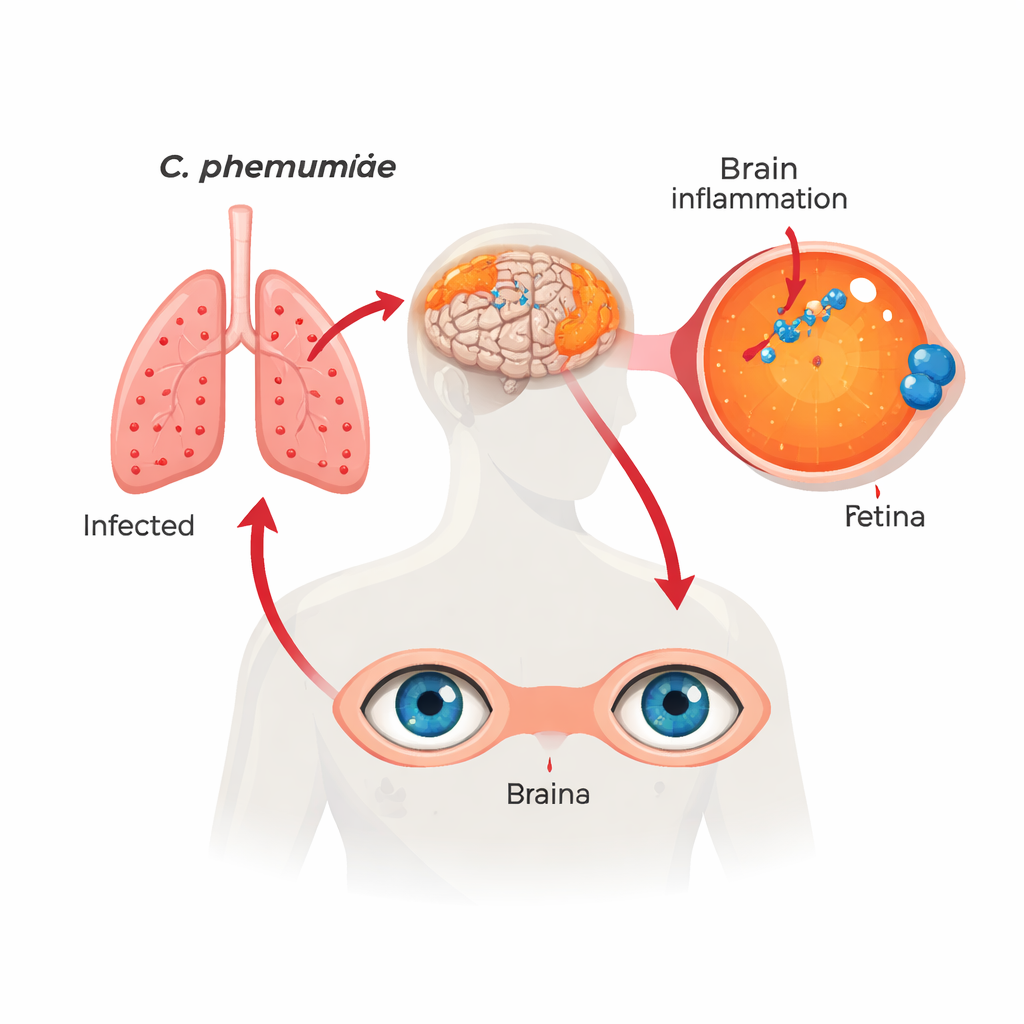
A familiar infection in an unexpected place
The researchers examined retinas and matching brain samples from over 100 deceased people who had normal thinking, mild cognitive impairment, or Alzheimer’s dementia. Using several independent techniques—including special antibodies, genetic probes, staining methods, and DNA tests—they found clear signs of Chlamydia pneumoniae inside retinal cells. These bacterial “inclusions” were most common and abundant in people with Alzheimer’s, less so in those with mild impairment, and least in those who had remained cognitively normal. The amount of bacteria in the retina closely mirrored the amount found in the brain, suggesting that both eye and brain are involved in the same long‑running infection.
Links between bacteria, plaques, and thinking ability
Finding the germ was only the first step; the key question was whether it mattered for disease. The team measured classic Alzheimer’s markers in the same tissues. They saw that higher bacterial load in the retina went hand‑in‑hand with more amyloid‑beta deposits—the sticky protein that forms plaques—as well as certain abnormal forms of tau protein. People whose brains carried more bacterial signal also had more nerve‑fiber tangles, worse overall Alzheimer’s pathology, and more brain shrinkage. Clinically, those with higher retinal bacterial burden tended to score lower on memory and thinking tests and had more advanced dementia. Carriers of the APOE ε4 gene variant, a strong genetic risk factor for Alzheimer’s, harbored more of the bacterium in both retina and brain, hinting at a gene–infection interaction.
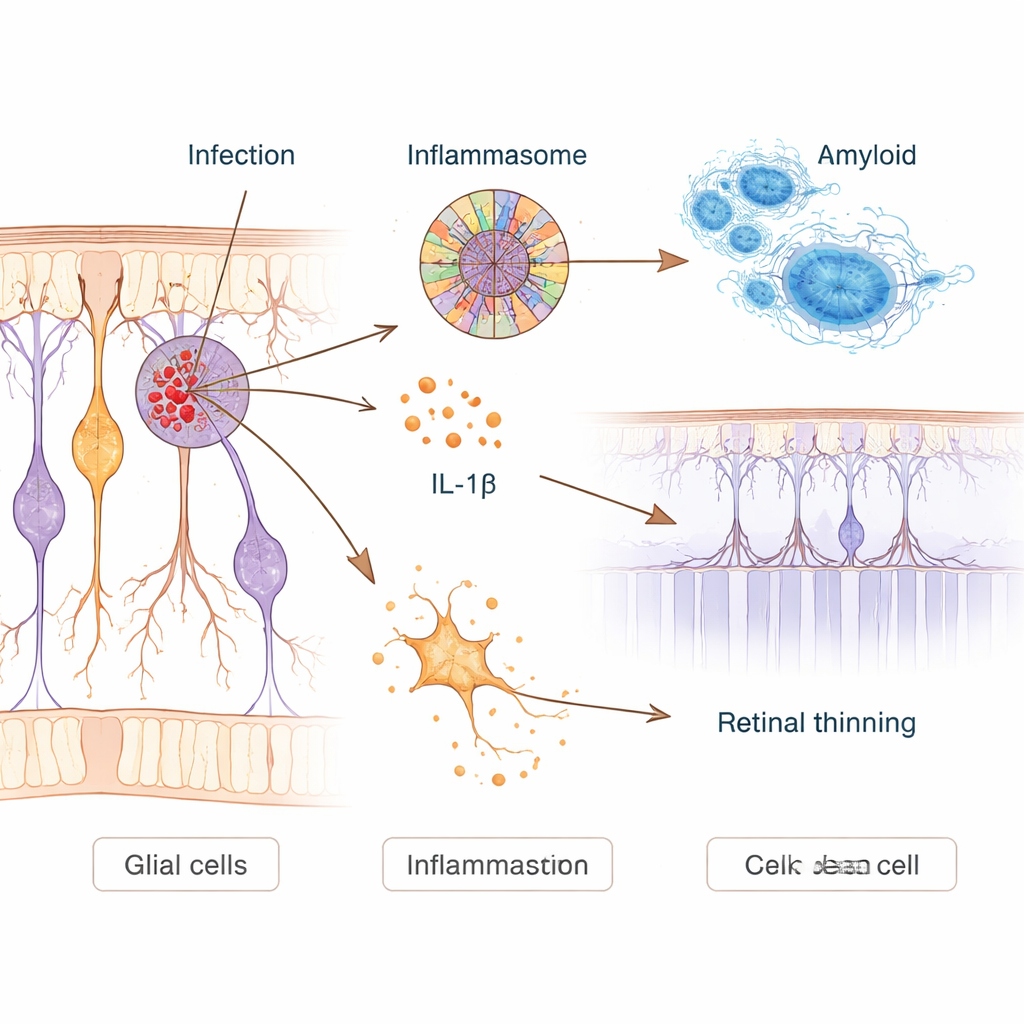
How infection may spark toxic inflammation
To probe cause and effect, the scientists infected human nerve‑like cells in the lab and Alzheimer‑model mice with Chlamydia pneumoniae. In cultured neurons, infection ramped up production of amyloid‑beta and switched on a molecular alarm system called the NLRP3 inflammasome. This system acts like a cellular tripwire: once triggered, it releases inflammatory messengers such as interleukin‑1β and can punch holes in cells, leading to an explosive form of cell death. Infected neurons showed clear signs of membrane damage and activation of these pathways. In mice, a single nasal dose of the bacterium was enough for it to reach the brain, where it increased amyloid plaques, activated immune cells, and worsened performance on memory and vision‑based maze tasks months later.
Retinal immune cells under pressure
In human eyes, the team mapped how infection interacted with the local support cells that normally protect neurons. Star‑shaped astrocytes and Müller cells, as well as microglia—the brain and retina’s resident “clean‑up” cells—clustered around infected sites. Chemical signatures of the NLRP3 inflammasome and its downstream death pathways were strongly elevated in retinas from people with Alzheimer’s, and were already rising in those with mild cognitive impairment. Many infected cells carried markers of both fiery “pyroptotic” death and more familiar apoptosis. Although more microglia were recruited in Alzheimer’s retinas, each bacterium appeared to be contacted or engulfed by fewer microglia than in healthy eyes, suggesting that the clean‑up system becomes overloaded or impaired as disease advances.
Using the eye to read brain health
Because the retina is accessible to high‑resolution imaging in living people, the researchers asked whether retinal measures related to Chlamydia pneumoniae could help predict Alzheimer’s status. They used machine‑learning models to combine retinal infection levels with markers such as amyloid‑beta deposits, inflammation, and local tissue thinning. Certain combinations—especially retinal bacteria together with amyloid—distinguished Alzheimer’s patients from controls with high accuracy and tracked with disease stage and cognitive test scores. While more work and larger studies are needed, the results suggest that a simple eye‑based test could someday reveal not only classic Alzheimer’s changes but also a hidden infectious component, opening the door to earlier use of antibiotics or drugs that dampen the inflammasome in people at risk.
What this could mean for patients
For non‑specialists, the take‑home message is that Alzheimer’s may not be driven solely by misbehaving proteins; long‑standing infections could act as fuel poured onto a slow fire. This study provides strong evidence that a common respiratory bacterium can settle in the retina and brain, stir up damaging inflammation, and worsen memory loss in susceptible individuals. If future clinical trials confirm that treating such infections or blocking the inflammasome pathway slows decline, routine retinal imaging and targeted therapies could become part of a more proactive, personalized approach to preventing or delaying dementia.
Citation: Gaire, B.P., Koronyo, Y., Vit, JP. et al. Identification of Chlamydia pneumoniae and NLRP3 inflammasome activation in Alzheimer’s disease retina. Nat Commun 17, 771 (2026). https://doi.org/10.1038/s41467-026-68580-4
Keywords: Alzheimer’s disease, retina, Chlamydia pneumoniae, neuroinflammation, amyloid-beta