Clear Sky Science · en
Large-scale serum N-glycomics tracks N-glycosylation dynamics in hepatocellular carcinoma progression and enables early diagnosis
Why Sugar Coatings on Blood Proteins Matter
Most people know that blood tests can reveal liver damage, but few realize that tiny sugar chains decorating blood proteins also change as liver disease worsens. This study shows that by reading these sugar “barcodes,” doctors may one day spot liver cancer, especially hepatocellular carcinoma (HCC), earlier and more accurately than with today’s standard tests.
Following Liver Disease from Health to Cancer
Hepatocellular carcinoma is one of the deadliest cancers worldwide and often grows silently in people with chronic hepatitis B, fatty liver disease or cirrhosis. Current screening tools, such as ultrasound and the blood marker alpha-fetoprotein (AFP), frequently miss early tumors. The researchers set out to track how the pattern of N-glycans—complex sugar structures attached to blood proteins—changes as the liver moves from health through chronic hepatitis and cirrhosis to full-blown cancer, and to test whether those changes could power better diagnostic tools.
A Nationwide Effort and a High-Throughput Sugar Scanner
The team analyzed blood samples from 1,074 people at three medical centers in China, spanning healthy volunteers, patients with chronic hepatitis B, patients with cirrhosis and patients with HCC. Using a high-throughput pipeline, they released, chemically tagged and enriched N-glycans from each serum sample, then measured them with a mass spectrometer—an instrument that weighs molecules with great precision. This produced detailed “glycan fingerprints” for 64 well-characterized sugar structures in each person, along with additional experiments on smaller subsets to map which specific proteins carried which sugars and how liver tissue genes controlling glycosylation were switched on or off.
How Liver Function and Cancer Shape the Sugar Landscape
By comparing glycan fingerprints with standard liver function tests, the scientists found that worsening liver health—regardless of the exact disease—was tightly linked to shifts in N-glycans. Poorer liver function went hand in hand with more highly branched sugar chains, more of a sugar called fucose, more “bisecting” sugar additions and less galactose and sialic acid at the tips of these chains. Some of these changes, long thought to be unique to cancer, turned out to reflect general liver failure. Yet the team also uncovered sugar features that were much more specific to HCC: large, highly branched, heavily fucosylated glycans that were elevated in cancer but not in cirrhosis, as well as distinct combinations of glycans that grouped HCC patients into three molecular subtypes with different liver function profiles and stages of disease.
Zooming In: Which Proteins and Genes Drive the Changes?
To understand what lies behind these altered sugar patterns, the researchers combined serum glycomics with glycoproteomics (which sugar is on which protein and at which site) and large public gene-expression datasets from liver tumors. They showed that a relatively small set of abundant blood proteins, such as acute-phase proteins involved in inflammation and clotting, contribute disproportionately to the overall glycan signal. At specific sites on these proteins, certain hybrid and highly branched glycans were strongly increased in liver disease, even when the total amount of the protein changed only modestly. In tumor tissue, many genes responsible for building, trimming and extending N-glycans were broadly turned up, especially those that create extra branches or add fucose, matching the complex sugar signatures seen in patient blood. Some of these glycosylation genes were also linked to how long patients survived and how quickly their disease progressed.
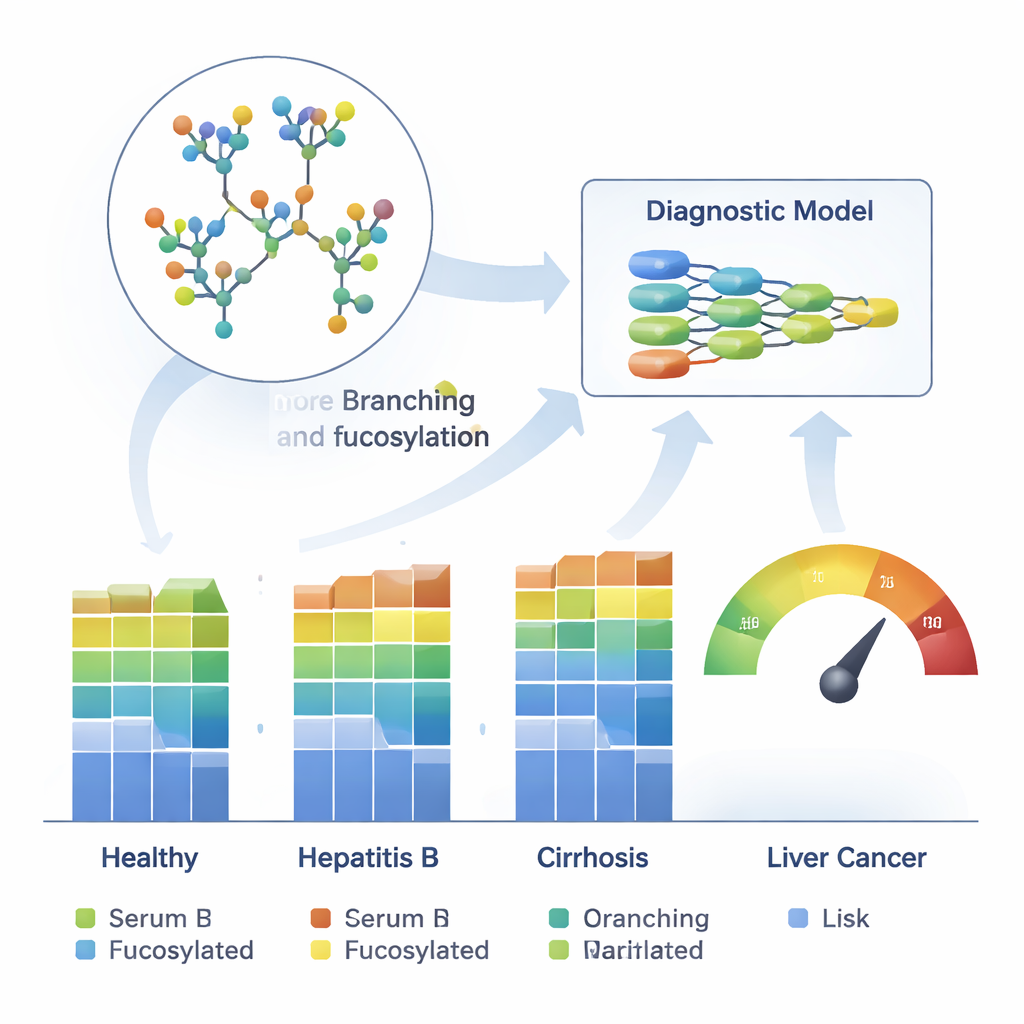
Teaching Machines to Read Sugar Codes
Finally, the team trained machine-learning models on 26 reliably measured glycans to distinguish HCC from healthy liver, chronic hepatitis B and cirrhosis. These models were developed on one large cohort and then tested on two independent patient groups. Across all tests, they achieved high accuracy, with areas under the receiver operating characteristic curve between 0.84 and 0.93. Crucially, they outperformed AFP and correctly flagged many cancers that AFP missed, including a substantial fraction of very early tumors. A small set of six glycans carried most of the diagnostic power, and a combined risk score from the main model also tracked how advanced a patient’s liver disease and cancer stage were.
What This Means for Patients
In everyday terms, this work suggests that the sugar decorations on common blood proteins provide a rich and sensitive readout of how sick the liver is and whether cancer has begun to develop. While more validation in diverse populations and long-term studies are needed, serum N-glycan profiling, paired with artificial intelligence, could complement imaging and existing blood tests to catch liver cancer sooner, especially in high-risk people with chronic liver disease.
Citation: Fu, B., Chen, J., Liu, X. et al. Large-scale serum N-glycomics tracks N-glycosylation dynamics in hepatocellular carcinoma progression and enables early diagnosis. Nat Commun 17, 1885 (2026). https://doi.org/10.1038/s41467-026-68579-x
Keywords: hepatocellular carcinoma, serum biomarkers, glycomics, early cancer detection, machine learning diagnostics