Clear Sky Science · en
Atroposelective interrupted CuAAC reaction using cyclic diaryliodoniums
Why this new chemistry matters
Chemists have long relied on a simple "click" reaction to snap molecular building blocks together quickly and cleanly, a trick that supports everything from drug discovery to self-healing materials. This article describes a clever twist on that classic click reaction that lets scientists build more complex, three-dimensional molecules with precise handedness—structures that are highly prized in modern medicines and advanced materials.
Giving a classic click reaction a new job
The starting point is the copper-catalyzed azide–alkyne cycloaddition, often simply called CuAAC. It joins two small components—an azide and an alkyne—into a five-membered ring called a triazole under mild conditions and with remarkable reliability. Traditionally, once copper helps form the triazole ring, the reaction ends there. In recent years, however, chemists have learned to "interrupt" this process, catching a fleeting copper–triazole intermediate with a third partner to build more elaborate products. Until now, those interruptions have not been able to control molecular handedness (chirality) in a general way, limiting their usefulness for making sophisticated, chiral molecules.
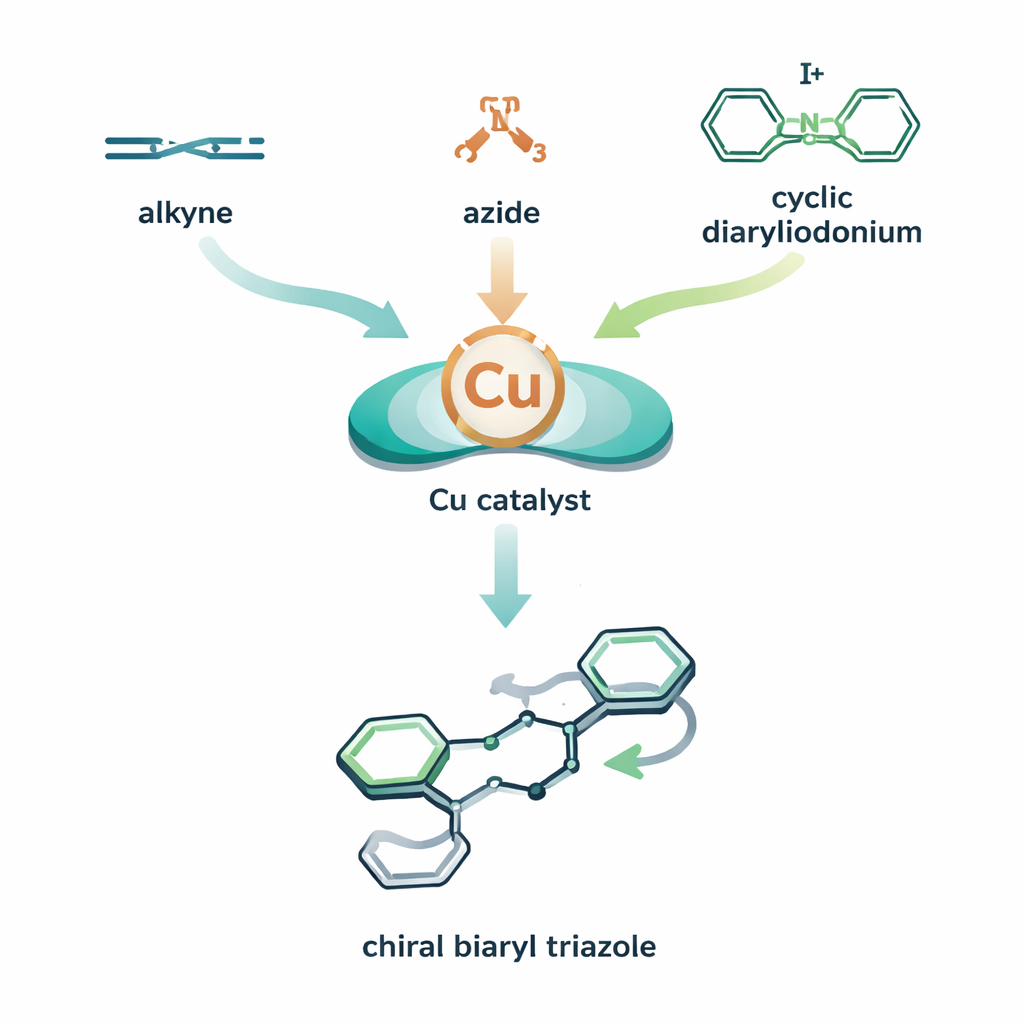
A three-piece molecular puzzle with a twist
The authors combine two copper-based reaction cycles into a single, orchestrated process. In their design, a copper catalyst first helps the alkyne and azide form a copper–triazole intermediate. Before this intermediate is neutralized, a third component—a ring-shaped, highly reactive iodine-containing molecule called a cyclic diaryliodonium—is brought in. Copper inserts into this ring and then opens it, stitching one of its aromatic rings onto the triazole. The result is a biaryl triazole: two ring systems joined around a bond that can act as a chiral axis, much like a propeller that can twist left or right. By pairing copper with a carefully chosen chiral ligand, the team biases the process so that one twist is formed far more than the other, achieving high atroposelectivity (control over which axial "hand" is made).
Testing how flexible and reliable the method is
To understand how general this reaction could be, the researchers systematically varied each of the three building blocks. They showed that many different alkynes, including those bearing electron-rich, electron-poor, and heteroaromatic rings, can participate while still giving good yields and strong preference for one chiral form. Certain bulky substitutions improved selectivity but could reduce yield, revealing a tradeoff between steric hindrance and efficiency. The azide partners worked best when they were derived from simple ester or amide groups or from benzylic positions, and a range of such azides delivered highly enriched chiral products. The cyclic diaryliodonium component could also be tuned: some substitutions preserved both yield and selectivity, while others, especially near the reactive iodine center, slowed the reaction or lowered its performance. Overall, the study delivered a broad collection of new atropisomeric biaryl triazoles accessible in one step.
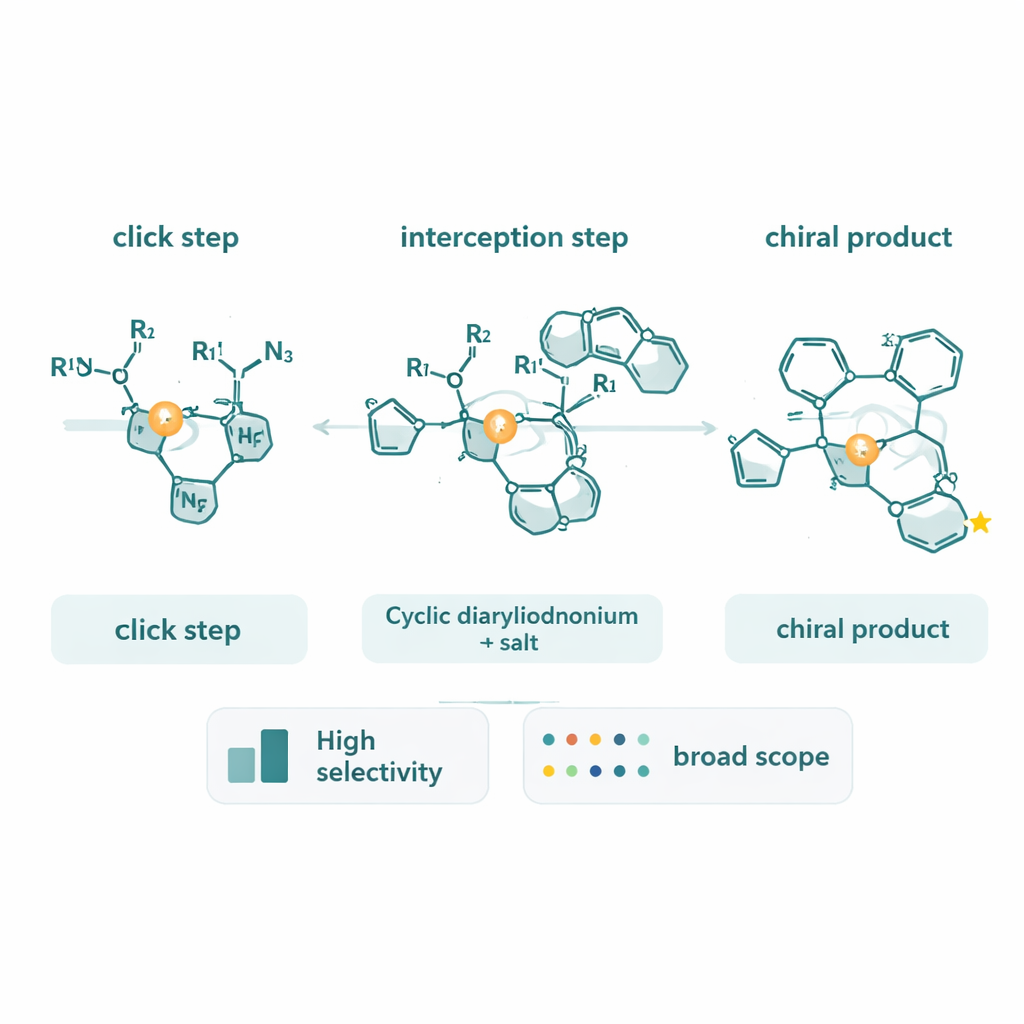
Peeking under the hood of the reaction
Beyond making new molecules, the team devoted substantial effort to finding out how the reaction actually works. Using deuterium-labeled alkynes, they observed an unusual inverse kinetic isotope effect, which points to a rate-limiting step where the alkyne carbon changes its bonding character rather than simply breaking a C–H bond. Calorimetry-based reaction progress analysis showed that the reaction rate depends strongly on the concentrations of the alkyne and azide, but only weakly on the cyclic diaryliodonium. These measurements support a picture in which forming and transforming the copper–triazole complex, and its oxidative addition into the diaryliodonium ring, together set the pace of the reaction. They also found that the chiral triazole products themselves can cling to copper and slow the catalyst down, hinting at ways to further optimize the system.
What this means going forward
In everyday terms, the researchers have taught a well-known click reaction a new trick: instead of simply snapping two pieces together, it now helps assemble three components into complex, propeller-like molecules while choosing almost exclusively one preferred twist. This provides a scalable route to chiral biaryl triazoles, structures that are valuable as potential drug molecules, catalysts, and functional materials. The mechanistic insights—especially how the key intermediate is intercepted and how the product can poison the catalyst—offer a roadmap for designing even more efficient and selective versions of this chemistry. For non-specialists, the key takeaway is that an already powerful molecular "Lego" tool has just become more versatile, enabling chemists to build more intricate and useful shapes with fine control over their three-dimensional form.
Citation: Li, Y., Yang, S., Duan, L. et al. Atroposelective interrupted CuAAC reaction using cyclic diaryliodoniums. Nat Commun 17, 944 (2026). https://doi.org/10.1038/s41467-026-68574-2
Keywords: click chemistry, copper catalysis, chiral biaryl, atropisomer, triazole