Clear Sky Science · en
A microphysiological human mini-bladder reveals urine-urothelium interplay in tissue resilience and UPEC recurrence in urinary tract infections
Why the State of Your Urine Matters
Most of us think of urine simply as waste, but its composition constantly changes with what we drink, eat, and how healthy we are. This study shows that those changes are not harmless background noise: they can directly influence how well the bladder defends itself against urinary tract infections (UTIs) and how easily infections come back after antibiotics. Using a tiny lab-grown “mini-bladder,” the researchers uncover how concentrated urine can quietly weaken bladder tissue and give disease-causing bacteria new ways to hide and survive treatment.
Building a Mini-Bladder in the Lab
To explore these hidden dynamics, the team engineered a thumb-sized human “mini-bladder” on a microfluidic chip. They seeded human bladder surface cells into a soft 3D scaffold and coaxed them to form several organized layers, similar to the real bladder lining. The topmost “umbrella” cells formed a tight, leak-resistant barrier, while deeper layers contained cells that can renew the tissue over time. The device also allowed urine-like liquid to flow through a central channel and the tissue to be gently stretched and relaxed, mimicking the filling and emptying cycles of a real bladder. Structural tests and high-resolution imaging confirmed that this mini-organ behaves much like its full-sized counterpart.
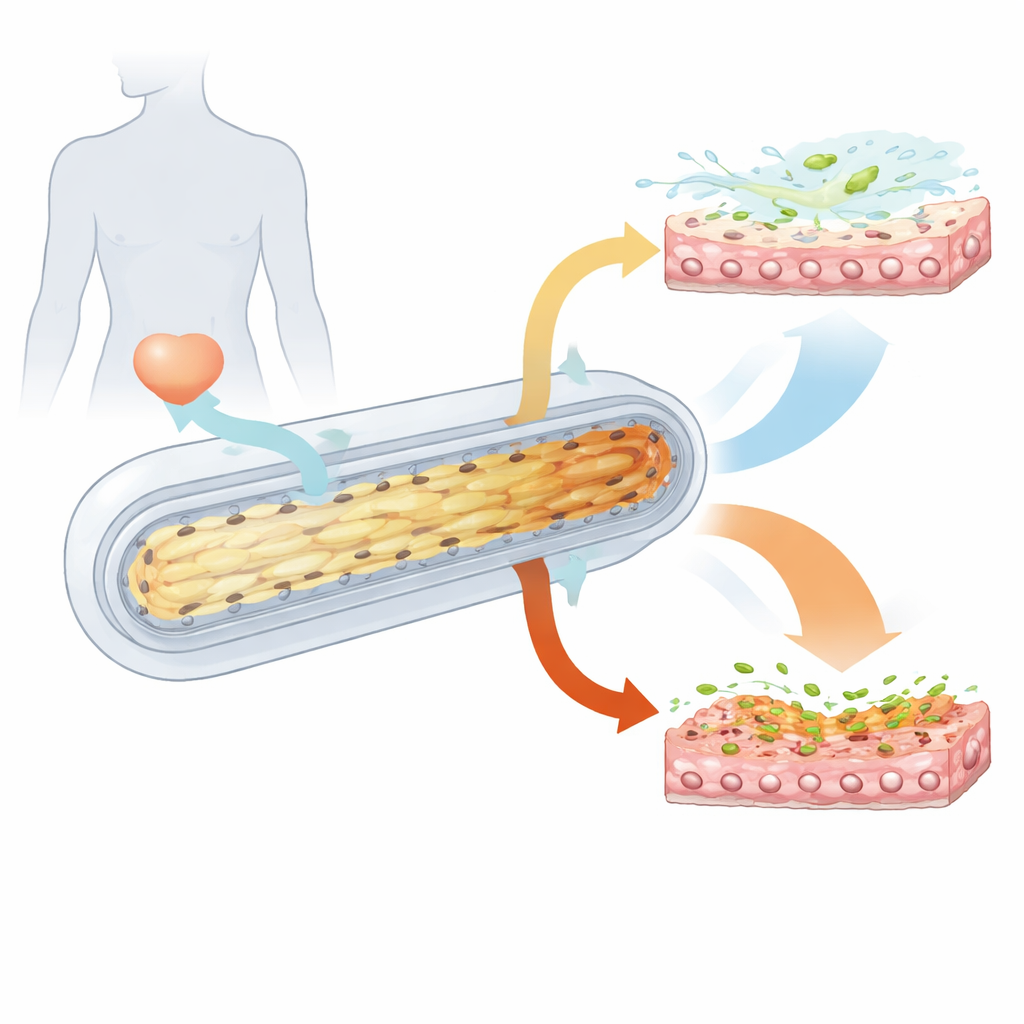
When Urine Turns Harsh
Human urine can be dilute or highly concentrated depending on hydration and health. The researchers created two synthetic urine mixtures: one “low-solute” and one “high-solute,” matching the lower and upper ends of natural human urine concentrations. When mini-bladders were bathed in high-solute urine for days, the tissue gradually deteriorated. More cells died, the total cell number dropped, and the normally tight seals between cells became leaky. Gene activity patterns showed reduced cell–cell adhesion, weaker tissue renewal, and dampened innate immune signaling. In contrast, low-solute urine supported cell growth, stronger tight junctions, and healthier differentiation of the bladder lining. In short, repeatedly exposing the bladder to very concentrated urine made the tissue structurally weaker and less ready to mount an early immune defense.
How Weak Tissue Favors Infection
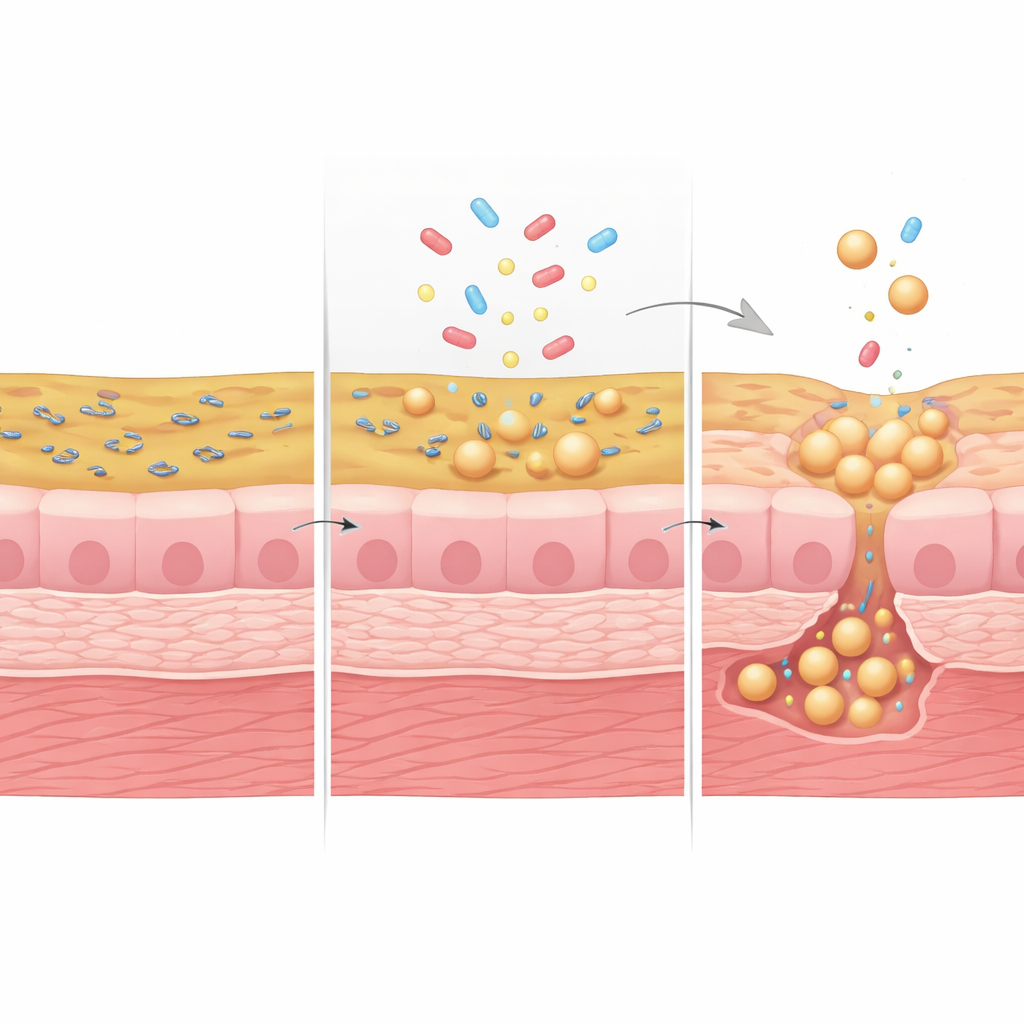
The team then infected the mini-bladders with uropathogenic Escherichia coli (UPEC), the main bacterial cause of UTIs, and periodically flushed the channel to imitate urination. After each “void,” many bacteria were washed out, but some clung to or invaded the bladder lining and gradually built up a hidden tissue-associated population. By blocking the bacteria’s main attachment mechanism with a sugar, D-mannose, the researchers reduced this embedded population and saw less tissue damage, showing that bacteria living on and within the tissue help reseed the urine between voids. When mini-bladders were pre-exposed to high-solute urine, more bacteria penetrated into the tissue, and standard antibiotics like ciprofloxacin were less able to clear them, even though free-floating bacteria in the urine were still removed effectively.
Hidden Survivors After Antibiotics
The story became even more striking with fosfomycin, a commonly prescribed antibiotic that targets bacterial cell walls. In high-solute urine, fosfomycin did a poor job of reducing the bacterial burden, and free bacteria often changed from rod-shaped into large, fragile, cell wall–deficient spheres. Similar spherical forms have been seen in urine from patients with recurrent UTIs. Using advanced 3D electron microscopy combined with fluorescence imaging, the researchers found that such spherical bacteria were not only floating in urine but also nestled between and beneath bladder cells deep within the mini-bladder wall. These sheltered forms survived treatment and later contributed to bacterial regrowth, helping explain why infections can flare up again after symptoms briefly improve.
What This Means for Patients
This work reveals that the bladder’s environment is not just a backdrop but an active player in infection and treatment outcomes. Concentrated urine weakens the bladder lining, blunts its immune defenses, and allows bacteria to invade deeper and adopt special cell wall–deficient forms that are hard for both antibiotics and immune cells to eliminate. The new mini-bladder platform shows how urine composition, bladder tissue health, and bacterial behavior are tightly linked. For people prone to recurrent UTIs, factors that make urine more concentrated—such as low fluid intake or certain medical conditions—may quietly increase risk by undermining tissue resilience and fostering hidden reservoirs of bacteria. Understanding and eventually adjusting this microenvironment, alongside better antibiotic strategies, could be key to breaking the cycle of repeat infections.
Citation: Paduthol, G., Nikolaev, M., Sharma, K. et al. A microphysiological human mini-bladder reveals urine-urothelium interplay in tissue resilience and UPEC recurrence in urinary tract infections. Nat Commun 17, 2322 (2026). https://doi.org/10.1038/s41467-026-68573-3
Keywords: urinary tract infection, bladder organoid, uropathogenic E. coli, antibiotic tolerance, urine concentration