Clear Sky Science · en
Biomarkers of response to neoadjuvant palbociclib plus anastrozole in endocrine-resistant estrogen receptor-positive/HER2-negative breast cancer: a phase 2 trial
Why this research matters to patients and families
Many women with a common form of breast cancer initially respond well to hormone-blocking drugs, only to see their tumors become resistant and start growing again. This study asks a pressing question: can adding a newer, cell-cycle–blocking drug to standard hormone therapy help in these hard-to-treat cases, and can we find warning signs in the tumor that predict who will, and will not, benefit?
A closer look at stubborn breast cancers
The trial focused on estrogen receptor–positive, HER2‑negative breast cancers, the most frequent subtype. All 34 patients had already been on standard pre-surgery hormone therapy (an aromatase inhibitor) but still showed active tumor cell growth, meaning the cancer was considered “endocrine‑resistant.” Researchers then treated these patients before surgery with a combination of the hormone drug anastrozole and the cell‑cycle blocker palbociclib. They measured how fast tumor cells were dividing using a marker called Ki67. If Ki67 dropped to very low levels after two weeks, the tumor was said to have reached “complete cell‑cycle arrest,” indicating that cancer cell division had been effectively shut down.
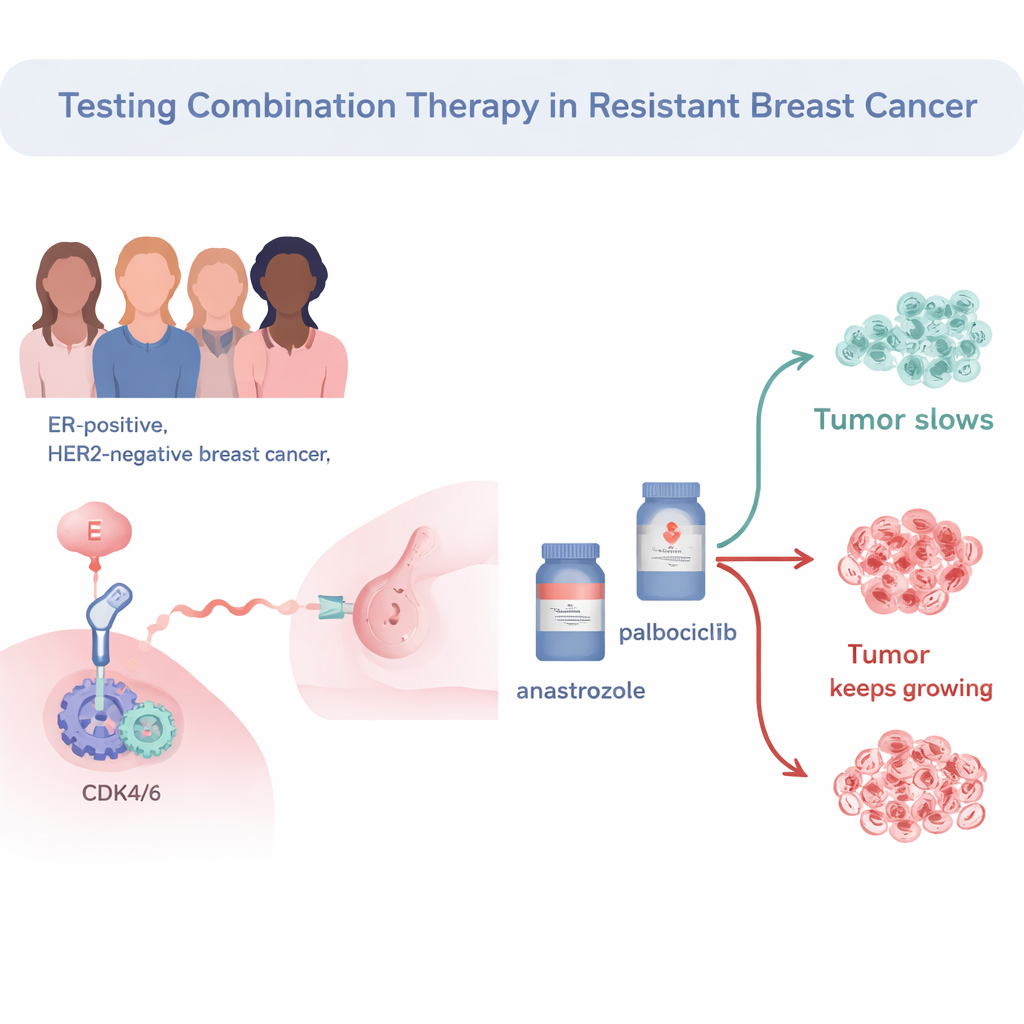
Who responded to the drug combination?
Among the 33 patients whose tumors could be fully evaluated, 57.6% reached complete cell‑cycle arrest after just two weeks of the combined treatment. When a more modest threshold was used—simply classifying tumors as “sensitive” if Ki67 fell below 10%—about two‑thirds of tumors were deemed responsive. Patients whose tumors remained highly active on Ki67 tended to have more aggressive cancers: higher grade, larger size, and higher starting Ki67 levels. They were also more likely to fall into so‑called “non‑luminal” molecular subtypes, which generally behave more aggressively than classic hormone‑driven “luminal A” tumors.
What makes some tumors resist treatment?
The team went beyond simple response rates and deeply analyzed tumor samples using DNA sequencing, RNA sequencing, and protein profiling. Resistant tumors were not defined by a single mutation; instead, they showed a pattern of “hyperactive wiring.” Compared with sensitive tumors, resistant cancers had dampened estrogen‑receptor signaling but stronger activity in pathways that drive cell division and growth, such as cell‑cycle genes, the mTOR growth pathway, and several inflammatory and interferon‑related networks. They also showed higher levels of immune “brakes,” known as immune checkpoints, including genes like IDO1 and PD‑L1 that can help tumors evade the body’s defenses.
From pathways to potential new treatments
To test whether these overactive pathways might be used against the cancer, the researchers created laboratory cell models that had become resistant to CDK4/6 inhibitors like palbociclib. These resistant cells showed the same signature of boosted cell‑cycle and interferon/inflammatory signaling seen in patients’ tumors. When exposed to drugs that block JAK‑STAT signaling—a key route downstream of interferon—one medicine, pacritinib, was particularly effective at slowing the growth of resistant cells and patient‑derived tumor organoids. This suggests that pairing CDK4/6 inhibitors with certain JAK‑targeting drugs could be a promising strategy for patients whose cancers have outsmarted current combinations.
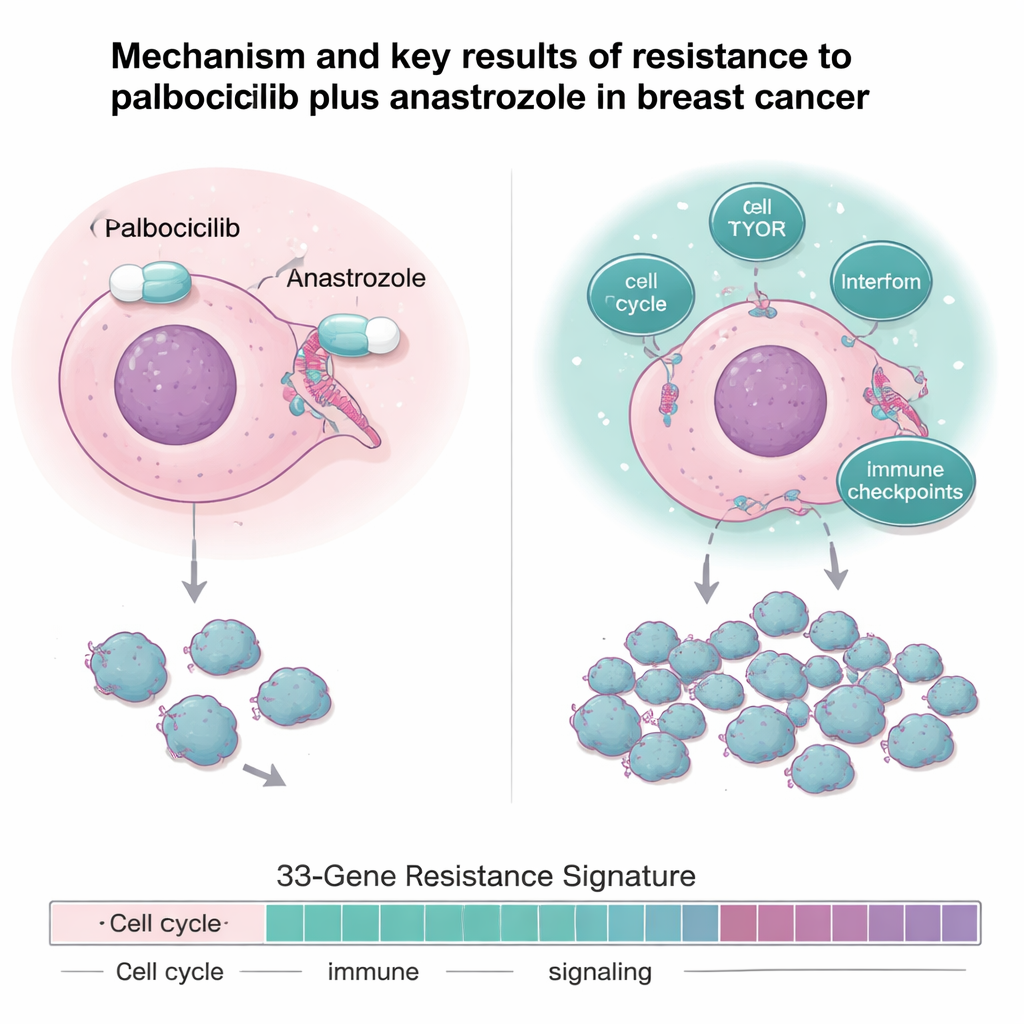
A gene fingerprint that predicts outcome
By comparing sensitive and resistant tumors across two related trials, the investigators built a 33‑gene “resistance signature” capturing genes involved in cell division, DNA repair, growth signaling, metabolism, and immune/inflammatory responses. This gene fingerprint was very accurate at distinguishing sensitive from resistant tumors in the trial. Importantly, when the same 33‑gene pattern was applied to an independent group of 151 patients with metastatic breast cancer treated in routine practice with CDK4/6 inhibitors plus hormone therapy, those whose cancers scored high for the resistance signature had shorter time before their disease progressed and shorter overall survival. In other words, this gene pattern measured before treatment could flag patients less likely to benefit for long from standard CDK4/6‑based regimens.
What this means for future care
For people facing hormone‑resistant estrogen receptor–positive breast cancer, this study offers both reassurance and a roadmap. It shows that adding palbociclib to anastrozole can still shut down tumor growth in over half of resistant cases. At the same time, it reveals that some tumors rely on alternative growth and immune‑related pathways, making them tougher to control. The newly defined 33‑gene signature and the discovery that JAK‑targeting drugs like pacritinib can curb resistant cells point toward more personalized treatment: identifying those unlikely to respond to standard combinations and steering them sooner toward trials testing new drug partners that directly target the cancer’s backup survival routes.
Citation: Kong, T., Mabry, A., Highkin, M. et al. Biomarkers of response to neoadjuvant palbociclib plus anastrozole in endocrine-resistant estrogen receptor-positive/HER2-negative breast cancer: a phase 2 trial. Nat Commun 17, 949 (2026). https://doi.org/10.1038/s41467-026-68570-6
Keywords: ER-positive breast cancer, CDK4/6 inhibitors, endocrine resistance, tumor biomarkers, palbociclib anastrozole