Clear Sky Science · en
Multimodal analysis disentangles the genetic and microbial associations between inflammatory bowel disease and other immune-mediated diseases across a harmonized population framework
Why some immune diseases travel in packs
Many people with inflammatory bowel disease (IBD) – long‑lasting inflammation of the gut – also develop other immune‑related illnesses, such as arthritis, psoriasis, or multiple sclerosis. Doctors see these overlaps every day, but it has been unclear how much they are driven by shared genes, by our everyday environment, or by the microbes that live in our intestines. This study uses nationwide data from Denmark, large genetic datasets, and gut microbiome profiles to untangle why certain immune diseases tend to cluster together and why Crohn’s disease and ulcerative colitis, the two main forms of IBD, do not always behave the same way.
Following disease patterns through families
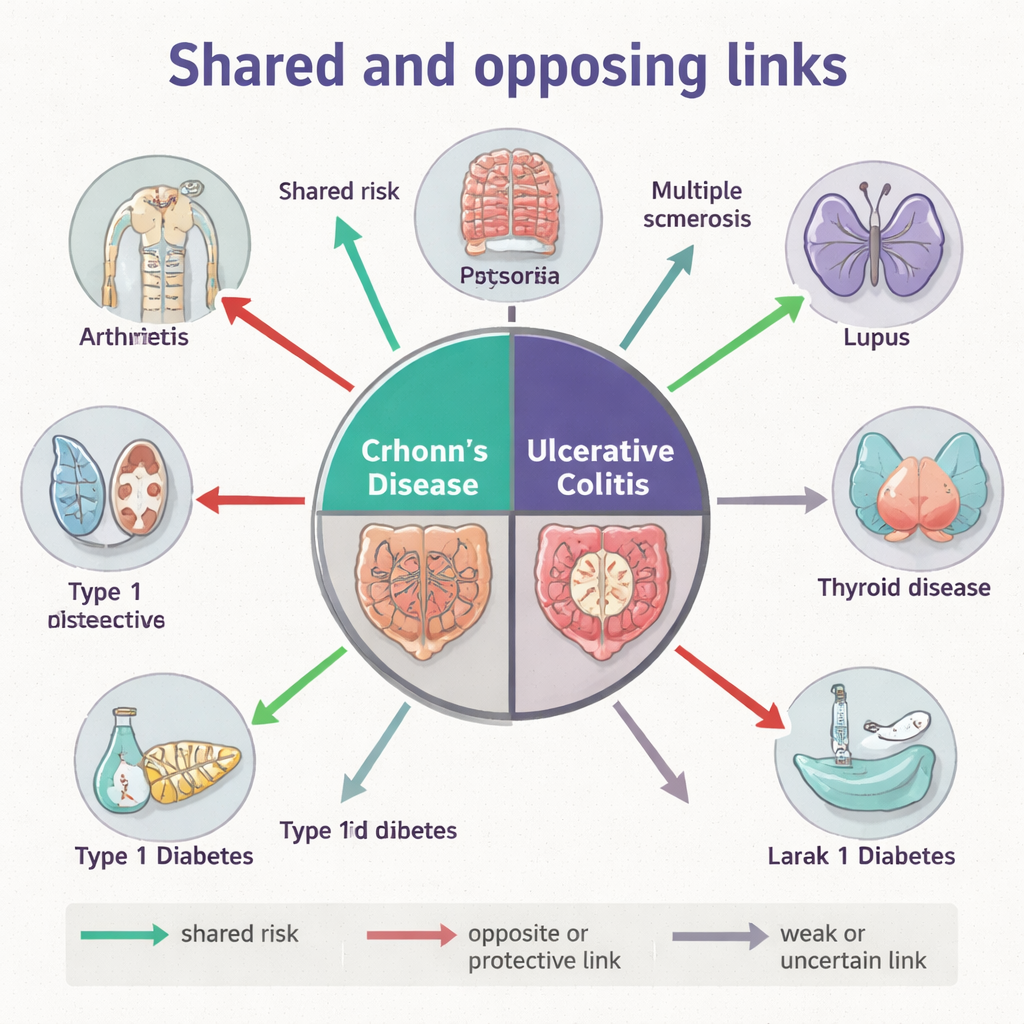
The researchers began by mapping out a gigantic family tree of the modern Danish population using civil registration data that link parents, children, and siblings. Focusing on more than two and a half million full siblings born between 1910 and 2010, they looked at how often Crohn’s disease or ulcerative colitis occurred together with 23 other immune‑mediated inflammatory diseases, including rheumatoid arthritis, psoriasis, type 1 diabetes, multiple sclerosis, and lupus. They found that both Crohn’s and ulcerative colitis run strongly in families, but they do so with different “companions.” For example, both bowel diseases were clearly linked within families to psoriasis, psoriatic arthritis, rheumatoid arthritis, and ankylosing spondylitis, while ulcerative colitis – but not Crohn’s disease – showed notable familial ties to conditions like sarcoidosis and lupus.
Peering into shared genes
Family patterns could reflect shared genes, shared environment, or both. To separate these influences, the team turned to genome‑wide association studies, which scan hundreds of thousands of genetic markers across the genome in large patient groups. They quantified how much of the genetic risk for Crohn’s and ulcerative colitis is shared with other immune diseases. As expected, the two bowel conditions shared a strong genetic backbone. Beyond this core, however, their genetic friendships diverged. Crohn’s disease showed positive genetic overlap with asthma, psoriasis, psoriatic arthritis, and rheumatoid arthritis, whereas ulcerative colitis shared more genetic risk with type 1 diabetes and primary biliary cholangitis, a liver disorder. Strikingly, both IBD types showed negative genetic correlations with multiple sclerosis, and ulcerative colitis also with lupus, suggesting that some genetic variants that raise the chance of gut inflammation may actually protect against certain brain or systemic immune diseases.
Listening to the gut’s microbial “accent”
Genes are only part of the story. The authors also assembled fecal microbiome data from thousands of people around the world, re‑processing all 16S rRNA sequencing reads in a harmonized way so that bacterial strains could be compared across studies. For seven immune diseases, they built “microbiota disease profiles” – statistical summaries of how hundreds of bacterial variants differ between patients and healthy controls. Comparing these profiles revealed that Crohn’s and ulcerative colitis share broadly similar microbial disruptions, and both overlap strongly with rheumatoid arthritis and celiac disease. Yet ulcerative colitis also showed microbiome similarities with multiple sclerosis and Graves’ disease even when the genetic overlap with those conditions was weak or negative. This mismatch between genes and microbes points to a powerful role for environment and lifestyle – including medications, diet, and antibiotic exposure – in shaping common microbial signatures across seemingly distant immune illnesses.
What this means for patients and doctors
Taken together, the family, genetic, and microbial layers reveal that Crohn’s disease and ulcerative colitis are not interchangeable labels but distinct members of the immune‑disease family. Some conditions, such as psoriasis and certain forms of arthritis, show consistent connections across all three layers, hinting at deeply shared biology. Others, like multiple sclerosis and lupus, display an almost “push‑pull” relationship, where gene effects and microbial patterns point in opposite directions. These nuances help explain why people with IBD can experience very different constellations of other immune problems and why the same drug may help in one disease but backfire in another.
Looking ahead: toward more tailored care
For a layperson, the key message is that immune diseases do not arise in isolation. This study shows that who gets which combination of conditions depends on a shifting mix of inherited genes, family background, and the microscopic life in the gut. It argues that Crohn’s disease and ulcerative colitis should be studied and treated as related but distinct entities, with careful attention to each patient’s broader immune profile and family history. In the future, combining genetic risk scores with microbiome signatures could help doctors better predict which additional immune conditions a person with IBD might face and design prevention and treatment strategies that are truly personalized rather than one‑size‑fits‑all.
Citation: Vestergaard, M.V., Alfaro-Núñez, A., Sazonovs, A. et al. Multimodal analysis disentangles the genetic and microbial associations between inflammatory bowel disease and other immune-mediated diseases across a harmonized population framework. Nat Commun 17, 1849 (2026). https://doi.org/10.1038/s41467-026-68564-4
Keywords: inflammatory bowel disease, autoimmune comorbidity, gut microbiome, genetic correlation, Crohn’s disease and ulcerative colitis