Clear Sky Science · en
Glass formation in hybrid metal halides via breaking molecular rotational order
Why this strange kind of glass matters
Glass is usually thought of as a frozen liquid made from sand, but researchers are now creating glasses from mixtures of metals and organic molecules that glow under X‑rays and can be molded like plastics. This article explores a new way to design such glasses by deliberately disrupting how the molecules can rotate, offering a recipe for better radiation detectors, optical parts, and other advanced technologies.
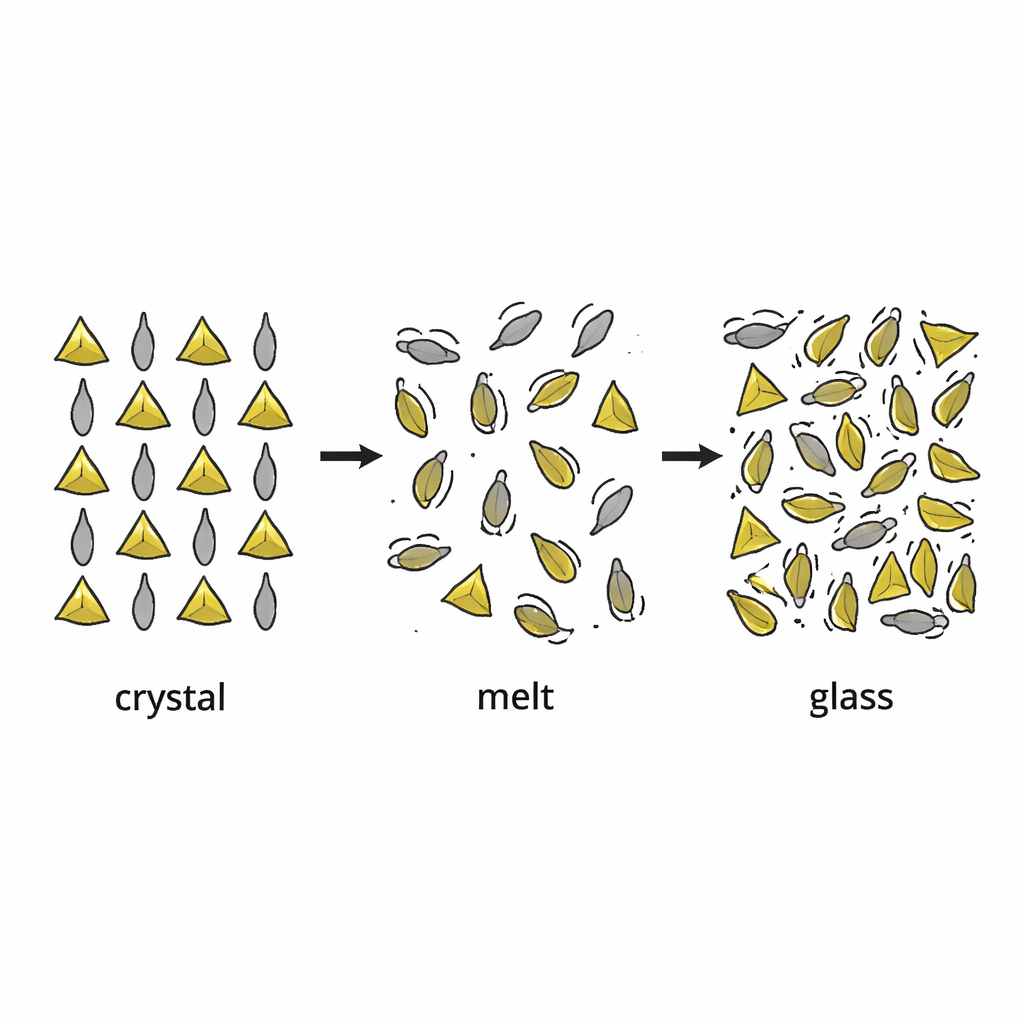
From tidy crystals to frozen disorder
In a crystal, atoms and molecules line up in a precise, repeating pattern. In a glass, that long-range order is lost: the building blocks are stuck in a jumble, like people frozen mid‑dance. The authors focus on a family of materials called zero‑dimensional hybrid metal halides, built from compact manganese–bromine units and larger organic molecules carrying positive charge. These ingredients can form either well‑ordered crystals or glassy solids, depending on how they are cooled from the melt. The key idea is that, as the liquid cools, the organic molecules slow down and eventually become locked into place with whatever orientations they happen to have, creating a disordered but stable structure.
Shaping molecules to control glass formation
The team designed nine related compounds by changing the shape and electrical surface of the organic phosphonium molecules. Swapping one of the ring‑shaped groups for small chains or different benzyl groups slightly distorts the molecule and alters how easily it can rotate and pack. When the researchers melted and rapidly cooled these materials, some compositions stayed crystalline, while others turned into true glasses that showed no sharp diffraction peaks—clear evidence that their regular long‑range order had vanished. Computer models confirmed that the manganese–bromine units keep their basic geometry, but the organic molecules adopt a wide variety of orientations, signaling strong rotational disorder in the glass.
Measuring motion that you cannot see
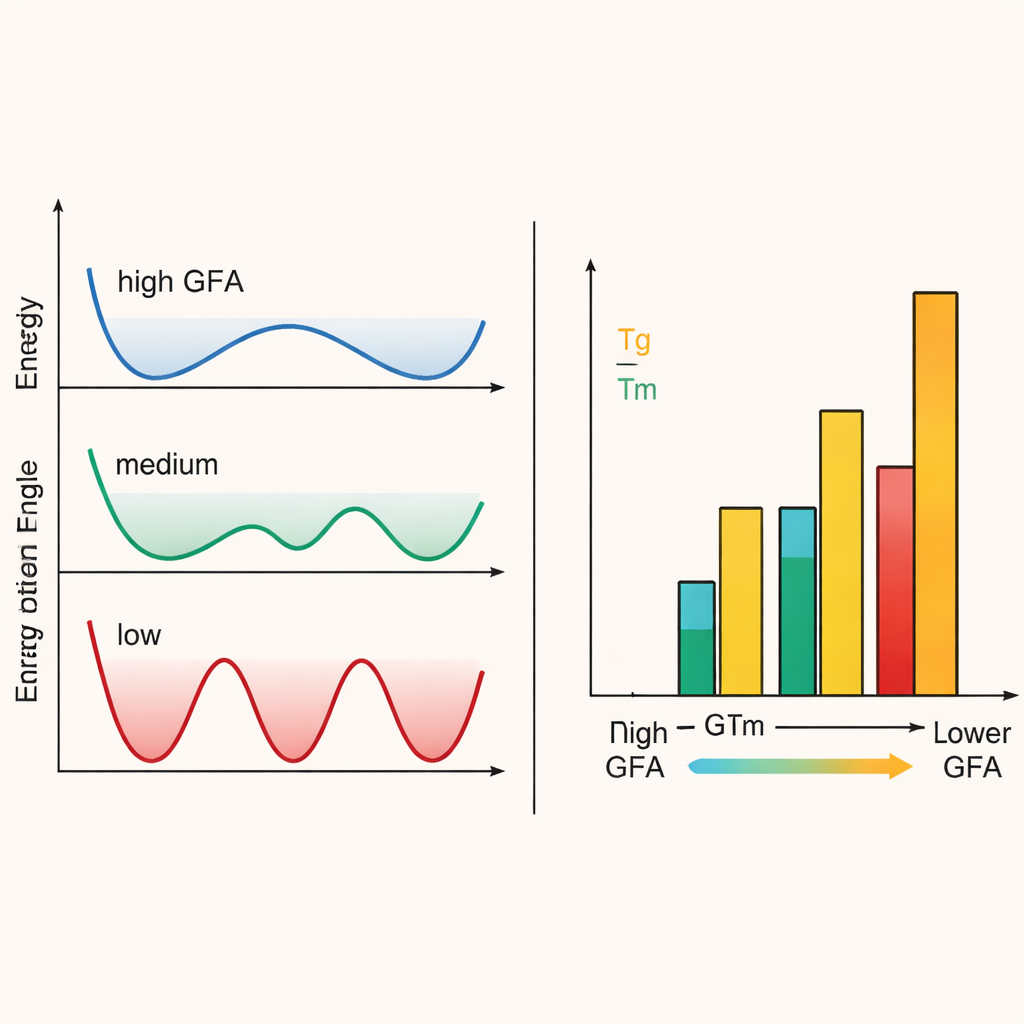
To connect this hidden motion to glass‑forming ability, the authors used both lab experiments and large‑scale simulations. Differential scanning calorimetry revealed each material’s melting temperature and glass transition temperature, whose ratio is a standard indicator of how easily a glass can form. They also built mathematical measures of how aligned the molecules are and how quickly they change orientation. Systems in which the organic molecules could explore many orientations, facing weaker and more uniform electrical interactions, showed flatter “energy landscapes,” shorter rotational correlation times, and higher glass‑forming ability. In contrast, more polar or elongated molecules faced deeper rotational energy wells and stronger locking‑in with neighbors, making it harder to avoid crystallization during cooling.
Glowing glasses for X‑ray detection
Beyond structure, these hybrid glasses have striking optical behavior. When excited by ultraviolet light, both crystals and glasses emit green light from the manganese centers, but the glassy versions show broader, slightly red‑shifted emission and shorter lifetimes, signatures of a more disordered environment. Under X‑ray illumination, the glasses act as efficient scintillators: they convert weak X‑ray doses into visible light with high sensitivity and good stability over many cycles. One composition in particular detects extremely low X‑ray doses, and another can be drawn into thin fibers that produce sharp X‑ray images, illustrating the practical value of controlling molecular motion during glass formation.
A design rule for tomorrow’s glasses
For non‑specialists, the core message is simple: by carefully adjusting how freely molecules can rotate and how evenly their charges are distributed, scientists can steer a material toward becoming a glass with chosen properties instead of a crystal. In these hybrid metal halides, molecules with compact shapes and gentle, uniform electrical surfaces give rise to highly formable glasses with lower working temperatures, while more polar or uneven molecules favor rigid, higher‑temperature glasses that are harder to form. This strategy—tuning rotational disorder rather than just composition—offers a powerful new guideline for designing next‑generation glasses and amorphous materials, from metal‑based glasses to other hybrid solids used in optics, electronics, and radiation detection.
Citation: Li, ZY., Feng, R., Li, ZG. et al. Glass formation in hybrid metal halides via breaking molecular rotational order. Nat Commun 17, 1850 (2026). https://doi.org/10.1038/s41467-026-68563-5
Keywords: glass formation, hybrid metal halides, molecular rotation, scintillator materials, amorphous solids