Clear Sky Science · en
Impact and cost-effectiveness of doxycycline post-exposure prophylaxis in Australian men who have sex with men
Why this matters for everyday sexual health
Sexually transmitted infections like chlamydia, gonorrhoea, and syphilis are rising in many countries, especially among gay, bisexual, and other men who have sex with men. A simple antibiotic pill taken soon after sex, called doxycycline post-exposure prophylaxis (doxy-PEP), has emerged as a promising way to prevent some of these infections. But using antibiotics in this way could also fuel drug-resistant “superbug” strains. This study asks a practical question with big public health implications: how can doxy-PEP be used in Australia to prevent the most infections at the lowest cost, while doing the least harm in terms of antibiotic resistance?
A pill after sex as a new prevention tool
Doxycycline is a long-used, inexpensive antibiotic. Recent clinical trials have shown that when certain men and transgender women take a dose within 72 hours after condomless sex, they sharply cut their chances of getting chlamydia and syphilis, and modestly reduce gonorrhoea infections. Australian experts already suggest that doxy-PEP could be offered to people at high risk of syphilis. However, there is a crucial catch: each extra dose of antibiotics gives bacteria another chance to evolve resistance. If gonorrhoea becomes less sensitive to doxycycline and related drugs, it could become harder and more expensive to treat. Policymakers therefore need to know not just “does it work?” but “who should get it, and what does that mean for resistance and cost?”
Simulating real-world sexual networks
To explore these trade-offs, the researchers built a detailed computer model of 10,000 Australian men who have sex with men and followed this virtual population from 2012 to 2034. The model captures how people form and end regular and casual sexual partnerships, how often they have sex, how reliably they use condoms, and how often they get tested for infections. It also tracks the spread of gonorrhoea, chlamydia, and syphilis and how gonorrhoea strains gain resistance to antibiotics over time. After making sure the model reproduced past Australian surveillance data for infection rates and resistance patterns, the team simulated five ways of offering doxy-PEP and compared these with a future in which nobody used it.
Who gets the pills: five different strategies
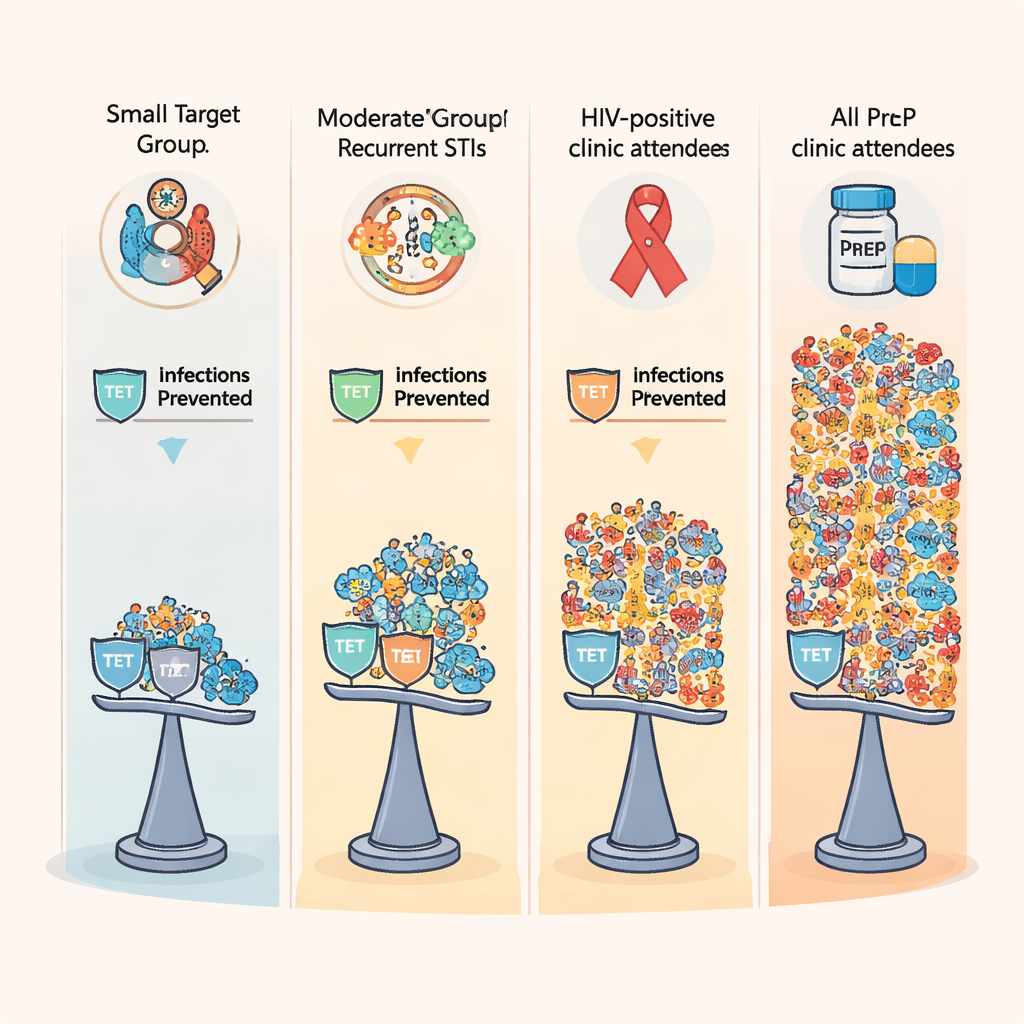
The five strategies ranged from highly targeted to very broad. One offered doxy-PEP only to men with a recent syphilis diagnosis. Two others focused on men who had multiple STI diagnoses within either six or twelve months, regardless of which infection. A fourth strategy offered doxy-PEP to all HIV-positive men attending sexual health clinics, and the broadest offered it to all clinic attendees using HIV pre-exposure prophylaxis (PrEP). For each strategy the model tracked how many antibiotic courses would be prescribed, how many infections would be prevented, how patterns of gonorrhoea resistance would shift, and how much money the health system would save or spend.
Fewer infections, more resistance
Across the board, doxy-PEP reduced total numbers of gonorrhoea, chlamydia, and syphilis infections over ten years, cutting overall STI cases by 13 to nearly 48 percent depending on the strategy. The broad PrEP-based approach prevented the most infections and could help meet World Health Organization targets for reducing chlamydia and syphilis. But there was a downside: all strategies increased the share of gonorrhoea that was highly resistant to tetracycline, a stand-in measure for doxycycline resistance. In the model, even without doxy-PEP, about three-quarters of new gonorrhoea infections were projected to be highly tetracycline-resistant by 2034. With broad doxy-PEP use, that figure climbed even higher, reaching about 97 percent when offered to all PrEP users. The narrower syphilis-focused strategy still raised resistance, but to a lesser extent, and resulted in the smallest increase in highly resistant gonorrhoea overall.
Balancing health benefits and costs
The study also weighed direct medical costs for testing and treatment and estimated quality of life lost due to infections. All five doxy-PEP strategies saved money compared with not using doxy-PEP at all, largely because preventing infections avoided clinic visits, tests, and treatments. Yet cost-effectiveness varied sharply. Targeting only men with a syphilis diagnosis gave the best “value for pill”: each prescription averted more than seven infections and produced the highest benefit–cost ratio, meaning every dollar spent on doxy-PEP returned many times that amount in savings. In contrast, the broad PrEP-based strategy, while preventing the most infections, required far more prescriptions per infection averted and had the lowest economic return. A combined ranking of infection reduction, resistance impact, and economic value consistently placed the syphilis-focused approach at or near the top.
What this means for sexual health policy
For a lay audience, the takeaway is that doxy-PEP can be a powerful, money-saving tool to cut syphilis and chlamydia among Australian men who have sex with men, but it is not a magic bullet and comes with resistance risks, especially for gonorrhoea. The research suggests that offering doxy-PEP mainly to people who have already had syphilis strikes the best balance: it protects those at highest risk, reduces serious infections, saves health-care costs, and slows (but does not stop) the rise of resistant gonorrhoea compared with broader rollouts. Policymakers considering doxy-PEP are therefore encouraged to pair it with strong resistance surveillance, ongoing STI screening, and investment in alternative measures such as vaccines, rather than relying on antibiotics alone.
Citation: Lai, H., Ong, J.J., Traeger, M.W. et al. Impact and cost-effectiveness of doxycycline post-exposure prophylaxis in Australian men who have sex with men. Nat Commun 17, 1846 (2026). https://doi.org/10.1038/s41467-026-68561-7
Keywords: doxycycline post-exposure prophylaxis, sexually transmitted infections, men who have sex with men, antimicrobial resistance, cost-effectiveness