Clear Sky Science · en
Microbial activation of the GLP-2R mitigates gastrointestinal inflammation
Why Gut-Friendly Protein Matters
As the world searches for sustainable ways to feed a growing population, scientists are looking beyond farms and fields to giant steel tanks growing microbes for food. This study explores whether one such microbe-based protein can do more than simply nourish us. The researchers asked: can a protein made from harmless bacteria both meet dietary needs and actively protect the gut from inflammation, a core problem in conditions like inflammatory bowel disease and chemotherapy-induced gut damage?
A New Kind of Protein From Microbes
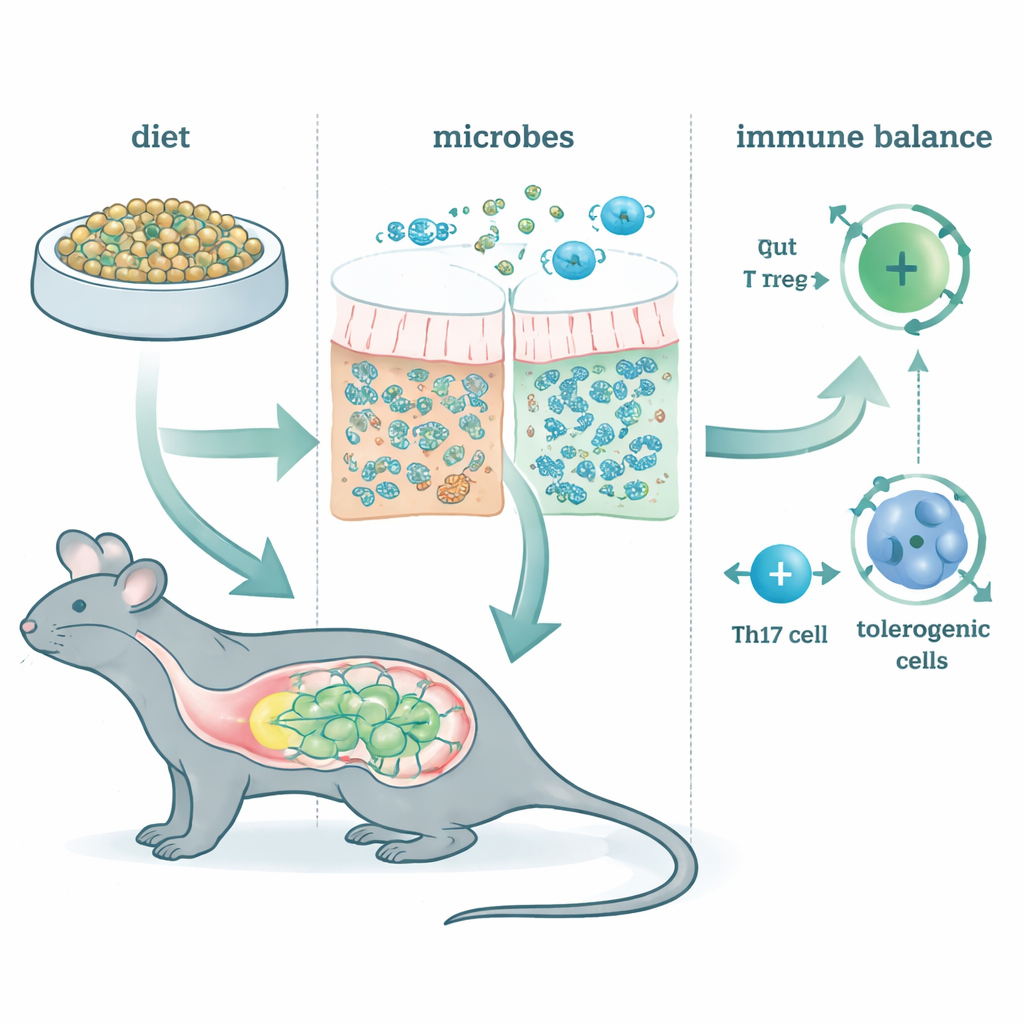
The protein at the center of this work comes from a soil bacterium called Methylococcus capsulatus Bath (McB). Instead of being eaten as live bacteria, McB is processed into a microbial lysate—essentially a powdered mix of broken bacterial cells—commercially produced under the name FeedKind®. Mice were fed diets in which this lysate supplied most of the protein, in either a simple or a more human-like "complex" diet. The team then tracked how this diet changed the mix of gut microbes, the immune cells that patrol the intestine, and how well the gut withstood different kinds of injury.
Reshaping the Microbial Neighborhood
Feeding McB rapidly and persistently altered the gut microbiota, regardless of which background diet the animals ate. Only a tiny fraction of McB itself ever appeared in the feces, meaning the material was almost completely used up in the gut. Instead, a handful of bacterial species from families such as Lachnospiraceae and Bacteroidaceae flourished. Genetic analyses showed that these communities were enriched for fermentation pathways that produce short-chain fatty acids—small molecules already known to nourish gut cells and influence immunity. In other words, McB acted less like a conventional protein and more like a targeted fuel that reshapes what resident microbes do.
Teaching the Immune System to Tolerate
The authors next examined key T cell types that help decide whether the gut reacts calmly or aggressively to what passes through it. One group, peripherally induced regulatory T cells (pTregs), helps prevent overreaction to food and friendly microbes. Another group, Th17 cells, can either drive damaging inflammation or help maintain a healthy barrier, depending on their “mood.” McB feeding strongly increased pTregs along the entire intestine, even when most gut microbes were wiped out with antibiotics. By contrast, McB’s effects on Th17 cells depended on the presence and normal function of gut bacteria. In microbially intact mice, McB nudged Th17 cells toward a more calming, IL‑10–producing, less inflammatory state. When fermentation was blocked, this tolerogenic shift vanished, underscoring that McB works partly by rerouting microbe–immune conversations.
Guarding the Gut During Injury
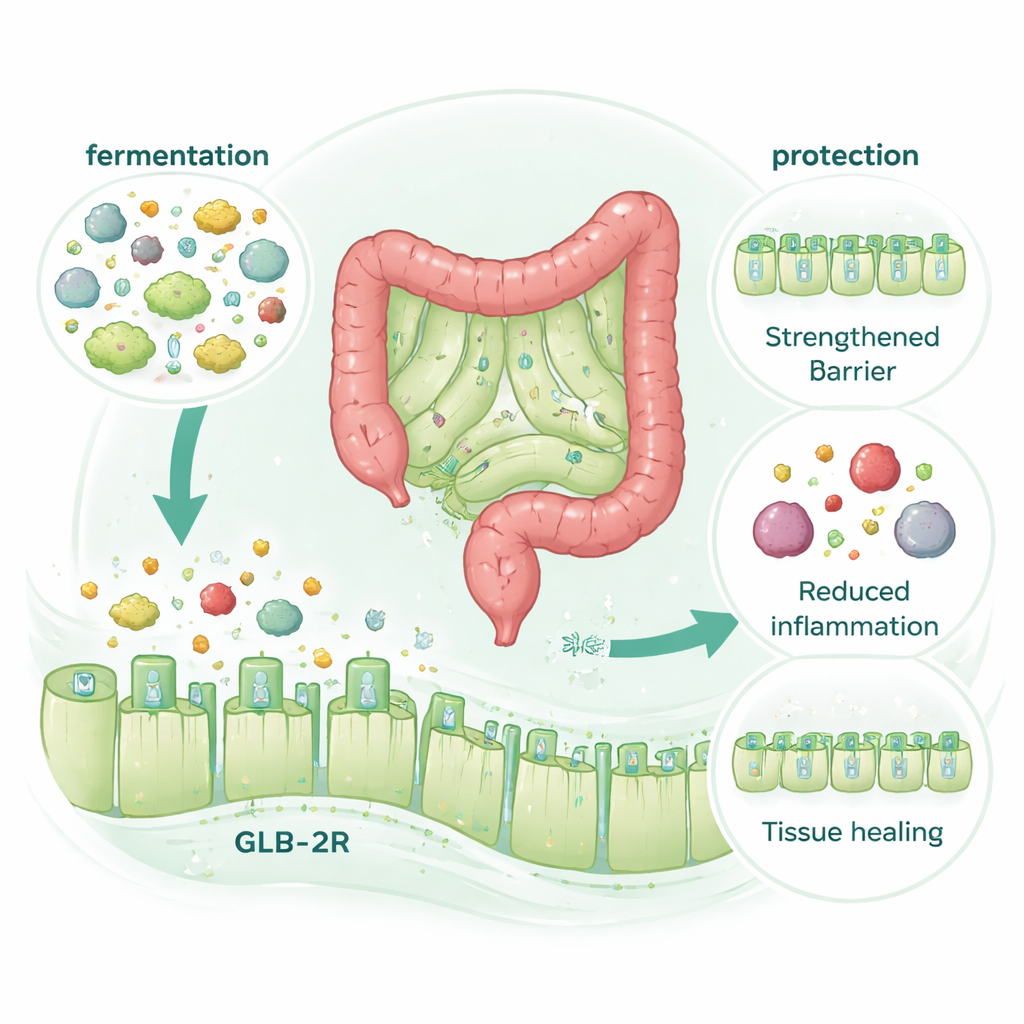
To see whether these changes translate into real-world protection, the researchers challenged mice with two harsh gut insults. One was a chemotherapy drug, 5‑fluorouracil, which causes widespread damage and shrinkage of the finger-like villi that absorb nutrients. The other was a chemical (DSS) that triggers colitis, a model of large-intestine inflammation. Mice on the McB diet lost less weight, maintained longer villi and colon length, showed fewer bleeding lesions, and had lower microscopic damage scores in both models. These benefits remained even when CD4 T cells were experimentally removed, suggesting that McB’s gut-protective actions are not solely due to adaptive immune cells but also involve direct effects on the intestinal lining.
A Hormone Receptor, but No Extra Hormone
A surprising twist was the discovery that McB’s protective effect absolutely required the gut’s receptor for a repair-promoting hormone called GLP‑2 (the GLP‑2 receptor, or GLP‑2R), but not the related GLP‑1 receptor. When mice lacked GLP‑2R, McB no longer shielded them from chemotherapy or colitis damage. Yet McB itself did not raise GLP‑2 levels in blood, even when digestion and hormone breakdown were carefully controlled. Instead, the benefits disappeared when microbial fermentation in the colon was chemically blocked, even though overall microbiota composition changed very little. This points to a mechanism in which microbial breakdown of McB generates small compounds that behave like GLP‑2 “mimics,” activating its receptor to strengthen the barrier and promote repair without extra hormone release.
What This Could Mean for Future Foods
To a lay reader, the take-home message is that some future protein sources might be designed not only to be climate-friendly, but also to actively support gut health. In mice, a commercially scalable McB lysate both rewired immunity toward tolerance and helped the intestine withstand severe injury, through two partially independent routes: a direct boost of regulatory T cells and a fermentation-driven activation of GLP‑2 receptors that aids tissue repair. While human studies are still needed, the work suggests that microbe-derived proteins could form a new class of "smart" foods—able to feed us, collaborate with our own microbes, and quietly reinforce the gut’s natural defenses against inflammation.
Citation: Yang-Jensen, S.K., Choi, B.SY., Nägele, N.S. et al. Microbial activation of the GLP-2R mitigates gastrointestinal inflammation. Nat Commun 17, 1839 (2026). https://doi.org/10.1038/s41467-026-68551-9
Keywords: gut inflammation, microbial protein, microbiome, GLP-2 receptor, regulatory T cells