Clear Sky Science · en
An open decoupled cell design achieving electricity generation and amplification through waste-to-energy conversion
Turning Wastewater into Extra Power
Imagine a battery that not only stores electricity, but also cleans up chemical waste and gives you back more electrical energy than you put in. This study introduces just such a concept: an "open" battery that feeds on industrial waste chemicals and salty solutions to boost its output. For anyone interested in cheaper, cleaner energy and smarter use of waste, this work hints at a new way to power our homes and grids while cutting both pollution and costs.
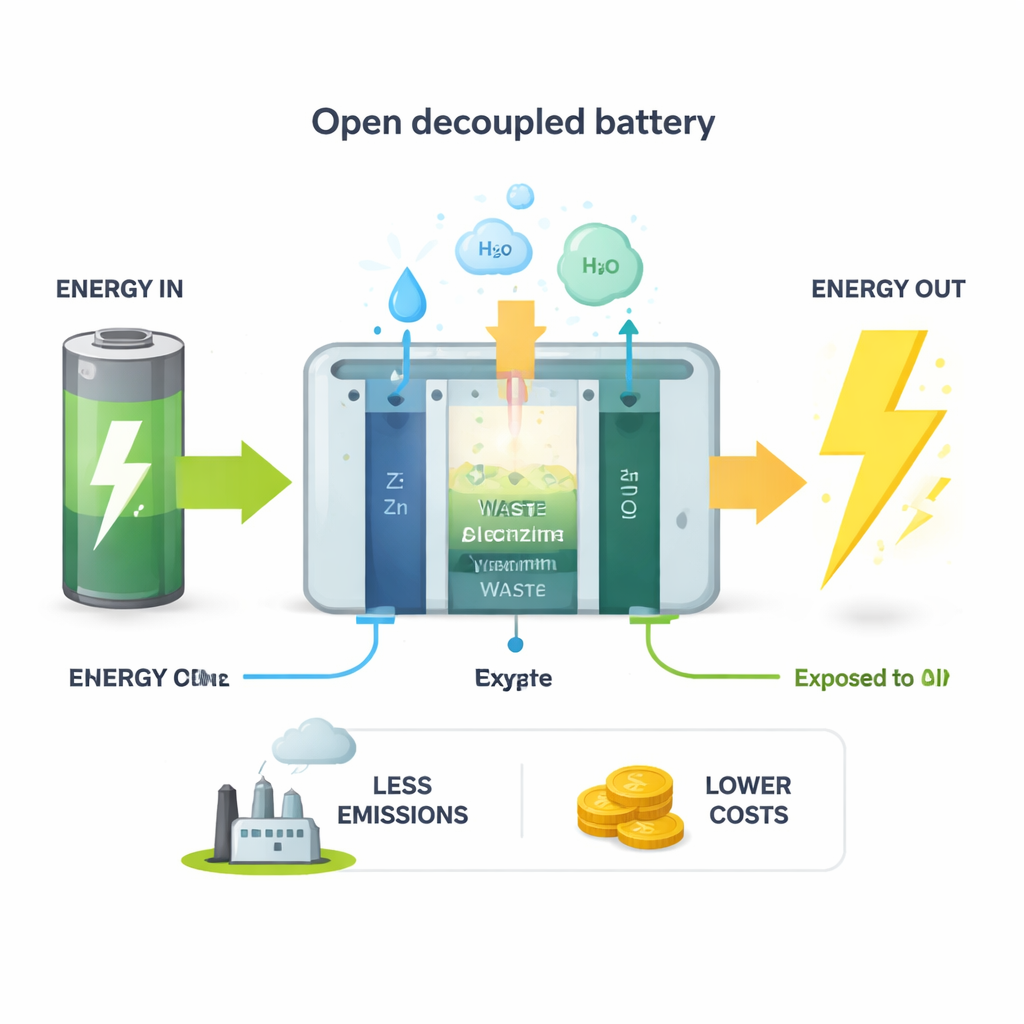
Why Regular Batteries Hit a Hard Limit
Conventional batteries are closed boxes. They can trade energy with the outside world, but no fresh material is allowed in or out during operation. Because of that, the best they can ever do is return roughly the same electrical energy that you feed them, minus some unavoidable losses. Their electrical energy efficiency is capped at 100% by design. That means every time you charge and discharge, you slowly lose stored electricity and must generate more somewhere else, often from fossil fuels. As grids fill up with solar and wind power, this limit becomes a costly bottleneck.
Opening the Battery to the World
The researchers propose an "open decoupled battery" that breaks this 100% barrier by taking in cheap or free materials from the environment. Instead of two electrodes and one shared liquid, they split the system into three key parts: a zinc metal electrode that shuttles charge, a positive electrode that breathes in oxygen from air during discharge, and a separate positive electrode that consumes a waste chemical—hydrazine—during charging. They also separate the liquids on each side and let dissolved salts create an extra voltage through a process called reverse electrodialysis, which taps the energy difference between concentrated and dilute solutions. Together, these three voltage sources form what the authors call a "3E" design.
How the New Cell Makes More than It Takes
In this design, the battery charges at a relatively low voltage because zinc is re-formed while hydrazine in wastewater is oxidized, a reaction that naturally wants to release energy. Discharging happens at a much higher voltage, as zinc is consumed and oxygen from the air is reduced. On top of that, the salt-concentration difference across a special membrane contributes extra push in the discharging direction. Because the output voltage is several times larger than the input voltage, the device can deliver more electrical energy than the electricity used to charge it—up to about 4.5 times at low current in the alkaline version, and even higher in an acidic variant. In large-scale tests, a 20-ampere-hour prototype ran stably and showed that such cells can be engineered at practical sizes.
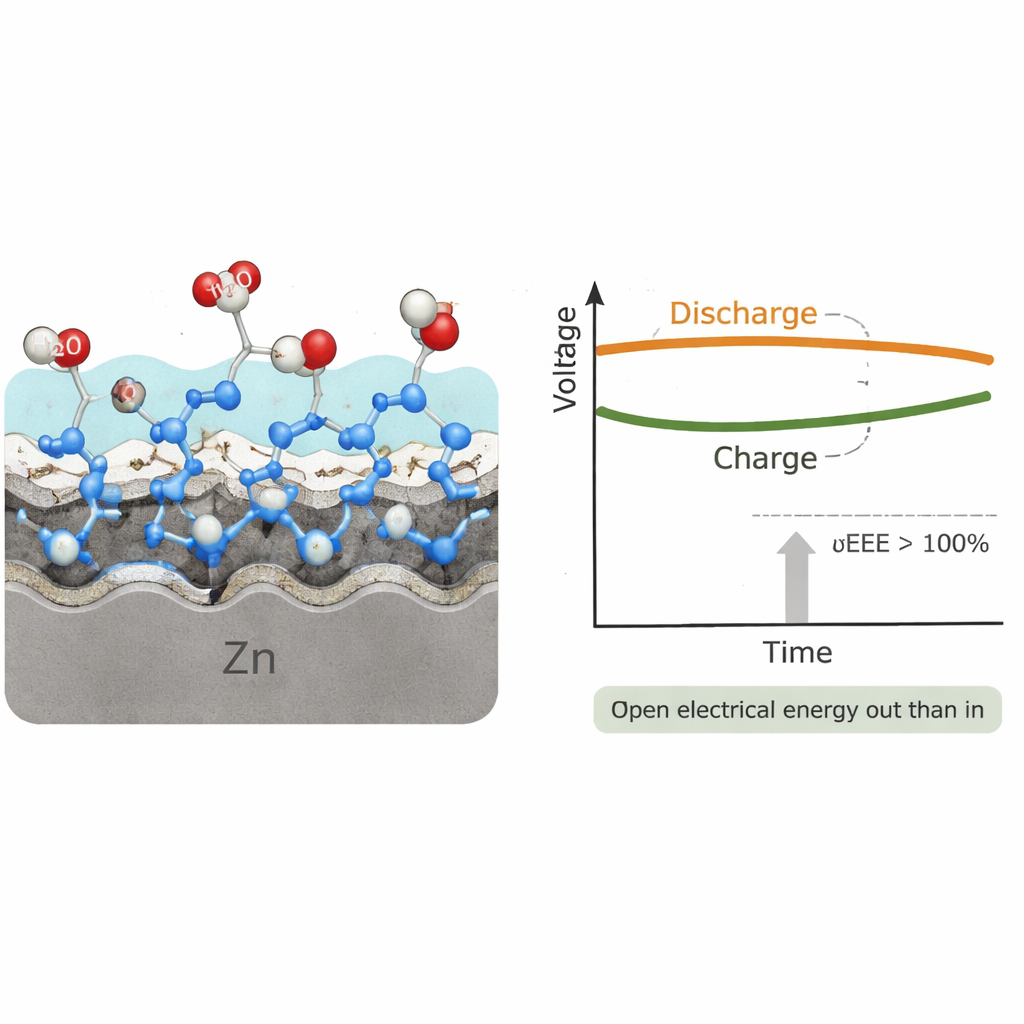
Protecting the Zinc and Extending Lifetime
A key challenge with zinc batteries is that the metal tends to corrode and dissolve, wasting material and shortening life. The team discovered that hydrazine does double duty: it is not only a fuel-like waste to be removed, but it also helps protect the zinc surface. Detailed computer simulations and in situ measurements show that hydrazine molecules stick to the zinc and rearrange the local electrons in a way that makes it harder for water to split, hydrogen gas to form, and zinc atoms to escape into the liquid. This "corrosion trilogy"—water splitting, gas formation, and metal loss—is slowed down, so the zinc can be used more deeply while the cell continues to operate for over a thousand hours and cycles under fast-charging conditions.
Cheaper, Cleaner Energy Systems
Because this open battery can return far more electrical energy than it absorbs from the grid, it acts like an electricity amplifier tied to waste treatment. Techno-economic analyses suggest that, for every megawatt-hour of electricity stored, the amount of power that must be generated upstream can drop by more than 80% compared with familiar storage systems like lithium-ion or lead-acid batteries. At the same time, using the cell to break down hydrazine wastewater costs much less than standard chemical treatments and sharply cuts carbon emissions when combined with solar, wind, or even natural gas plants. In simple terms, the authors show a path toward batteries that do not just lose energy slowly, but instead grow it, while cleaning up industrial streams—a potential shift in how we think about both energy storage and waste management.
Citation: Zheng, Z., Zheng, FY., Huang, B. et al. An open decoupled cell design achieving electricity generation and amplification through waste-to-energy conversion. Nat Commun 17, 1838 (2026). https://doi.org/10.1038/s41467-026-68550-w
Keywords: waste-to-energy, zinc batteries, energy storage, hydrazine wastewater, electricity efficiency