Clear Sky Science · en
Ether lipids influence cancer cell fate by modulating iron uptake
How a Little-Known Fat Could Expose Cancer’s Weak Spot
Cancer is often described as a “disease of genes,” but this study shows that the fats making up a cell’s outer skin can be just as important. The researchers uncover how a special class of fats, called ether lipids, help dangerous cancer cells pull in iron from their surroundings—fueling both their ability to spread and their vulnerability to a newly recognized form of cell death. Understanding this hidden connection between cell membranes, iron, and tumor behavior could open doors to therapies that selectively wipe out the most aggressive cancer cells.
A Different Way to Think About Cancer Cells
Most cancer research focuses on mutations in DNA and the proteins they encode. This work shifts attention to the cell membrane—the thin oily shell that surrounds every cell. Within that shell live many kinds of lipids (fats), each with distinct shapes and physical properties. The authors focus on ether lipids, which make up about one-fifth of the phospholipids in many mammalian cells but have been relatively understudied. They find that highly metastatic, stem‑like cancer cells pack their membranes with ether lipids, giving those membranes low tension and high fluidity. These physical traits, in turn, change how the cells import iron, a metal that can drive both tumor growth and a destructive process called ferroptosis, an iron‑dependent form of cell death.
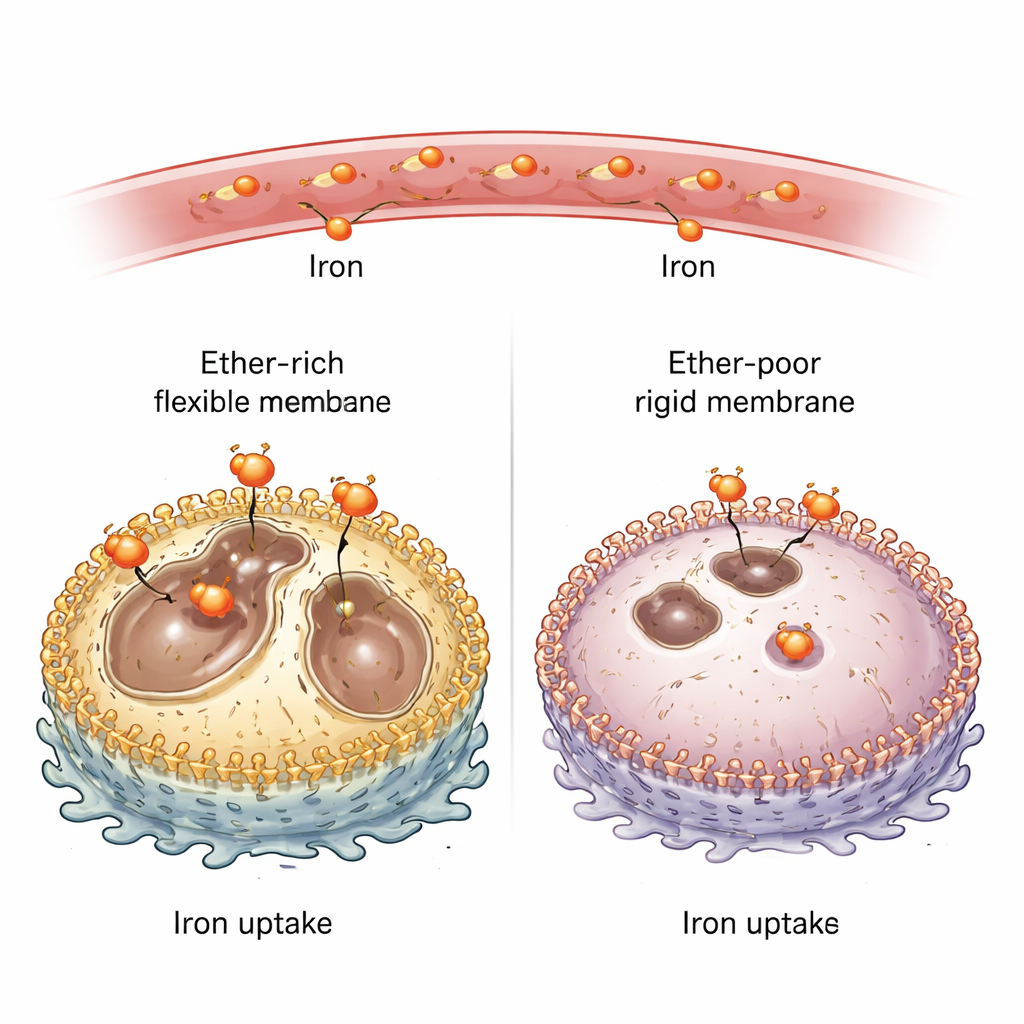
Connecting Membrane Fats to Iron Intake
To probe ether lipids, the team deleted key enzymes required to make them in several mouse and human cancer cell lines. Without these enzymes, ether lipids in the plasma membrane plummeted from roughly a quarter of all lipids to just a few percent. Surprisingly, these ether‑poor cells carried much less “redox‑active” iron—the chemically reactive form that participates in oxidation reactions—both in culture and in implanted tumors in mice. When the scientists supplied extra iron from the outside, the cells’ iron levels rose again and they regained their sensitivity to ferroptosis‑inducing drugs. This showed that ether lipids do more than simply serve as burnable fuel for oxidation; they actively control how much iron gets inside the cell.
A Specialized Iron Gate: The CD44 Pathway
The researchers then asked how membrane composition changes iron uptake. Cells typically import iron through a well‑known protein called the transferrin receptor, but aggressive, stem‑like cancer cells also rely on another route: a surface protein named CD44 that pulls in iron bound to a sugar molecule called hyaluronan. By tracking fluorescent probes, the team showed that knocking out ether lipids barely affected transferrin receptor uptake but sharply slowed CD44‑mediated endocytosis—an alternate, clathrin‑independent route of bringing material into the cell. Restoring ether lipids, or adding them back in liposome form, revived this CD44 pathway. Disrupting CD44 itself or digesting hyaluronan lowered cellular iron, confirming that this ether‑dependent route is a major iron gate in these cancer cells.
How Softer Membranes Change Cell Behavior
Why do ether lipids matter so much for CD44 but not for the classic iron receptor? The answer lies in physics. Using precision tools that pull tiny tethers from the cell surface, the authors showed that ether‑rich membranes have lower tension and are easier to deform. When ether lipids were removed, membrane tension increased and the lipids packed more tightly, making the membrane stiffer. These changes strongly hindered forms of endocytosis that depend on flexible, raft‑like membrane patches—exactly the kind CD44 uses—while leaving clathrin‑based uptake largely intact. Ether lipids also helped stabilize “lipid rafts,” tiny ordered islands in the membrane where CD44 clusters before it is pulled inward. In short, ether lipids tune the physical landscape of the membrane to favor CD44‑driven iron uptake.
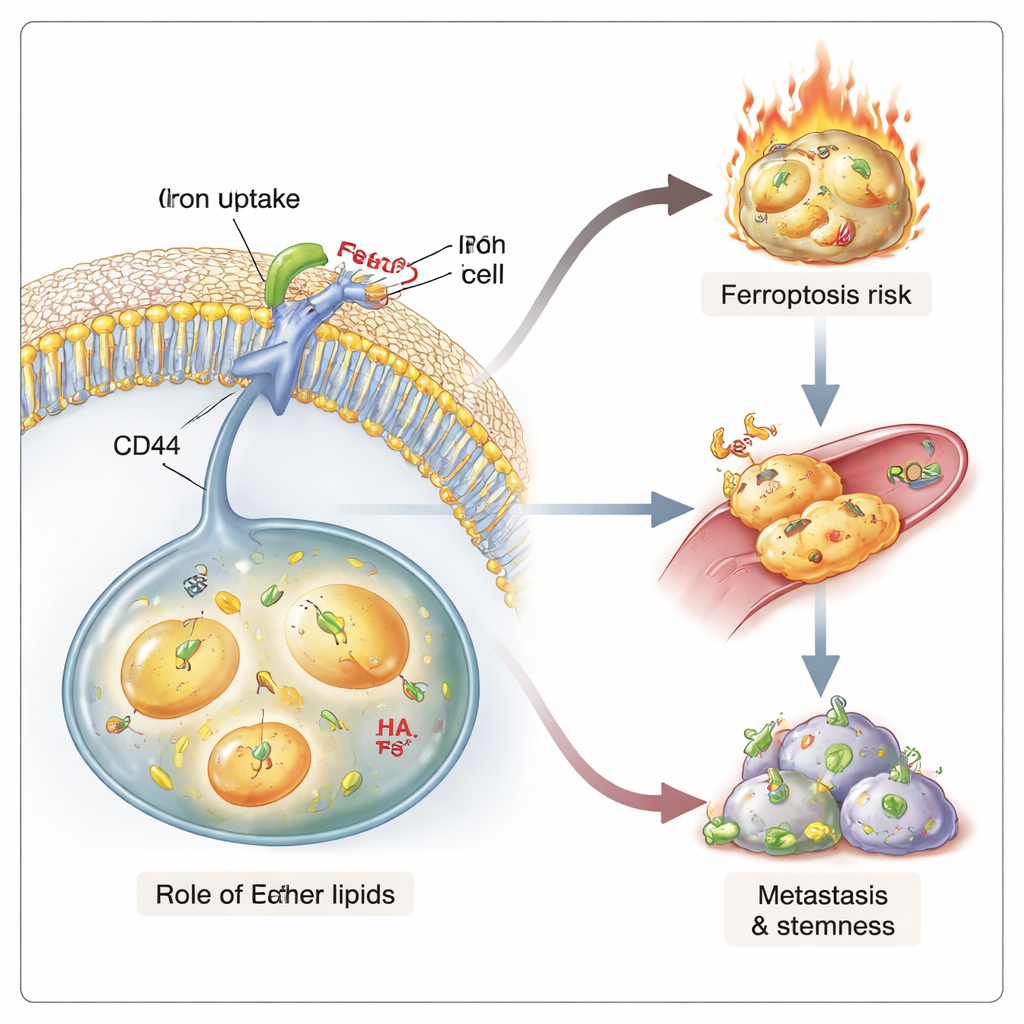
Fueling Both Spread and Self‑Destruction
High iron levels and low membrane tension are linked to traits that make cancers deadly: the ability to seed new tumors and to survive therapy. In three‑dimensional microvessel models, ether‑rich cancer cells more readily squeezed out of blood‑vessel‑like structures—an early step in metastasis. In mice, ether‑deficient cancer cells formed fewer metastases, even though their primary tumors grew at similar rates. These cells also showed reduced “stemness,” measured by their diminished capacity to form mammospheres in culture and to initiate tumors in limiting‑dilution implants. At the same time, ether‑rich cells, with their higher iron load and more oxidizable lipids, were more susceptible to ferroptosis when challenged with specific drugs or immune cues. Thus, ether lipids create a double‑edged state: they support aggressive, stem‑like behavior while rendering those same cells uniquely killable through iron‑driven lipid damage.
Why This Matters for Future Cancer Treatments
For non‑specialists, the key message is that not all cancer cells are equally dangerous, and their outer membranes help determine both how threatening and how vulnerable they are. Ether lipids reshape the cell surface so that iron can flow in through a specialized doorway, energizing metastatic, stem‑like cells but also loading them with the ingredients for their own destruction. Therapies that alter ether lipid production, disrupt CD44‑based iron uptake, or carefully trigger ferroptosis could selectively target these hard‑to‑treat cancer cell populations while sparing normal tissue. By revealing how a little‑known class of fats links membrane physics, metal metabolism, and cell fate, this study opens a new path toward smarter, more precise cancer interventions.
Citation: Mansell, R.P., Müller, S., Yang, JS. et al. Ether lipids influence cancer cell fate by modulating iron uptake. Nat Commun 17, 1835 (2026). https://doi.org/10.1038/s41467-026-68547-5
Keywords: ether lipids, cancer stem cells, iron uptake, ferroptosis, metastasis