Clear Sky Science · en
Genome-wide discovery and phenotyping of non-coding transcripts in A. fumigatus reveals lncRNAs with a role in antifungal drug sensitivity
Why a mould in our homes matters
Most of us think of mould as a nuisance on bread or in damp corners, but one common mould, Aspergillus fumigatus, is responsible for more than two million deaths every year—more than malaria and HIV combined. Doctors rely on a narrow arsenal of antifungal drugs to keep this pathogen in check, and resistance to these drugs is rising fast. This study explores a hidden layer of the fungus’s genome—long non‑coding RNAs, or lncRNAs—that do not make proteins but can still influence how the fungus responds to treatment. Understanding these “silent” genetic elements could open new ways to predict, track, and eventually counter antifungal resistance.
Hidden messages in fungal DNA
Classically, genes have been thought of as stretches of DNA that code for proteins, the workhorses of the cell. Over the last decade, scientists have discovered that vast regions of the genome are transcribed into RNA molecules that never become proteins. These long non‑coding RNAs can still affect how cells behave, including how they respond to drugs. While lncRNAs have been mapped in humans and in yeast, they were largely uncharted in disease‑causing moulds such as A. fumigatus. The authors set out to change that by building a genome‑wide catalogue of these mysterious transcripts and asking whether any of them tip the balance between drug sensitivity and resistance.
Listening to the fungus under drug attack
To uncover lncRNAs, the team exposed A. fumigatus to six different antifungal compounds, including widely used azoles that target the fungal cell membrane, and then sequenced all the RNAs produced. Using a custom bioinformatics pipeline, they assembled tens of thousands of transcripts and systematically stripped away anything corresponding to known protein‑coding genes or short housekeeping RNAs. After multiple rounds of filtering and manual curation, they arrived at a high‑confidence set of 1,089 new long non‑coding RNAs spread across the genome. Most lay either between known genes or overlapped them in the opposite direction, and together they expanded the fraction of the fungal genome known to be actively transcribed from roughly two‑thirds to more than four‑fifths.
Coordinated responses and conserved hotspots
When the researchers compared how these lncRNAs changed under different drug doses, they found that the fungus does not deploy them randomly. Instead, lncRNAs fell into about 15 distinct response patterns, some shared across multiple drugs and others unique to particular treatments. For example, drugs hitting similar biochemical pathways tended to trigger overlapping lncRNA signatures, while a protein‑synthesis inhibitor produced many unique responses. Many lncRNAs sat very close to genes already known to influence azole sensitivity, such as those involved in iron capture or the biosynthesis of ergosterol, a key component of fungal cell membranes. In several cases, a nearby lncRNA and a drug‑response gene were switched up or down together, hinting that these non‑coding elements may help coordinate crucial survival programs.
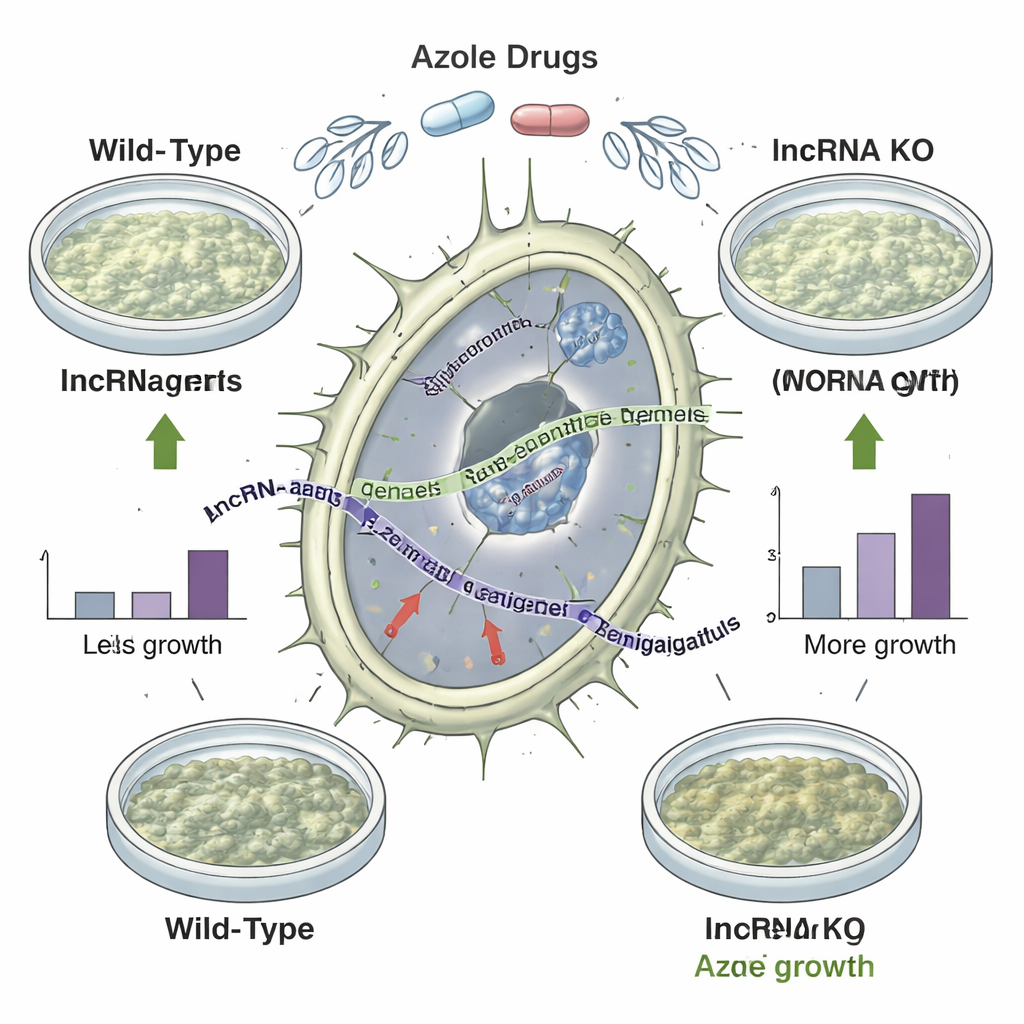
Knocking out “silent” genes changes drug sensitivity
Cataloguing lncRNAs is one thing; proving they matter is another. The team deleted 92 selected lncRNA regions from the fungal genome and compared how the mutants grew under a range of stresses, including high temperature, low iron, and exposure to three different azole drugs. Sixty mutants showed condition‑specific changes in fitness, and 35 actually grew better than the original strain when challenged with azoles. One standout deletion strain displayed improved growth across all tested azole drugs without simply boosting nearby protein‑coding genes, strongly suggesting that the missing lncRNA itself was restraining drug tolerance. Looking across many clinical and environmental isolates with known drug susceptibility profiles, the authors also found that the presence or absence of certain lncRNA genes tracked with how easily each strain was inhibited by azoles.
What this means for fighting deadly fungal infections
For non‑specialists, the key message is that parts of the fungal genome once written off as “junk” are actively shaping how dangerous A. fumigatus can be and how well our drugs work against it. By building the first comprehensive map of long non‑coding RNAs in this major pathogen and tying dozens of them to measurable changes in drug response, this work opens up a new class of genetic markers and potential targets. In the long run, lncRNAs could help explain why some strains are naturally harder to treat, guide the design of more effective diagnostics, and inspire therapies that disarm resistance not by killing the fungus outright, but by silencing the silent regulators that help it endure.
Citation: Weaver, D., Qi, T., Chown, H. et al. Genome-wide discovery and phenotyping of non-coding transcripts in A. fumigatus reveals lncRNAs with a role in antifungal drug sensitivity. Nat Commun 17, 1832 (2026). https://doi.org/10.1038/s41467-026-68543-9
Keywords: Aspergillus fumigatus, antifungal resistance, long non-coding RNA, azole drugs, fungal genomics