Clear Sky Science · en
Unravelling the nucleation–elongation mechanism of one-pot catenation
Why tiny mechanical links matter
At first glance, molecules that loop through one another like links in a chain may sound like chemical curiosities. But these "catenanes"—interlocked molecular rings and cages—are the building blocks of future molecular machines, advanced materials and nanoscale devices. To harness them, chemists need to do more than make them once in a flask; they must understand, and ultimately control, how these intricate structures grow from simple ingredients. This article uncovers how certain cage-shaped molecules thread and lock together in a single reaction mixture, revealing general rules that could make complex molecular links easier and faster to build on demand.
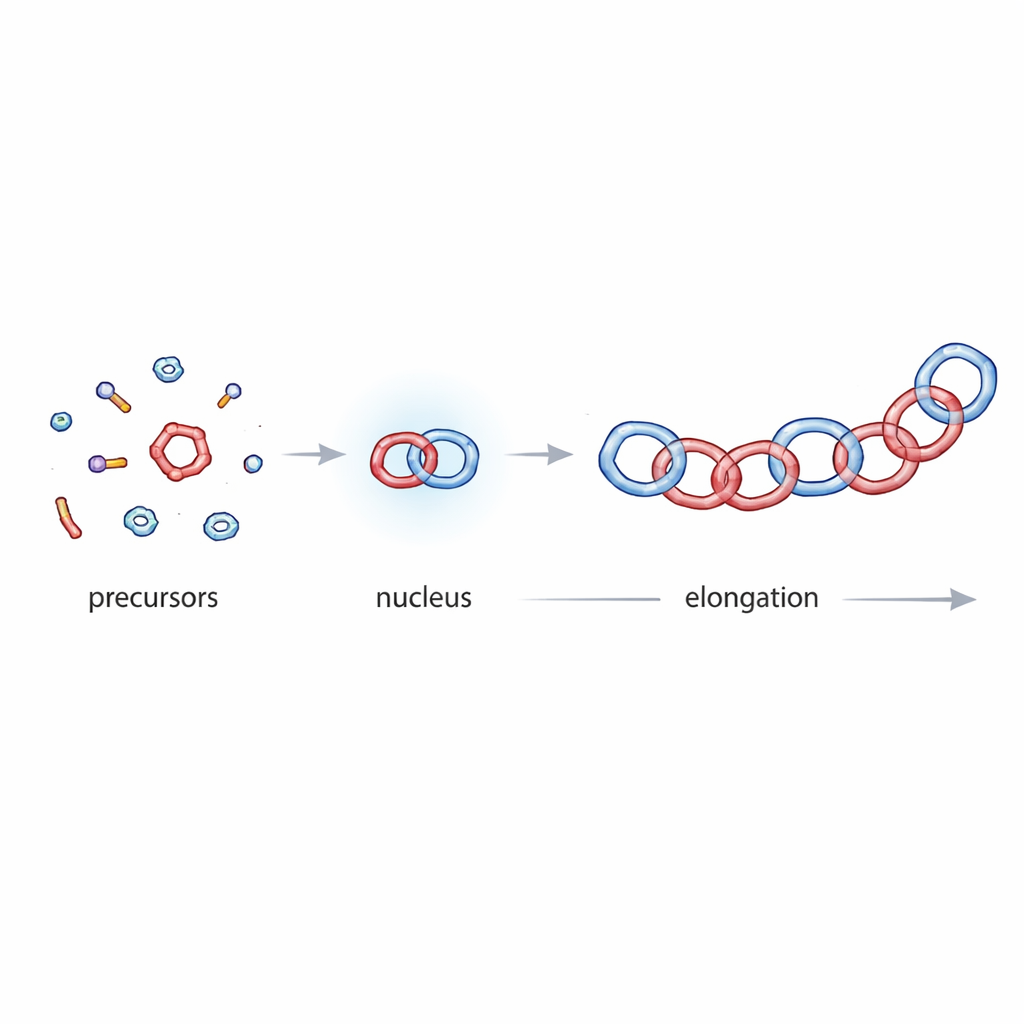
From loose pieces to locked links
The researchers focused on catenanes built from rigid, cage-like molecules rather than simple rings. Each cage is assembled from flat aromatic "panels" and flexible linkers that snap together through reversible chemical bonds. Under the right conditions, these cages can slip through one another and lock, much like two key rings interlinked. The team studied two target structures: a dimeric cage-catenane (two cages interlocked, called DCC) and a trimeric cage-catenane (three cages interlocked, called TCC). Both form in what chemists call a "one-pot" process, meaning all building blocks and catalyst are mixed at once, and the system is left to organize itself into the final interlocked products.
A familiar growth pattern from biology
To decode how these catenanes appear, the authors borrowed ideas from the way protein fibrils and supramolecular polymers grow. Those systems often follow a nucleation–elongation mechanism: first, a small but rare nucleus forms (nucleation), then additional units quickly add on (elongation), producing a characteristic S-shaped growth curve and an initial lag time. By carefully monitoring the reaction using nuclear magnetic resonance spectroscopy across many concentrations, the team showed that DCC and TCC follow the same general pattern. Both display a sharp "critical concentration" below which little catenane forms and above which growth suddenly becomes efficient, a hallmark of nucleation–elongation behavior.
Two growth routes for two kinds of chains
Despite this shared framework, DCC and TCC grow in strikingly different ways. For DCC, a single type of monomeric cage, dubbed MC-1, plays a starring role. MC-1 binds the flat panel components very strongly, acting as a powerful template. Once enough MC-1 forms, it captures an extra panel, helping two cages interlock and rapidly elongate into the dimeric catenane. When the researchers added a small amount of purified MC-1 to a fresh reaction (a "seeded" experiment), the usual lag phase nearly vanished and DCC appeared much faster, directly confirming MC-1 as an efficient nucleus for growth. Even pre-made DCC could speed up its own formation via an autocatalytic process, though less effectively than MC-1.
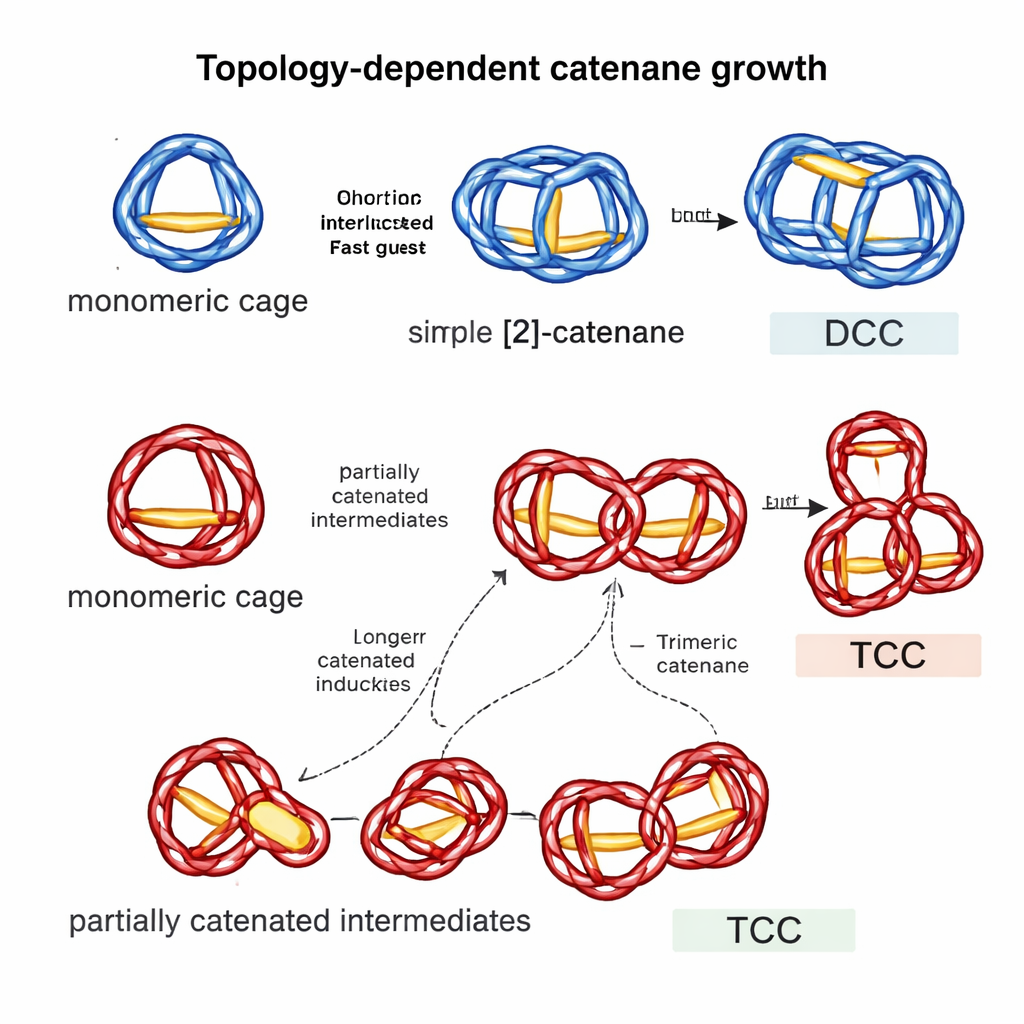
Complex links need more complex beginnings
The trimeric cage-catenane TCC told a subtler story. Its monomeric cage, MC-2, binds panels more weakly overall, so it does not serve as a strong nucleus in the same way. Kinetic experiments again showed a lag phase and critical concentration, but seeding the mixture with MC-2 did not shorten the lag at all. Only when the team added a small amount of preformed TCC did the induction period shrink, suggesting that elusive, partially interlocked intermediates—rather than simple monomeric cages—are the key stepping stones to the final three-cage structure. Mass spectrometry supported the existence of these species, even though they are too unstable to isolate. By comparing binding strengths and mapping out plausible routes, the authors proposed that TCC forms through several parallel pathways that all rely on these partially catenated intermediates to drive elongation.
Design rules for future molecular chains
By untangling these microscopic pathways, the study shows that not all interlocked molecules grow alike: the topology—whether two cages or three, and how they weave—changes which intermediates act as nuclei and how easily growth proceeds. The authors also introduce a simple dimensionless parameter that captures how difficult nucleation is and how strongly it can be tuned by changing concentration or adding seeds. For non-specialists, the takeaway is that chemists are learning to treat molecular links much like polymers or protein fibers, with controllable starts and growth rates. This mechanistic insight opens the door to rationally designing more elaborate chains and networks of catenanes, which could underpin future smart materials and nanoscale machines built from precisely interlocked molecular parts.
Citation: Chen, Z., Lv, X., Xue, N. et al. Unravelling the nucleation–elongation mechanism of one-pot catenation. Nat Commun 17, 1830 (2026). https://doi.org/10.1038/s41467-026-68541-x
Keywords: catenanes, self-assembly, supramolecular polymerization, molecular machines, dynamic covalent chemistry