Clear Sky Science · en
Nonequilibrium pulsed heating freezes sintering of supported metal nanocatalysts
Why tiny metal particles matter for everyday technology
From clean energy to making chemicals and cleaning exhaust, much of modern technology relies on catalysts: materials that speed up reactions without being used up. Many of the best catalysts are made of metal nanoparticles—ultra‑small bits of metal—sitting on a solid support. These particles work so well because they have a huge surface area. But there is a serious problem: at high temperature they tend to clump together into larger chunks, losing their special properties. This study shows that by heating them in very fast pulses instead of slowly, we can largely stop this clumping and create tougher, longer‑lasting catalysts.
How heat quietly ruins powerful catalysts
Traditional catalyst manufacturing, and many real‑world reactions, require heating metal nanoparticles for long periods to high temperatures. Under these conditions, small particles wander across the support and merge—a process called sintering. As they merge, the total surface area shrinks and the catalyst becomes less effective. This is a major barrier to using precious metals like platinum efficiently in fuel cells, pollution control, and chemical plants, because a great deal of expensive metal can become underused once it forms big, inactive lumps.
A new way to heat: fast pulses instead of slow baking
The researchers explored a very different heating strategy known as ultrafast pulsed heating. Instead of slowly ramping the temperature up and holding it there, they repeatedly flashed the temperature of a platinum‑on‑graphene sample up to about 1000 °C for just 50 thousandths of a second, then cooled it back down very quickly. Using an electron microscope that can look inside materials while they are being heated, they watched how the nanoparticles formed and moved on the surface in real time. They compared this pulsed approach to a conventional, slow heating schedule reaching the same peak temperature but over many hundreds of seconds.
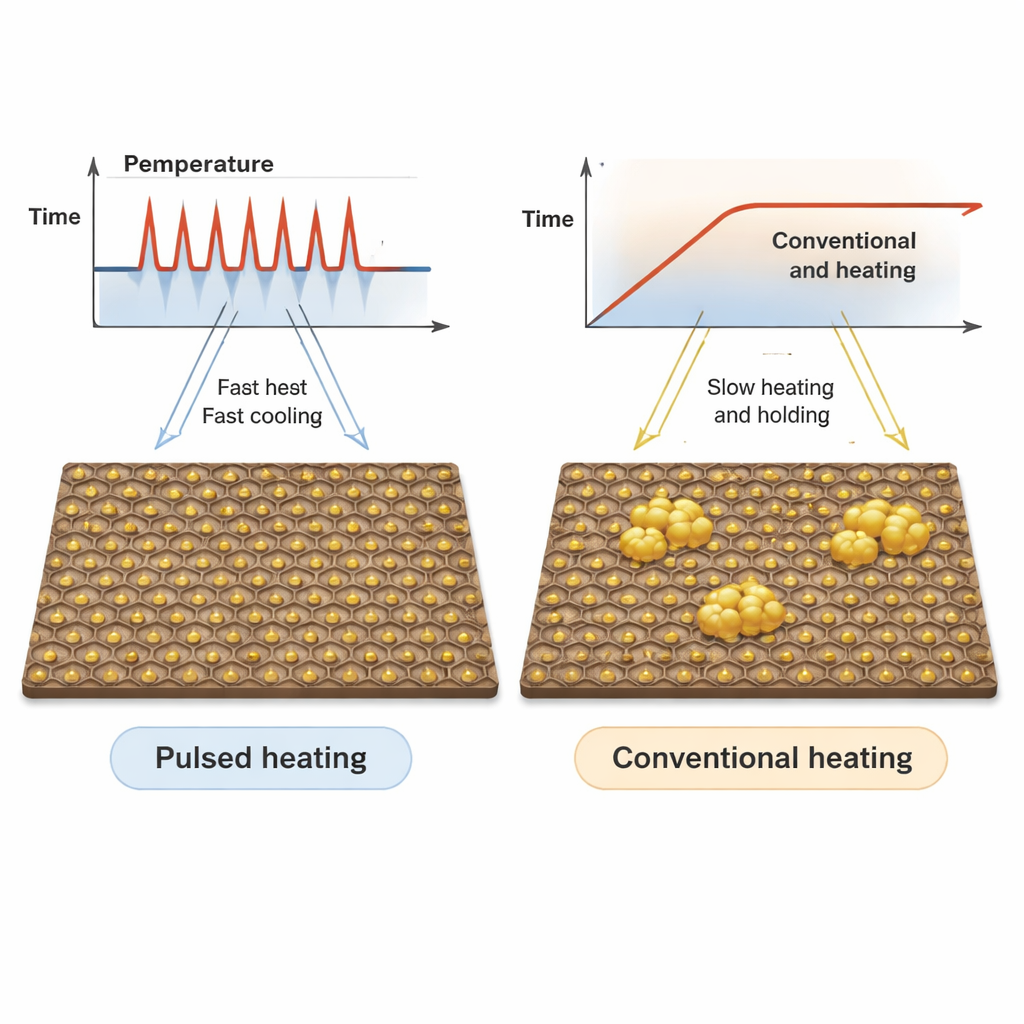
What they saw when particles faced pulsed versus slow heat
Under pulsed heating, the platinum precursor quickly broke down into many tiny nanoparticles less than 3 nanometers across, spread uniformly over the graphene. Even after ten pulses, most particles stayed small and well separated, and after one hundred pulses they still showed only slight growth. By contrast, with conventional heating the number of visible particles dropped sharply while the remaining ones grew much larger, clear evidence of sintering and even evaporation of the smallest clusters. Careful measurements confirmed that, although both methods produced well‑ordered crystal structures, the pulsed approach gave a narrower size distribution and far better resistance to coalescing.
Locking nanoparticles into a sweet spot
Beyond size, the team examined how the atomic structure and the contact between platinum and graphene changed. With repeated pulses, the particles gradually reshaped from irregular blobs into neatly faceted, hexagon‑like crystals whose orientation lined up with the underlying graphene lattice. Electron spectroscopy showed that the electronic signature of the carbon support shifted, a sign of stronger bonding and charge sharing between platinum and graphene. Computer simulations backed this up: they suggested that pulsed heating keeps the system in a “metastable” state—one that is not the lowest‑energy arrangement overall, but is protected by kinetic barriers because the particles never stay hot long enough to move very far. Slow heating, in contrast, gives atoms plenty of time to diffuse, merge, and spread across the surface.
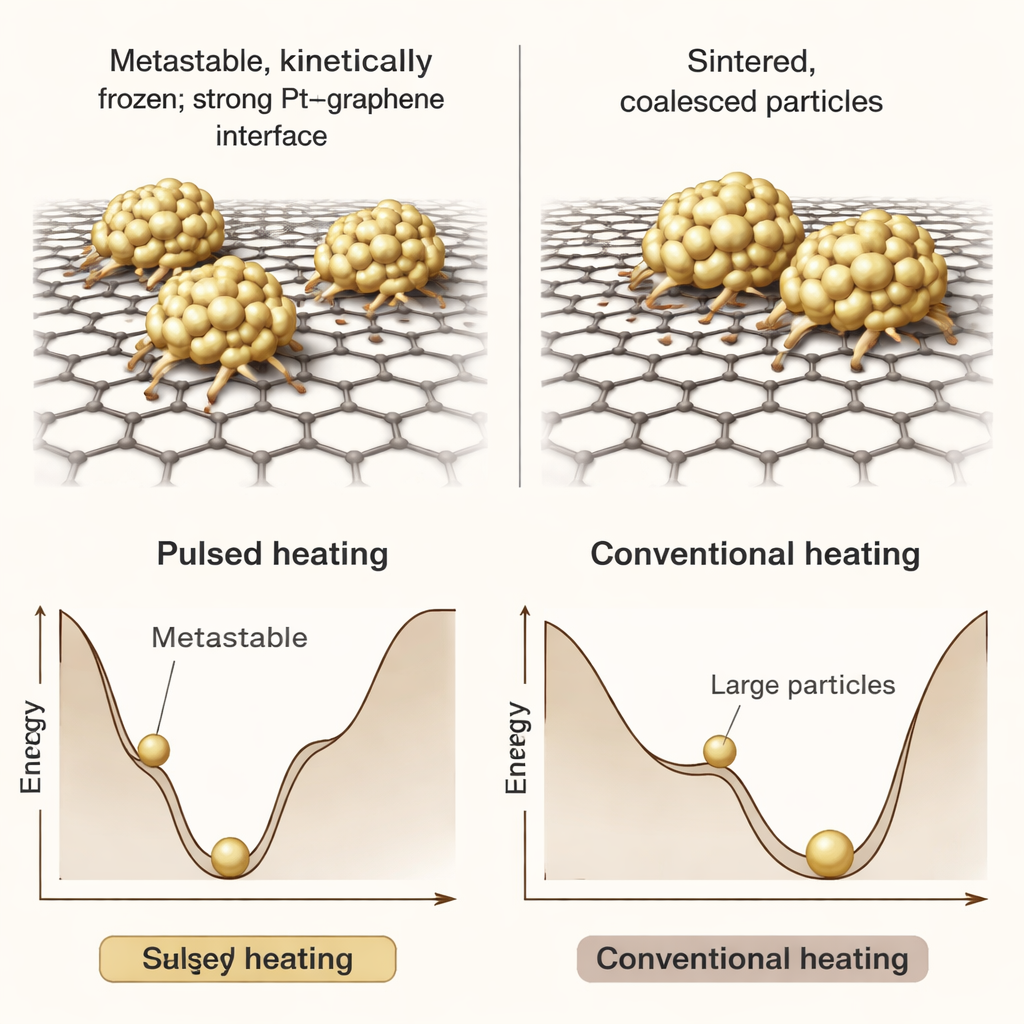
Why this matters for real‑world catalysts
In simple terms, pulsed heating works like quickly toasting bread and then pulling it out before it burns: the nanoparticles get enough energy to organize themselves and bond firmly to the support, but not enough time at high temperature to wander around and clump together. The end result is a dense layer of ultra‑small, highly crystalline platinum particles that are strongly attached to graphene and can survive even prolonged high‑temperature exposure without serious sintering. This nonequilibrium route could be applied broadly to make tougher catalysts that use less precious metal, last longer, and perform better in demanding energy and chemical processes.
Citation: Huang, J., Zhang, Z., Wang, G. et al. Nonequilibrium pulsed heating freezes sintering of supported metal nanocatalysts. Nat Commun 17, 1828 (2026). https://doi.org/10.1038/s41467-026-68539-5
Keywords: nanocatalysts, pulsed heating, platinum nanoparticles, sintering resistance, graphene support