Clear Sky Science · en
Ependymoglial cells are critical for cortex regeneration in axolotls
How Some Animals Rebuild Their Brains and Spines
Most people know that a broken bone can heal, but few realize that some animals can rebuild far more complex body parts, including parts of the brain and spinal cord. This study looks at the axolotl, a salamander famous for regrowing limbs, and asks a striking question: which exact cells let it repair its central nervous system—and can we turn that knowledge into a tool to study regeneration in detail?
The Salamander That Refuses to Scar
Axolotls can regrow lost limbs, tails, and even large portions of their brain and spinal cord. Instead of forming permanent scars, their tissues reorganize and rebuild. But regeneration is not magic: it depends on specific cell types taking on specific jobs. In the brain and spinal cord, one key player is a population of support cells called ependymoglial cells. These cells line fluid-filled spaces and normally help maintain the nervous system. Researchers suspected they might also act as stem cells, creating new neurons after injury. Until now, however, there was no precise way to remove only these cells in living axolotls to test just how essential they are.
A Genetic Switch to Remove Chosen Cells
The team adapted a clever bacterial trick to axolotls. They engineered animals so that certain cell types produce an enzyme called nitroreductase. On its own, the enzyme is harmless. But when the axolotl is bathed in a matching “prodrug” compound, the enzyme converts that compound into a toxin—but only inside the marked cells, killing them while leaving neighbors intact. By linking the enzyme to genetic switches that turn on only in chosen cells, the scientists could selectively erase those cells on demand. They created several axolotl lines: some in which ependymoglial cells glowed red and carried the enzyme, and others in which specific cortical neurons did the same.
Proving Which Cells Truly Rebuild the Nervous System
With this system in place, the researchers asked what happens if ependymoglial cells are removed before injury. Using a refined drug, they were able to nearly wipe out these cells in the spinal cord and telencephalon (the front part of the brain), without harming surrounding support cells or muscle stem cells. When they then injured the spinal cord or brain, regeneration simply failed. The spinal cord did not regrow into the tail, the brain injury filled with scar-like tissue instead of new neurons, and the usual burst of dividing cells at the wound site was almost completely absent. In transplanted “chimera” animals, where only donor tissue carried the sensitive cells, deleting ependymoglial cells in that region alone was enough to block local repair. These experiments show that these cells are not just helpful—they are the main, and possibly the only, source of new nerve cells after injury in the axolotl central nervous system.
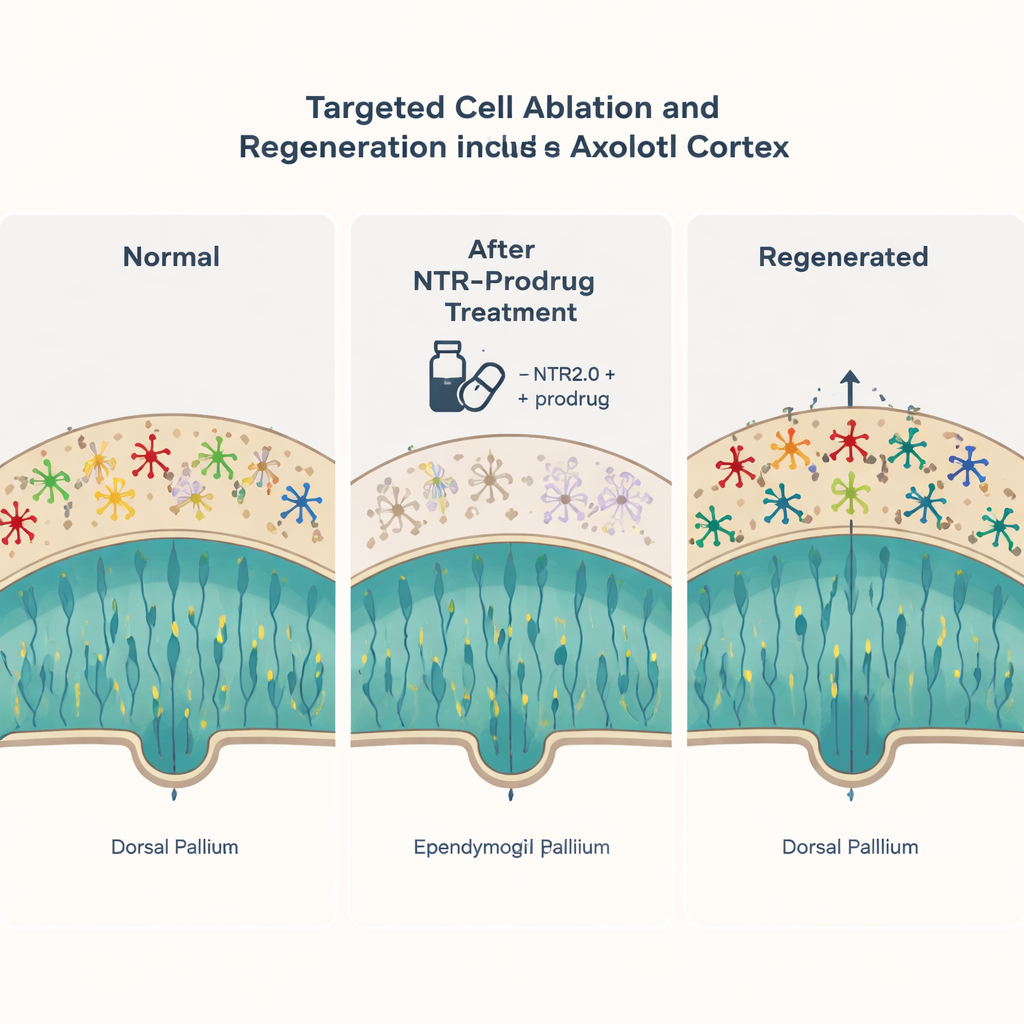
Erasing and Rebuilding the Cortex
The scientists next turned to large-scale loss of neurons, similar to what happens in neurodegenerative diseases. Initial attempts to remove cortical neurons with the original enzyme were inefficient, so they adopted a more powerful variant called NTR2.0. In animals engineered to express this stronger enzyme only in certain cortical neurons, a short treatment eliminated more than 95 percent of those neurons. The outer brain layer thinned dramatically, and the animals temporarily lost the ability to swallow food properly. Remarkably, over the following weeks and months, new neurons appeared in the same brain region, arising from the spared ependymoglial cells. By labeling cells born at different times, the researchers showed that these new neurons settled into an orderly “outside-in” pattern, matching how the axolotl cortex is built during development. Many distinct neuron subtypes were restored, and behavior recovered as the tissue rebuilt.
A Versatile Toolbox for Future Regeneration Research
To make their approach broadly useful, the team also created a flexible transgenic line in which the toxic enzyme can be switched on only in cells that express a separate “Cre” gene. Because many axolotl lines already exist with Cre active in different tissues, crossing them with this new line will allow researchers to delete almost any chosen cell type with a simple drug treatment. In plain terms, this study both proves that a single support-cell population underlies the axolotl’s ability to rebuild its brain and spinal cord, and delivers a precise on–off switch for removing specific cells. This combination will help scientists untangle how complex tissues regenerate and could eventually inform strategies to coax more limited human tissues to repair themselves.
Citation: Fu, S., Zeng, YY., Peng, C. et al. Ependymoglial cells are critical for cortex regeneration in axolotls. Nat Commun 17, 1827 (2026). https://doi.org/10.1038/s41467-026-68538-6
Keywords: axolotl regeneration, brain repair, spinal cord repair, stem-like glial cells, targeted cell ablation